Efficacy of Stereotactic Body Radiation Therapy in Recurrent Intrahepatic Cholangiocarcinoma
Images
Case Summary
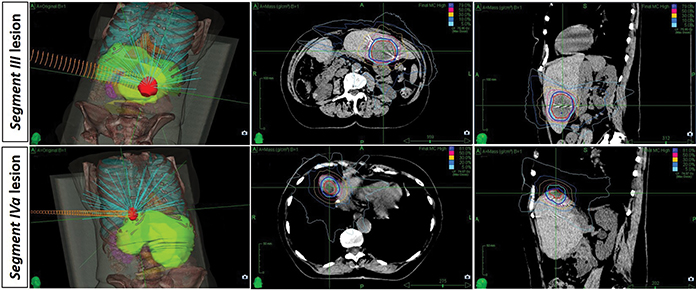
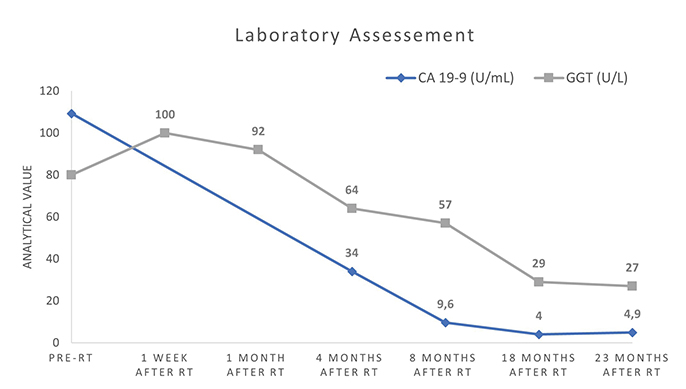
An 81-year-old woman was referred to the hospital in January 2017 with nausea, progressive worsening anorexia, and weight loss of about 6 kg in one year. A computed tomography (CT) scan showed a 9--6.5-cm mass in the right hepatic lobe involving the right branch of the portal vein, and a slight ectasia of the intrahepatic bile ducts. Hepatic biopsy revealed a histological diagnosis of cholangiocarcinoma. The patient underwent percutaneous portal embolization in February 2017 followed by right hepatectomy extended to segment I in April 2017. Histopathological examination revealed a diagnosis of intrahepatic cholangiocarcinoma (ICC), stage pT2G2N0R0 (AJCC 8th edition staging manual).1 She remained asymptomatic and with no signs of recurrence until January 2019 when she appeared with anorexia, nausea and weight loss of about 3 kg over a month. Laboratory study revealed an elevation of the tumor marker carbohydrate antigen (CA 19-9) (109.30 U/mL) and gamma-glutamyltransferase (GGT) (80 U/L). Carcinoembryonic antigen (CEA) was not altered. Abdominal MRI showed two focal hepatic lesions, one measuring 4.2 cm in segment III, and the other measuring 1.7 cm in segment IVa. At this point, the patient refused systemic treatment with palliative chemotherapy and, after a multidisciplinary board meeting, it was decided to locally treat both lesions with stereotactic body radiation therapy (SBRT). Two weeks after ultrasound-guided placement of fiducial markers, the patient underwent SBRT with a Cyberknife system (Accuray Incorporated) directed to the segment III liver lesion, at a dose of 60 Gy in 5 fractions of 12 Gy each, on alternate days. Five days after concluding treatment of segment III, she initiated SBRT of the segment IVa lesion, at a dose of 60 Gy, in 3 fractions of 20 Gy each, on alternate days (Figure 1). A 5-fractionation scheme was adopted for the segment III lesion to fulfill radiation dose constraints for adjacent normal tissues, according to UK Consensus on Normal Tissue Dose Constraints for Stereotactic Radiotherapy.2 Planning target volume (PTV) for the segment III lesion was 84.87 cm3 and for segment IVa was 17.09 cm3. Radiation plans were prescribed to the isodose line that provided greater than 95% coverage of the PTV. The prescription isodose line was 79% and 81%, for segment III and IV lesions, respectively. The Cyberknife Synchrony Respiratory Tracking System was used to continuously monitor respiratory movements and correlate the data with movements of a fiducial-marked target lesions.
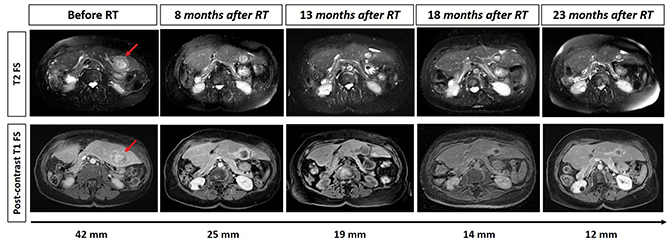
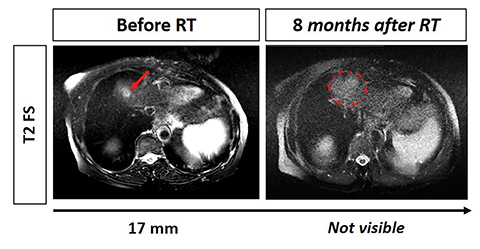
The patient concluded SBRT treatment in April 2019 uneventfully and with good tolerance. Subsequently, she maintains 6-monthly imaging and laboratory surveillance. Total follow-up time since SBRT was 23 months. At the last follow-up date (March 2021), the patient had no signs of gastrointestinal, skin or hematologic toxicity, with evidence of gradual remission of the hepatic segment III lesion and total disappearance of the hepatic segment IVa lesion. Furthermore, she has no signs of local recurrence or metastasis, even though she has not undergone any systemic treatment (Figures 2, 3).
