Unfavorable Intermediate- and High-Risk Prostate Cancer Treated With Predominantly Brachytherapy Alone With Long-Term Follow-Up
Applied Radiation Oncology — Vol. 12, Issue 4, pp. 20-28
Published: December 1, 2023
1 Department of Radiation Oncology, Kaiser Permanente Southern California, Los Angeles, CA
2 Department of Research and Evaluation, Kaiser Permanente, Pasadena, CA
* Corresponding author: Barry Goy (barry.w.goy@kp.org)
Abstract
Keywords
prostate cancer, brachytherapy, high risk, radiation therapy
Introduction
Low-dose-rate (LDR) brachytherapy (BT) using radioactive seeds has shown excellent 10-year results in multiple studies.1-3 However, most patients in these trials were low risk. The National Comprehensive Cancer Network (NCCN) guidelines recommend local therapy for treating patients with unfavorable intermediate-risk prostate cancer (U-IRPC) and high-risk prostate cancer (HRPC), but do not recommend BT as monotherapy without the use of supplemental external-beam radiation therapy (S-EBRT) or neoadjuvant androgen deprivation therapy (NADT).4 These guidelines tend to emphasize the use of pituitary ablation as opposed to prostate ablation. Our prior experience of patients with favorable IRPC and U-IRPC showed impressive results using predominantly BT alone compared with radical prostatectomy (RP) and external-beam radiation therapy (EBRT) with or without NADT.5 Our aim was to perform a retrospective analysis of U-IRPC and HRPC, comparing BT with or without S-EBRT, with long-term follow-up.
Materials and Methods
Patient Characteristics
There were 156 patients with U-IRPC and HRPC who underwent BT at our integrated, multifacility health care system between January 2004 and December 2016. Patients were clinically staged, with a digital rectal examination (DRE) for T-stage from the 2002 American Joint Committee Cancer staging.6 Other tests included initial prostate-specific antigen (PSA) scoring prior to treatment and biopsies of the prostate with a Gleason score (GS) assessment. IRPC was classified as clinical stage T2b-c, GS 3 + 4 or 4 + 3 (group 2 or 3), and/or initial PSA of 10.1-20.0. Percentage positive biopsy core (PPBC < 50%) calculated from the pathology report was also considered an intermediate risk factor. U-IRPC was defined as GS 4 + 3 (group 3) or those with ≥ 2 intermediate-risk factors.7 HRPC was defined as any patient with GS 8-10 (group 4 or 5), initial PSA >20.0, and/or clinical T3a disease by DRE, but not by MRI.8 We defined favorable HRPC as ≤ T2c, Gleason group 4, and PSA ≤ 10.0. U-HRPC patients were those with clinical T3a, Gleason group 5, Gleason group 4 with a PSA > 10.0, or Gleason groups 2-5 with a PSA > 20.0. Charlson comorbidity index was assigned to each patient to assess overall health status.9
Therapy
Patients underwent S-EBRT using either 3-dimensional conformal therapy with a 6-field approach or intensity-modulated radiation therapy, with 0.8-cm planning target volume around the prostate and seminal vesicles, but a 0.6-cm posterior. The S-EBRT dose prescribed was 45-46 Gy in 1.8-2.0 Gy fractions over 5 weeks covering the prostate and seminal vesicles. The pelvic lymph nodes were not treated. NADT was given using leuprolide, typically for 3-6 months, starting 2-3 months prior to S-EBRT or BT, and concurrently with S-EBRT or BT. For BT, stranded iodine-125 radioactive seeds were inserted transperineally using ultrasound guidance, a stepper-stabilizer unit, and fluoroscopy. Planning ultrasound was done with the placement of a urethral catheter to define the urethra and prostate base. A minimum peripheral dose of 145 Gy to the prostate and proximal seminal vesicles was prescribed using 0.4 mCi per seed with a modified peripheral loading technique, whereas 110 Gy was prescribed for the BT boost patients about 4 weeks after completion of S-EBRT.10,11 Postimplant dosimetry was performed using CT 1-2 weeks after BT, utilizing the VariSeed 8.0.1 (Varian) fusion program. V100 (percent volume that received ≥ 100% of the prescribed dose) and D90 (percentage of the prescribed dose delivered to 90% of the prostate) were calculated for the prostate.
