Using Natural Killer Cells Could Bolster Immune-Checkpoint Therapy
 Scientists at Albert Einstein College of Medicine describe findings in The Journal of Clinical Investigation (JCI) that could bolster the effectiveness of immune-checkpoint therapy.
Scientists at Albert Einstein College of Medicine describe findings in The Journal of Clinical Investigation (JCI) that could bolster the effectiveness of immune-checkpoint therapy.
The introduction a decade ago of immune checkpoint inhibitors that work by unleashing the immune system’s T cells to attack tumor cells marked a major advance in cancer therapy. However, only 10% to 30% of treated patients experience long-term improvement.
Rather than rally T cells against cancer, the Einstein research team used different human immune cells known as natural killer (NK) cells—with dramatic results. “We believe the novel immunotherapy we’ve developed has great potential to move into clinical trials involving various types of cancer,” said study leader Xingxing Zang, MMed, PhD, the Louis Goldstein Swan Chair in Cancer Research and professor of microbiology & immunology, of oncology, of urology, and of medicine at Einstein and a member of the Cancer Therapeutics Program of the Montefiore Einstein Cancer Center.
The surfaces of immune cells are studded with receptors known as “checkpoint” proteins, which prevent immune cells from straying beyond their usual targets (pathogen-infected cells and cancer cells). When checkpoint receptors on immune cells bind with proteins expressed by the body’s own normal cells, the interaction puts the brakes on a possible immune-cell attack. Diabolically, most types of cancer cells express proteins that bind with checkpoint proteins, tricking immune cells into standing down and not attacking the tumor.
Immune checkpoint inhibitors are monoclonal antibodies designed to short-circuit immune-cell/cancer-cell interactions by blocking either the tumor proteins or the immune-cell receptors that bind with tumor proteins. With no brakes to impede them, immune cells can attack and destroy cancer cells.
The limited effectiveness of checkpoint inhibitors prompted Dr Zang and other scientists to look at checkpoint pathways involving NK cells, which—like T cells—play major roles in eliminating unwanted cells. A cancer-cell protein called PVR soon captured their attention. “We realized that PVR may be a very important protein that human cancers use to hobble the immune system’s attack,” said Dr Zang.
PVR protein is usually absent or very scarce in normal tissues but is found in abundance in many types of tumors including colorectal, ovarian, lung, esophageal, head and neck, stomach, and pancreatic cancer as well as myeloid leukemia and melanoma. Moreover, PVRs appeared to inhibit T cell and NK cell activity by binding to a checkpoint protein called TIGIT—prompting efforts to interrupt the TIGIT/PVR pathway by using monoclonal antibodies made against TIGIT. More than 100 clinical trials targeting TIGIT are now in progress worldwide. However, several clinical studies including two large phase 3 clinical trials have recently failed to improve cancer outcomes.
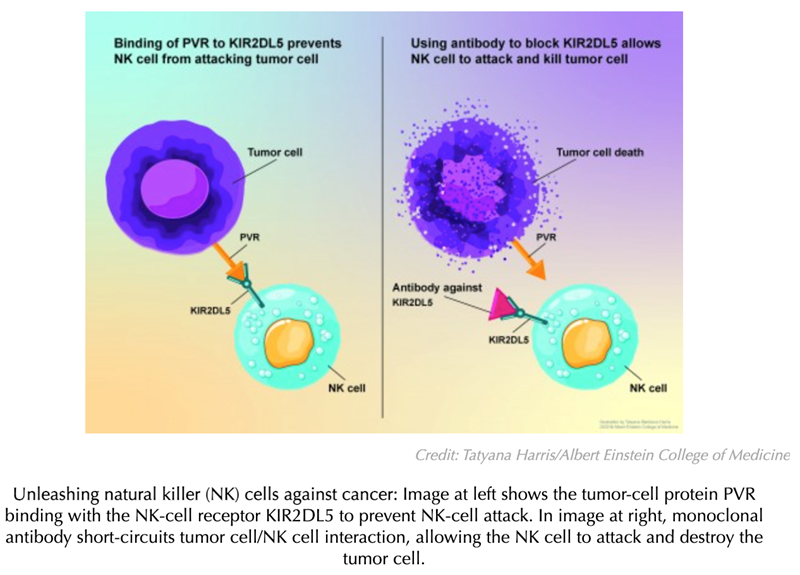
Meanwhile, the cancer-cell protein PVR was found to have another “binding partner” on NK cells: KIR2DL5. “We hypothesized that PVR suppresses NK cell activity not by binding with TIGIT but by binding with the recently recognized KIR2DL5,” said Dr Zang. To find out, he and his colleagues synthesized a monoclonal antibody targeting KIR2DL5 and carried out in vitro and in vivo experiments using the antibody.
In their JCI paper, Dr Zang and colleagues demonstrated that KIR2DL5 is a commonly occurring checkpoint receptor on the surface of human NK cells, which PVR cancer proteins use to suppress immune attack. In studies involving humanized animal models of several types of human cancers, the researchers showed that their monoclonal antibody against KIR2DL5—by blocking the KIR2DL5/PVR pathway—allowed NK cells to vigorously attack and shrink human tumors and prolong animal survival (see accompanying illustration). “These preclinical findings raise our hopes that targeting the KIR2DL5/PVR pathway was a good idea and that the monoclonal antibody we’ve developed may be an effective immunotherapy,” said Dr Zang.
Einstein has filed a patent application for KIR2DL5/PVR immune checkpoint including antibody drugs and is interested in a partnership to further develop and commercialize the technology.
Dr Zang has previously developed and patented more than 10 immune checkpoint inhibitors. One of those inhibitors is now being tested in China in phase 2 clinical trials involving several hundred patients with advanced solid cancers (non-small cell lung cancer, small cell lung cancer, nasopharyngeal cancer, head and neck cancer, melanoma, lymphoma) or recurrent/refractory blood cancers (acute myeloid leukemia, myelodysplastic syndromes). Another of Dr Zang’s immune checkpoint inhibitors will be evaluated starting next year in cancer clinical trials in the United States.