Imaging Findings
Abdominal MRI revealed a 4.2 cm nodular formation at the lower/medial limit of hepatic segment III, with low uptake of contrast product and accentuated filling in the late phase, suggesting metastatic cholangiocarcinoma. It also revealed another 1.7-cm nodular lesion on the cranial margin of the segment VIa with the same characteristics. Macrovascular invasion has not been observed. Follow-up with abdominal MRI every 6 months after SBRT has revealed gradual volumetric reduction of the segment III lesion over time (Figure 2A) and disappearance of segment IVa lesion (Figure 2B).
CT scan during follow-up excluded other sites of secondary disease such as lungs, lymph nodes or bone metastasis.
Diagnosis
Intrahepatic cholangiocarcinoma previously resected — stage pT2G2N0R0, with subsequent appearance of two liver metastasis
Discussion
ICC is a rare entity with an incidence increasing worldwide, probably due to improved ability to establish the diagnosis.3 Surgical resection is considered the only treatment with curative intent for localized ICC. However, negative margin resection is challenging in patients with locally advanced tumors, and extended hepatic resection has been demonstrated as an independent risk factor for major postoperative complications.4 Approximately 70% of patients are unresectable at diagnosis due to multiple intrahepatic tumors, vascular invasion or distant metastases. For these patients, who are unable to achieve optimal resection and are mainly treated with palliative chemotherapy, the median survival ranges 2.3 to 9 months.3
The use of RT in intrahepatic malignancies was traditionally limited by concerns over hepatic tolerance and the resulting inability to deliver a sufficient treatment dose, particularly in patients who may have compromised hepatic function. The delivery of tumoricidal doses of RT has become feasible with the development of modern techniques, including charged particles and SBRT; however, evidence is scarce about its applicability in ICC. Studies regarding robotic radiosurgery of liver metastasis have shown that a total dose of 45 to 60 Gy administered in 3 to 5 fractions, with a fraction dose of 9 to 20 Gy was safe and effective as a local treatment option for secondary liver tumors.5 Moreover, a prospective dose-escalation study that used different doses (30 Gy in 3 fractions, 50 Gy in 5 fractions, and 60 Gy in 6 fractions) determined that the rate of complete and partial response (at 6 and 12 months), as well as the local control rate at 12 months, were significantly higher in the 60 Gy group.6
In this work, we present a case of a patient diagnosed with ICC 52 months ago, who underwent total resection of the tumor, with subsequent appearance of two liver metastases effectively and safely treated with SBRT, without any type of systemic treatment. After a follow-up period of 23 months, the patient remains with no signs of disease recurrence or progression. In fact, she has a survival superior to the overall survival (OS) reported in the vast majority of ICC metastatic cancer patients without extrahepatic disease.7 Some studies have been published in the last decade about the role of RT as locoregional therapy in patients with unresectable or metastatic ICC without extrahepatic disease.8-10 A single-institution retrospective study carried out by Tao and colleagues included 79 patients with localized inoperable ICC, treated with 3D-conformal intensity-modulated RT photon beam or passive scatter proton beam techniques, with doses ranging 35 to 100 Gy in 3 to 30 fractions, for a median biologic equivalent dose (EQD2) of 80.5 Gy.8 Median OS after the first diagnosis was 30 months. Authors concluded that higher EQD2 (> 80.5 Gy) correlated with an improved 3-year OS (73% vs 38%) and 3-year local control (78% vs 45%).8 However, an important factor could have highly contributed to the observed 3-year OS and local control, as the great majority of patients included in that study (89%) received systemic chemotherapy prior to RT, unlike the patient described in this case report.
A prospective phase II multi-institutional trial including 37 patients with unresectable ICC showed that hypofractionated proton therapy (median EQD2 of 58 Gy) resulted in a median progression-free survival (PFS) rate of 8.4 months, and 1-year and 2-year PFS rates of 41.4% and 25.7%, respectively.9 The median OS was 22.5 months, with 1-year and 2-year OS rates of 69.7% and 46.5%, respectively.9 Also, in this study 61.5% of patients underwent chemotherapy before RT. Based on these results, a recent NRG study (NRG-GI001) attempted to evaluate the use of hypofractionated external-beam RT (EBRT) in this patient population; however, this trial closed due to poor accrual. Last year, Smart and colleagues reported 2-year results of a cohort of 66 patients with unresectable or locally recurrent cholangiocarcinoma treated with hypofractionated proton or photon RT, with a median dose of 58.05 Gy (median of biologically effective dose of 80.52 Gy) in 15 fractions.10 Note that 42% of patients included in this study received prior chemotherapy, and 70% of patients presented with only one tumor lesion before RT. The median OS from the date of diagnosis was 25 months for the entire cohort, and 2-year OS and 2-year local control rates were 58% and 84%, respectively, with a relatively low severe toxicity rate (grade 3 < 11%).10 These studies suggest that ablative doses of radiation allow for a high local control rate and encouraging results in terms of survival outcomes.