Follow-Up
Time zero was the date of BT. Freedom from biochemical failure (FFBF) was defined based on the American Society of Therapeutic Radiology and Oncology-Phoenix definition of biochemical failure of PSA nadir + 2 ng/mL threshold.12 Patients experiencing biochemical failure typically underwent androgen deprivation therapy (ADT). Complications were graded according to the Radiation Therapy Oncology Group grading system for late effects.13 A minimum of 12 months of follow-up was required for this study.
Statistics
Patient characteristics were delineated with percentages for categorical factors, and median and range were utilized for continuous factors. The Pearson chi-square test was utilized to assess differences in categorical characteristics between larger groups, whereas the Fisher exact test was used for smaller sample size comparisons. The Kruskal-Wallis test was performed to calculate the differences in continuous factors between groups. Kaplan-Meier estimates were performed at 10 years, and the log-rank statistic was performed to estimate the differences between local therapies, using two-sided P < 0.05.14 Multivariable analysis (MVA) using the Cox proportional hazards regression model was utilized to find independent prognostic factors.15 Outcomes were then adjusted by weighting the observations according to the inverse probability of treatment, based on the propensity scores, to account for selection bias between treatment groups, based on factors that were independently prognostic on MVA.16
Results
Patient Cohort and Prognostic Factors
Median follow-up of BT vs S-EBRT was 8.2 vs 8.3 years, with a range for all patients of 1.4-18.2 years. In total, 124 (79.5%) had U-IRPC, while 32 (20.5%) had HRPC. Also, 129 (82.7%) underwent BT alone, while 27 (17.3%) underwent S-EBRT using BT as a boost (Tables 1 and 2).
| BT ALONE(n = 129) | S-EBRT + BT(n = 27) | TOTAL(n = 156) | P VALUE | |
|---|---|---|---|---|
| Age, median (IQR) | 67.7 (62.3, 73.1) | 67.7 (63.6, 70.9) | 67.7 (62.4, 73.0) | .90* |
| Race/ethnicity, n (%) | ||||
| Asian | 12 (9.3%) | 4 (14.8%) | 16 (10.3%) | 0.56@ |
| Black | 29 (22.5%) | 7 (25.9%) | 36 (23.1%) | |
| Hispanic | 21 (16.3%) | 2 (7.4%) | 23 (14.7%) | |
| White | 67 (51.9%) | 14 (51.9%) | 81 (51.9%) | |
| Charlson score, median (IQR) | 2.0 (1.0, 3.0) | 2.0 (1.0, 2.0) | 2.0 (1.0, 3.0) | .55* |
| Clinical T-stage, n (%) | ||||
| T1c | 79 (61.2%) | 12 (44.4%) | 91 (58.3%) | 0.22@ |
| T2a | 26 (20.2%) | 11 (40.7%) | 37 (23.7%) | |
| T2b | 20 (15.5%) | 4 (14.8%) | 24 (15.4%) | |
| T2c | 3 (2.3%) | 0 (.0%) | 3 (1.9%) | |
| T3a | 1 (.8%) | 0 (.0%) | 1 (.6%) | |
| Initial PSA, median (IQR) | 9.2 (6.1, 11.7) | 9.4 (6.5, 12.7) | 9.2 (6.2, 11.8) | .87* |
| Initial PSA | ||||
| ≤ 10.0 | 69 (53.5%) | 15 (55.6%) | 84 (53.8%) | 0.54@ |
