Antibody Targets a Secretory Protein for Anti-cancer Therapy
Scientists have created antibodies to neutralize it and stop cancer growth by identifying how the cancer-promoting protein chitinase-3-like-1 is released by cells. The discovery made by a team from Taiwan’s National Cheng Kung University unearthed how the secretory glycoprotein, chitinase-3-like-1 (CHI3L1, is secreted by cells, identified a new protein biomarker for cancer progression, and then developed CHI3L1-neutralizing antibodies that can arrest the growth of cancers and supplement existing standard chemotherapies.
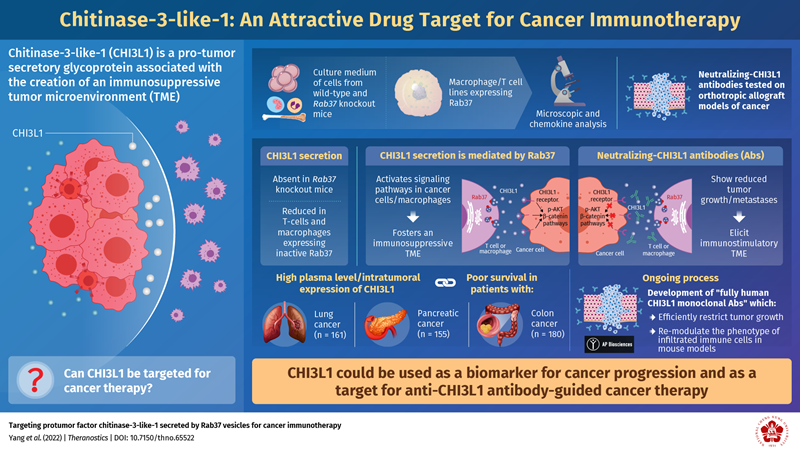
Researchers from National Cheng Kung University, Taiwan, have recently been successful in identifying more details on how CHI3L1 promotes tumorigenesis (the development of cancer). The group’s findings, published in Theranostics, successfully described CHI3L1’s possible secretory and immunosuppressive mechanisms and developed and validated the efficacy of anti-CHI3L1 antibodies in solid tumors.
The team used a variety of experimental approaches and cutting-edge techniques, such as vesicle isolation, fluorescence microscopy, and immunohistochemistry to come to their conclusions. For instance, the team monitored the real-time movement of the CHI3L1 protein inside cells using light-emitting fluorescent “trackers.” To further validate their findings, the researchers also conducted experiments using laboratory mice.
Dr. Yi-Ching Wang, Chair Professor at the National Cheng Kung University, Taiwan, explained “CHI3L1 is typically secreted by a variety of cells, including macrophages, T cells, neutrophils, epithelial cells, smooth muscle cells, fibroblasts, and cancer cells. We found that a protein called Rab37 mediated the transport of CHI3L1 out of these cells via a pathway that was dependent on guanosine triphosphate (GTP) for energy. This was evidenced by the fact that CHI3L1 secretion was attenuated in mice, which were genetically modified to not produce Rab37.”
Once outside the cell, CHI3L1 activated a series of sub-cellular signaling pathways, resulting in the rapid development of pro-tumor microenvironments. This finding led the team to focus its attention on the development of novel antibodies to counter subsequent cancer progression.
“CHI3L1 blockage by neutralizing antibodies may shift tumor microenvironments towards an immunostimulatory phenotype, therefore reducing cancer progression,” said Dr. Wang. “Hence, we developed CHI3L1-neutralizing antibodies, which we tested on cancer models in mice and on cancer cell lines.”
As expected, the neutralizing antibodies reduced tumor growth. They also reduced metastases—the spread of cancer from its original location to one or more nearby/distant body organs. Moreover, they triggered a significant “biological reversal” by creating physiological conditions that facilitate the identification and elimination of cancer cells by the immune system. The researchers observed this reversal in lung, pancreatic, and colon tumor models.
Dr. Yan-Shen Shan, senior author and Distinguished Professor, and Dr. Wu-Chou Su, Professor and Director at the Department of Internal Medicine (both from the National Cheng Kung University), further explained that the CHI3L1-neutralizing antibodies appear to be non-toxic and safe in the short-term. Combining them with current chemotherapies used in cancer treatment could lessen adverse effects and shorten patients’ hospital stay, they added.
After observing the great efficacy of these antibodies, for further clinical application, the team recently developed fully human monoclonal antibodies against CHI3L1 with AP Biosciences, Inc. Two out of eleven candidate clones will be validated with more in vivo examinations to determine which has the best antitumor efficacy and lowest toxicity.