SBRT remains an option for the treatment of all primary and metastatic liver cancers. In fact, local control rates reported in a recently published phase III randomized trial for recurrent hepatocellular carcinoma were 70% to 80% at 3 years, with proton beam therapy shown to be noninferior to radiofrequency.11 SBRT consists of a noninvasive and well-tolerated treatment that may allow patients sustained QOL benefits. Our patient did not require systemic treatment and tolerated SBRT excellently. Furthermore, SBRT in monotherapy was able to sustain good systemic disease control, prompting us to hypothesize about a potential abscopal response.
Conclusion
This case report is about a patient diagnosed with ICC 52 months ago. The patient was initially submitted for right hepatectomy; however, there was subsequent appearance of two liver metastases. She rejected systemic therapy and proceeded with SBRT of liver metastasis. After a total follow-up of 23 months since SBRT, the patient not only retains an excellent survival, but also has no signs of disease recurrence or progression.
Management of cholangiocarcinoma is often difficult, with limited options for salvage treatment. Although radiation has an established role in the treatment of cholangiocarcinoma, SBRT remains an option. This case report highlights the potential efficacy of SBRT in monotherapy for treating multiple intrahepatic metastases of ICC. Future randomized control trials attempting SBRT of the liver are needed for better management of advanced ICC.
References
- Lee AJ, Chun YS. Intrahepatic cholangiocarcinoma: the AJCC/UICC 8th edition updates. Chin Clin Oncol. 2018;7(5):9-11. doi:10.21037/cco.2018.07.03
- Hanna GG, Murray L, Patel R, et al. UK consensus on normal tissue dose constraints for stereotactic radiotherapy. Clin Oncol. 2018;30(1):5-14. doi:10.1016/j.clon.2017.09.007
- Endo I, Gonen M, Yopp AC, et al. Intrahepatic cholangiocarcinoma: rising frequency, improved survival, and determinants of outcome after resection. Ann Surg. 2008;248(1):84-96. doi:10.1097/SLA.0b013e318176c4d3
- De Jong MC, Nathan H, Sotiropoulos GC, et al. Intrahepatic cholangiocarcinoma: an international multi-institutional analysis of prognostic factors and lymph node assess
- ment. J Clin Oncol. 2011;29(23):3140-3145. doi:10.1200/JCO.2011.35.6519
- García R, Santa-Olalla I, Lopez Guerra JL, Sanchez S, Azinovic I. Robotic radiosurgery for the treatment of liver metastases. Rep Pract Oncol Radiother. 2017;22(2):111-117. doi:10.1016/j.rpor.2016.10.002
- Rule W, Timmerman R, Tong L, et al. Phase i dose-escalation study of stereotactic body radiotherapy in patients with hepatic metastases. Ann Surg Oncol. 2011;18(4):1081-1087. doi:10.1245/s10434-010-1405-5
- Kang SH, Hwang S, Lee YJ, et al. Prognostic comparison of the 7th and 8th editions of the American Joint Committee on Cancer staging system for intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2018;25(4):240-248. doi:10.1002/jhbp.543
- Tao R, Krishnan S, Bhosale PR, et al. Ablative radiotherapy doses lead to a substantial prolongation of survival in patients with inoperable intrahepatic cholangiocarcinoma: a retrospective dose response analysis. J Clin Oncol. 2016;34(3):219-226. doi:10.1200/JCO.2015.61.3778
- Hong TS, Wo JY, Yeap BY, et al. Multi-institutional phase II study of high-dose hypofractionated proton beam therapy in patients with localized, unresectable hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J Clin Oncol. 2016;34(5):460-468. doi:10.1200/JCO.2015.64.2710
- Smart AC, Goyal L, Horick N, et al. Hypofractionated radiation therapy for unresectable/locally recurrent intrahepatic cholangiocarcinoma. Ann Surg Oncol. 2020;27(4):1122-1129. doi:10.1245/s10434-019-08142-9
- Kim TH, Koh YH, Kim BH, et al. Proton beam radiotherapy vs. radiofrequency ablation for recurrent hepatocellular carcinoma: a randomized phase III trial. J Hepatol. 2021;74(3):603-612. doi:10.1016/j.jhep.2020.09.026
Citation
M A, C S, C F, C C, S V, G C, C C, P C. Efficacy of Stereotactic Body Radiation Therapy in Recurrent Intrahepatic Cholangiocarcinoma. Appl Radiat Oncol. 2021;(3):68-71.
October 5, 2021