| 10.1-20.0 | 51 (39.5%) | 12 (44.4%) | 63 (40.4%) | |
| > 20.0 | 9 (7.0%) | 0 (.0%) | 9 (5.8%) | |
| Gleason grade group, n (%) | ||||
| Group 1 (GS 6) | 17 (13.2%) | 0 (.0%) | 17 (10.9%) | 0.01@ |
| Group 2 (3 + 4) | 48 (37.2%) | 12 (44.4%) | 60 (38.5%) | |
| Group 3 (4 + 3) | 49 (38.0%) | 7 (25.9%) | 56 (35.9%) | |
| Group 4 (GS 8) | 12 (9.3%) | 4 (14.8%) | 16 (10.3%) | |
| Group 5 (GS 9-10) | 3 (2.3%) | 4 (14.8%) | 7 (4.5%) | |
| PPBC > 50%, n (%) | 35 (27.1%) | 15 (55.6%) | 50 (32.1%) | <.01# |
| Use of NADT, n (%) | 9 (7.0%) | 16 (59.3%) | 25 (16.0%) | <.01# |
| FFBF | FFST | MFS | PCSS | OS | |
|---|---|---|---|---|---|
| Age (older vs younger) | HR = .56P = .13 | HR = .46P = .08 | HR = .62P = .44 | HR = .96P = .97 | HR = 1.78P = .14 |
| Race (Black vs non-Black) | HR = 1.52P = .31 | HR = 1.01P = .98 | HR = 1.13P = .87 | HR = .31P = .36 | HR = .58P = .19 |
| Charlson score (3+ vs 1 and 2) | HR = .97P = .95 | HR = 1.65P = .29 | HR = 1.51P = .54 | HR = .94P = .96 | HR = 2.58P = .01 |
| Clinical stage (T2b-3a vs T1c-T2a) | HR = 2.15P = .07 | HR = 2.30P = .07 | HR = 6.00P < .01 | HR = 3.44P = .22 | HR = .86P = .72 |
| Initial PSA (>10.0 vs ≤10.0) | HR = 1.23P = .57 | HR = 1.38P = .45 | HR = 2.36P = .20 | HR = .83P = .84 | HR = .85P = .65 |
| Gleason group (5,4 vs 3,2,1) | HR = 2.41P = .06 | HR = 2.57P = .08 | HR = 4.66P = .04 | HR = 1.72P = .71 | HR = 1.31P = .60 |
| % + cores > 50% | HR = .91P = .82 | HR = .60P = .30 | HR = .40P = .20 | HR = 1.55P = .68 | HR = .90P = .79 |
| Use of NADT (N vs Y) | HR = .39P = .07 | HR = .84P = .80 | HR = .67P = .68 | HR = .15P = .14 | HR = .56P = .24 |
| Use of S-EBRT (N vs Y) | HR = .72P = .56 | HR = 1.12P = .87 | HR = 1.01P = .99 | HR = .74P = .78 | HR = .36P = .07 |
Median initial PSA for BT alone vs S-EBRT + BT was 9.2 (range, .9-50.0) vs 9.4 (range, 4.2-18.0), P = 0.87. The clinical stage was not significantly different between BT alone vs S-EBRT, P = 0.22. The S-EBRT group had a higher proportion with higher a Gleason group (P = .01) and PPBC > 50% (P < .01), but also had a significantly higher percentage of patients undergoing NADT (P < .01). Median duration of NADT for BT vs S-EBRT + BT was 3 months (interquartile range [IQR], 3, 6) vs 6 months (IQR, 6, 6), P = 0.01, ranging for all patients from 3 to 18 months. NADT using leuprolide was given to 9 (7.0%) vs 16 (59.3%) of BT alone vs S-EBRT, P < 0.01. Only 25 patients (16%) underwent NADT, and most received 3-6 months, while only 2 patients received long-term NADT of 12 and 18 months, which were in the S-EBRT group.
On MVA, higher clinical stage (P < .01) and higher Gleason group (P = .04) independently predicted a lower 10-year MFS, while a higher Charlson score predicted a lower overall survival (OS) (P = .01) (Table 2).
