CHOPIN Trial Publishes Phase I Results of Percutaneous Hepatic Perfusion with Immunotherapy
 Updated results from the Phase 1b CHOPIN Trial, conducted at Leiden University Medical Center on the use of the Delcath CHEMOSAT Hepatic Delivery System with Melphalan (CHEMOSAT) in combination with the immune checkpoint inhibitors (ICI) ipilimumab and nivolumab to treat patients with metastatic uveal melanoma with liver metastases, were published in Cardiovascular and Interventional Radiology.
Updated results from the Phase 1b CHOPIN Trial, conducted at Leiden University Medical Center on the use of the Delcath CHEMOSAT Hepatic Delivery System with Melphalan (CHEMOSAT) in combination with the immune checkpoint inhibitors (ICI) ipilimumab and nivolumab to treat patients with metastatic uveal melanoma with liver metastases, were published in Cardiovascular and Interventional Radiology.
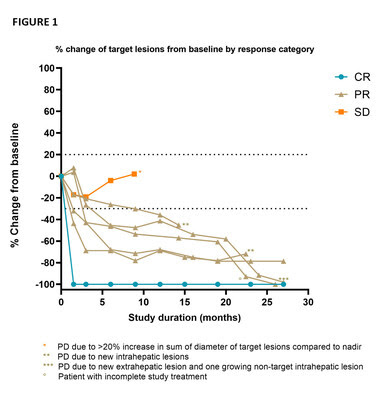
The goal of the CHOPIN trial is to study the safety and potential synergistic effects of systemic ICI therapy ipilimumab plus nivolumab (IPI+NIVO) when combined with Delcath's proprietary liver-targeted percutaneous hepatic perfusion (PHP) treatment in metastatic uveal melanoma patients. The just released publication presented updated safety and efficacy results from the Phase 1b portion of the trial which were previously presented in June 2022 at the American Society of Clinical Oncology Annual Meeting. The Phase 1b portion of the trial enrolled seven patients each of which were treated with two courses of PHP (melphalan 3mg/kg, max 220 mg per cycle) combined with four courses IPI+NIVO escalating the dosing from 1mg/kg each IPI+NIVO (cohort 1) to IPI 1mg/kg + NIVO 3mg/kg (cohort 2). As previously reported, the Best Overall Response included 1 complete response, 5 partial responses and 1 stable disease accounting for an Objective Response Rate of 85.7% and a Disease Control Rate of 100% (FIGURE 1). At the cut-off date of November 15, 2022, the median follow-up was 29.1 months (range 8.9 – 30.2), the median PFS was 29.1 months (95% CI 11.9 – 46.3) and the median duration of response was 27.1 months (range 7.4 –28.5). All patients are still alive and three of four patients who subsequently experienced PD continued with treatment in the form of repeated melphalan PHP (M-PHP) cycles.
The ongoing randomized phase 2 part of the CHOPIN trial comparing M-PHP alone with M-PHP plus IPI/NIVO, which will include another 76 patients (38 per arm), is approximately 50% enrolled and will provide more insight to the efficacy.
The determination of a safe and effective dose was a primary goal of the Phase 1b portion of the CHOPIN trial. Grade 1/2 adverse events were seen in all patients and 71.4% experienced grade 3/4 toxicities. In this phase 1b dose-escalation study combining M-PHP with IPI/NIVO the safe treatment dose was established at IPI 1mg/kg and NIVO 3mg/kg. The authors did observe low-grade immune-related toxicities and PHP-related hematological toxicities in the treated groups. Hematological toxicity is a common adverse event after M-PHP, affecting approximately three-quarters of patients. All 7 patients in the study experienced grade 1/2 anemia. To prevent severe leukopenia/neutropenia, G-CSF was administered within 48 hours after M-PHP in their treatment center. The phase 2 part of the CHOPIN study will provide more information on both hepatic and systemic toxicity associated with the combination therapy.
"If similar results are observed in the larger, randomized second phase of this trial, it would represent a meaningful improvement over current treatment options for this patient population," said Dr Johnny John, Senior Vice President, Medical and Clinical Affairs. "In addition, further investigation of this combination protocol may be warranted in patients with liver dominant disease in other tumor types currently treated with ICI agents."
The rationale for combining ICI therapy with M-PHP in metastatic uveal melanoma is based on both uveal melanoma's specific characteristics and the unique immunomodulatory role of the liver. Uveal melanoma is characterized by a different set of driver mutations and lower mutational load compared to cutaneous melanoma, leading to limited neoantigen presentation and lower efficacy of ICI. By combining M-PHP with
IPI/NIVO, the authors noted that they aim to increase the efficacy of ICI by turning a 'cold tumor' into a 'hot tumor'. In addition, while PHP can provide long-lasting disease control in the liver, it does not control extrahepatic disease. Conversely, IPI/NIVO treatment shows a trend towards control of extrahepatic lesions, but hepatic disease progression regularly occurs. With combined treatment, the authors aim to control hepatic disease, as well as prevent extrahepatic disease in follow-up. In addition, it is well documented that the liver has a unique immune modulating role and liver metastases diminish ICI efficacy systemically regardless of the type of primary tumor and in animal models it has been shown that this effect can be overcome by localized hepatic therapy. Current available evidence from studies on isolated limb perfusion (ILP) and isolated hepatic perfusion (IHP), which is the surgical counterpart of M-PHP, show that ILP and IHP can lead to T-cell activation following the procedures. The authors hypothesize that this is also the case for M-PHP leading to an improved activation of the immune system together with ICI.