Dosimetry and Use of MRI
Postimplant dosimetry of BT alone vs S-EBRT + BT revealed a median V100 of 96.8% vs 96.6% (P = .99), and a D90 of 115.7% vs 115.9% (P = .51). Median prostate size for BT alone vs S-EBRT + BT was 36.9 cm3 (range, 12.0-72.9 cm3) vs 29.6 cm3 (range, 20.4-48.6 cm3), P = 0.03.
Fourteen (8.9%) underwent MRI prior to treatment as part of risk assessment, and 2 of these underwent S-EBRT and had T2 disease on MRI. Twelve underwent BT alone, and 2 of these were upgraded. The first was T2a on DRE and upgraded to T3a on MRI, and their PSA was < 0.1 at 6.7 years. The second patient was upgraded from T2b on DRE to T3a/b on MRI with extracapsular extension and proximal seminal vesicle invasion, and was also biochemically free of disease at 8.7 years.
Main Outcomes
None of the survival outcomes were significantly different between BT alone vs S-EBRT + BT boost. The 10-year FFBF for BT vs S-EBRT was not significant in both unadjusted (77.0% vs 71.6%, P = .53) and adjusted models (76.8% vs 72.9%, P = .70). The propensity-adjusted 10-year MFS, prostate cancer-specific survival (PCSS), and overall survival (OS) for BT vs S-EBRT were 90.8% vs 87.3% (P = .81), 98.4% vs 87.3% (P = .36), and 73.4% vs 78.0% (P = .18), respectively (Figure 1, Table 3).
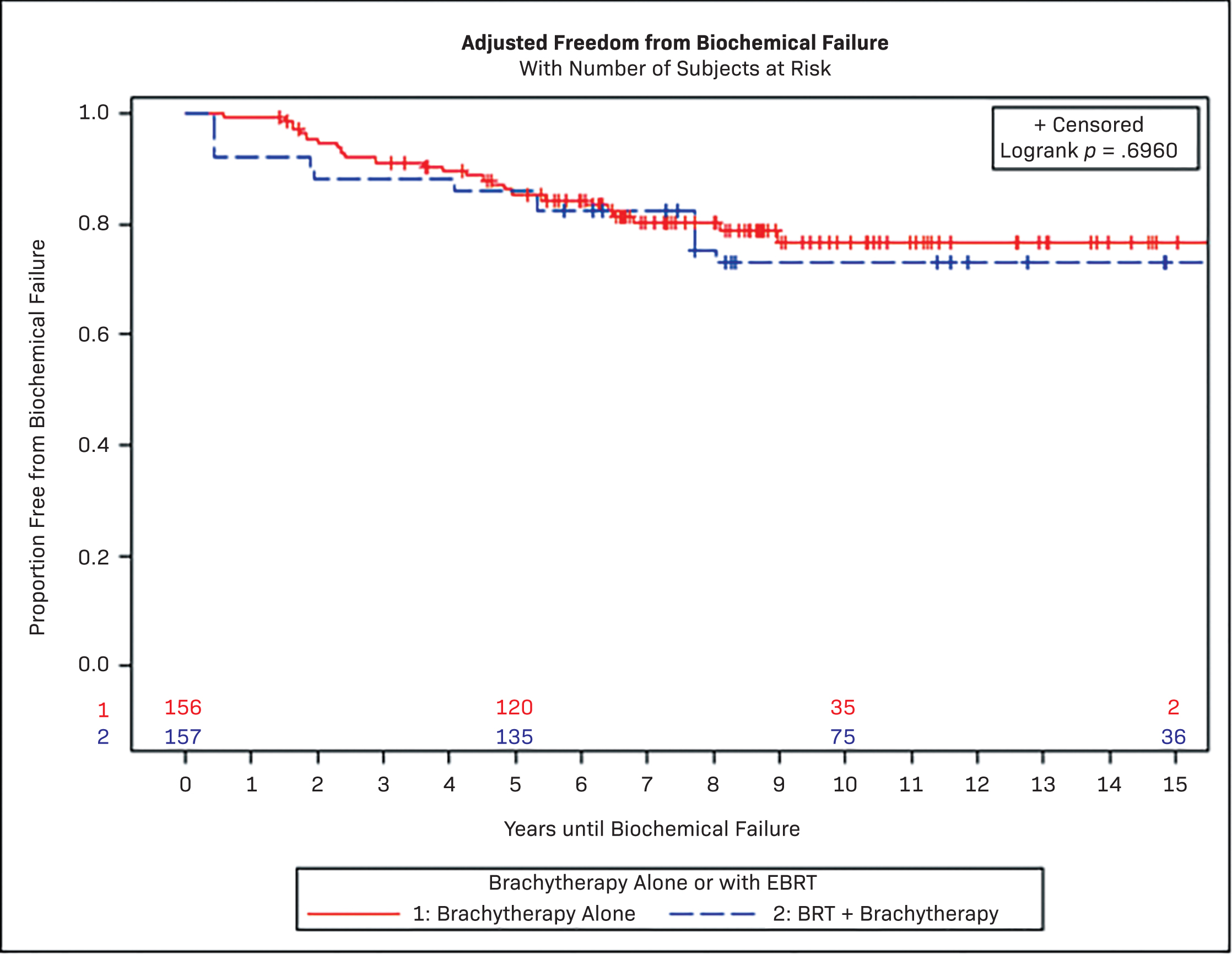
| # SUBJECTS | OBSERVED EVENTS | 10 y UNADJUSTED PROBABILITY | P LOG RANK | 10 y ADJUSTED PROBABILITY | P LOG RANK | |
|---|---|---|---|---|---|---|
| FFBF | ||||||
| BT | 129 | 25 | 77.0% (67.2%, 84.2%) | 0.53 | 76.8% (66.9%, 84.0%) | 0.7 |
| S-EBRT + BT | 27 | 7 | 71.6% (48.9%, 85.5%) | 72.9% (48.7%, 87.1%) | ||
| MFS | ||||||
| BT | 129 | 11 | 91.1% (84.1%, 95.1%) | 0.86 | 90.8% (83.7%, 94.9%) | 0.81 |
| S-EBRT + BT | 27 | 3 | 87.1% (64%, 96%) | 87.3% (63%, 96%) | ||
| PCSS | ||||||
| BT | 129 | 4 | 98.4% (89.4%, 99.8%) | 0.94 | 98.4% (89.3%, 99.8%) | 0.36 |
| S-EBRT + BT | 27 | 2 | 93.8% (63.2%, 99.1%) | 87.3% (55.0%, 97.0%) | ||
| OS | ||||||
| BT | 129 | 34 | 74.1% (63.5%, 82.0%) | 0.07 | 73.4% (62.6%, 81.5%) | 0.18 |
| S-EBRT + BT | 27 | 5 | 83.7% (56.5%, 94.6%) | 78.0% (48.6%, 91.8%) |
Median follow-up of U-IRPC vs HRPC was 8.5 (1.6-18.2) vs 7.8 years (6.9-8.5), P = 0.06. Ten-year FFBF, freedom from salvage therapy (FFST), MFS, PCSS, and OS for U-IRPC vs HRPC were 80.7% vs 55.6% (P < .01), 86.0% vs 66.3% (P = .01), 94.5% vs 72.6% (P < .01), 97.1% vs 100.0% (P = .55), and 77.5% vs 60.4% (P = .60), respectively.
Subset analysis of 32 patients with HRPC revealed 13 with favorable HRPC and 19 with U-HRPC. The 10-year FFBF for favorable HRPC vs U-HRPC was 91.7% vs 31.7% (P < .01), and the 10-year MFS was 100.0% vs 53.7% (P = .01). Of those with favorable HRPC, 11 of 13 underwent BT alone.
Salvage Therapy
The 10-year FFST was 83% vs 78% for BT vs S-EBRT + BT, P = 0.89. One patient underwent salvage cryotherapy, which failed, and went on to systemic therapy. All other patients underwent ADT as first-line salvage therapy. Three patients who had BT alone received abiraterone and enzalutamide, 1 of whom also received docetaxel. Two patients treated with S-EBRT received docetaxel, 1 of whom also underwent abiraterone and enzalutamide.
Patterns of Failure
FFBF and MFS for U-IRPC were significantly higher than HRPC, with 10-year FFBF of U-IRPC and HRPC being 80.7% vs 55.6%, P < 0.01, and MFS of 94.5% vs 72.6%, P < 0.01, respectively. Analyzing the patterns of failure, there were few patients with local recurrences: 2 experiencing isolated seminal vesicle recurrence, 1 with seminal vesicle plus prostate recurrence, and 1 with prostate recurrence alone, who later underwent cryotherapy. The patients in this study were in the era prior to prostate-specific membrane antigen PET (PSMA-PET) imaging, while MRI and biopsies were utilized to evaluate patients at the time of biochemical failure (Table 4).
| U-IRPC (n = 20) | HRPC (n = 12) | |
|---|---|---|
| Biochemical failure only | 14 | 3 |
| Isolated prostate failure | 1 | 0 |
| Isolated seminal vesicle failure | 1 | 1 |
| Prostate and seminal vesicle failure | 0 | 1 |
| Prostate, seminal vesicle, and pelvic nodal failure | 0 | 1 |
| Positive prostate biopsy | 2/5 | 0/3 |
| Pelvic nodal metastases only | 0 | 2 |
| Peri-rectal nodal metastases only | 1 | 0 |
| Para-aortic and pelvic nodal metastases | 2 | 1 |
| Bone metastases | 5 | 2 |
| Lung metastases | 0 | 1 |
Prevalence of Complications
There was a significantly higher prevalence of urinary complications using S-EBRT of 33.3% vs 16.3% for BT alone, P = 0.04; however, severe grade 3 and 4 complications for BT vs S-EBRT were not significantly different, 5.4% vs 3.7%, P = 0.99. There was also a higher prevalence of rectal complications using S-EBRT vs BT of 22.2% vs 6.2%, P < 0.01; most of which were grades 1 and 2, and only 1 severe grade 4 fistula. Most of the severe grade 3 and 4 complications were obstructive urinary symptoms, with 2 transurethral resections of prostate, 2 needing daily clean intermittent catheterization, 1 requiring a daily indwelling urethral catheter, 1 requiring percutaneous nephrostomy tubes, and 1 experiencing grade 3 urinary incontinence (Table 5).
| BT (n = 129) | S-EBRT + BT (n = 27) | P VALUE | |
|---|---|---|---|
| Urinary complications | 21 (16.3%) | 9 (33.3%) | .04* |
| Grade 1 and 2 urinary | 14 (10.9%) | 8 (29.6%) | .01* |
| Grade 3 and 4 urinary | 7 (5.4%) | 1 (3.7%) | 0.99@ |
| Rectal complications | 8 (6.2%) | 6 (22.2%) | <.01* |
| Grade 1 and 2 rectal | 7 (5.4%) | 6 (22.2%) | <.01* |
| Grade 3 and 4 rectal | 1 (.8%) | 0 (.0%) | 0.99@ |
Discussion
The NCCN guidelines historically only recommended monotherapy BT for the treatment of low-risk prostate cancer, and only since 2015 recommended its use for favorable IRPC.4 The NCCN currently only recommends BT as a boost for U-IRPC and HRPC, along with S-EBRT or whole pelvic radiation, and NADT. Whole pelvic radiation is endorsed by the NCCN, along with 6 months of NADT for U-IRPC and a minimum of 18 months of NADT for HRPC, although conflicting data exist regarding whole pelvic radiation, and none of these trials used BT.4,17-19 The most recent trial that was positive for pelvic radiation, but did not use BT, showed an improvement in biochemical failure, disease-free survival, and distant metastases-free survival, and also incorporated the use of PSMA-PET to assess distant disease, which is much more sensitive than prior imaging.19 Increasing the sensitivity of detecting distant disease will influence oncological outcomes, making MFS closer to a surrogate of FFBF or progression-free survival, and less of a predictor of PCSS. For patients not undergoing RP, the standard recommendation has been long-term NADT with EBRT, considered category 1 by the NCCN. One problem with this recommendation is that patients with HRPC are a heterogeneous mix of patients.20 One approach could be to segregate HRPC into a favorable vs unfavorable category, in which favorable could represent those with a reasonable probability of having organ-confined disease or disease into the capsule. With the increasing use of MRI and PSMA-PET, selecting U-IRPC and HRPC that may have localized disease should become more feasible, making these patients amenable to BT alone, which provides more ablative doses than EBRT.5 Our preference would be to offer BT alone to those favorable HRPC with Gleason group 4, PSA ≤ 10.0, T1c-T2c disease, including T3a seen on MRI (not T3a on DRE). Also, it seems that our favorable HRPC patients did just as well as those with U-IRPC, suggesting that these could be merged together as a risk group so that favorable HRPC could be categorized as U-IRPC. However, we recommend excluding those with seminal vesicle invasion and patients with a high risk of systemic disease. The majority of our patients received neither S-EBRT nor NADT, and the patients who did undergo NADT were short-term of 3-6 months. Also, none of our patients underwent whole pelvic radiation. A recent randomized study showed no benefit with the addition of S-EBRT to BT for favorable IRPC, but this study did not include U-IRPC or HRPC.21 Despite the lack of additional therapies, the BT alone cohort showed excellent FFBF, MFS, and PCSS at 10 years, although for U-HRPC, FFBF and MFS were much lower at 10 years at 31.7% and 43.3%, and no significant difference was found with the addition of S-EBRT, despite 59.3% of S-EBRT + BT boost undergoing NADT vs only 7.0% for the BT-alone group. PCSS remained high for both U-IRPC and HRPC, likely due to the availability of effective salvage therapies that can prolong survival, but are not curative. Although the S-EBRT group did have higher Gleason grouping and higher PPBC > 50%, MVA and propensity score matching did not show any benefit in regards to oncological outcomes with S-EBRT, despite a majority of S-EBRT undergoing NADT. Is it possible that the ablative doses given to the prostate and surrounding capsule are the most important factor in treating U-IRPC and favorable HRPC, while having very high rates of PCSS? The ASCENDE-RT trial treated all patients with NADT of 1 year along with pelvic radiation, comparing high-dose EBRT to the prostate vs BT boost, and this showed improvement of progression-free survival in those who underwent BT, although the BT boost arm did experience more urinary and rectal toxicities.22,23 This trial indicates that the ablative doses provided by BT still have an important role in the treatment of U-IRPC and favorable HRPC. A publication of 2 randomized studies by Merrick et al was done on patients with intermediate- and high-risk prostate cancer, which did not show any improvement with the addition of S-EBRT, compared with BT alone.24 It seems that one difference between the ASCENDE-RT trial vs the Merrick study was that the ASCENDE-RT used pelvic radiation, while the Merrick study may have included only the prostate and seminal vesicles when using S-EBRT. Additionally, a Surveillance, Epidemiology, and End Results Medicare analysis of 5835 patients also confirmed the lack of benefit of S-EBRT in addition to BT in regards to PCSS.25
One retrospective study also showed that BT was reasonable for HRPC, which compared 2557 HRPC with a median follow-up of 63.5 months, comparing RP vs EBRT vs BT, with NADT given in 19% vs 93% vs 53%, respectively, P < 0.0001.26 Biochemical relapse-free survival and clinical relapse-free survival (cRFS) were equivalent between BT and EBRT, but both were higher than RP. This may reflect differences on how PSA failures were defined, as the definition for failure is more sensitive for RP compared with EBRT.12,27 Also, the RP group had more failures in cRFS, as RP patients who underwent adjuvant radiation were counted as failures despite not having failed biochemically.
The main reason for the benefit of BT is that it provides more ablative doses than can be achieved by EBRT with or without NADT by producing a lower PSA nadir of <0.1.28 Although ablative doses may be effective in eradicating prostate cancer, high doses can cause significant morbidity. However, one can properly select patients who can tolerate these ablative doses by using the American Urological Association urinary score to select which patients would have a lower probability of long-term urinary effects.29 In the current study, we had acceptable side effects, and these side effects were more common in those undergoing S-EBRT, although most of these were grades 1 and 2, with severe grade 3 and 4 complications rates being low.
The limitations of this study include that the majority of patients were U-IRPC, with only 20.5% being HRPC. Also, there was an imbalance of GS and PPBC > 50%, with worse patients in the S-EBRT group, showing that the BT-alone patients were subjected to selection bias. We tried to account for these differences by using propensity score matching, although this is a relatively small study with the limitation of being retrospective. Unfortunately, there are few large randomized trials using prostate BT. Low reimbursements, combined with LDR BT requiring more training and skill, give little motivation for physicians to offer LDR BT to their patients.30-32 This has led to a decline in the use of LDR BT in clinical practice and residency training, with the potential downstream effect of fewer publications on the role of LDR BT in patients with prostate cancer, and potentially leading to the unavailability to many patients of one of the most successful treatment options for prostate cancer. Thus, we publish our 10-year results on the use of LDR BT on patients with more advanced disease, which, if done properly, can yield favorable oncological outcomes with acceptable rates of side effects, suggesting LDR BT to be a reasonable option in the treatment of U-IRPC and selected HRPC.
Conclusion
LDR BT using iodine-125 alone is a reasonable treatment option for U-IRPC and favorable HRPC, which is effective, convenient, and cost-effective.
References
- Blasko J, Grimm P, Sylvester J. Palladium-103 brachytherapy for prostate carcinoma. Int J Radiat Oncol Biol Phys. 2000;46(4):839-850. doi:doi:10.1016/s0360-3016(99)00499-x.
- Grimm P, Blasko J, Sylvester J, Meier R, Cavanagh W. 10-year biochemical (prostate-specific antigen) control of prostate cancer with 125-I brachytherapy. Int J Radiat Oncol Biol Phys. 2001;51(1):31-40. doi:doi:10.1016/s0360-3016(01)01601-7.
- Lawton C, Hunt D, Lee W. Long-term results of a phase II trial of ultrasound-guided radioactive implantation of the prostate for definitive management of localized adenocarcinoma of the prostate (RTOG 98-05). Int J Radiat Oncol Biol Phys. 2011;81(1):1-7. doi:doi:10.1016/j.ijrobp.2010.05.056.
- .
- Goy B, Burchette R, Soper M, Chang T, Cosmatos H. Ten-year treatment outcomes of radical prostatectomy vs external beam radiation therapy vs brachytherapy for 1503 patients with intermediate risk prostate cancer. Urology. 2020;136:180-189. doi:doi:10.1016/j.urology.2019.09.040.
- Greene F, Page D, Fleming I. AJCC Cancer Staging Handbook. 2002. doi:doi:10.1007/978-1-4757-3656-4.
- Zumsteg Z, Spratt D, Pei I. A new risk classification system for therapeutic decision making with intermediate-risk prostate cancer patients undergoing dose-escalated external-beam radiation therapy. Eur Urol. 2013;64(6):895-902. doi:doi:10.1016/j.eururo.2013.03.033.
- D’Amico A, Whittington R, Malkowicz S. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969-974. doi:doi:10.1001/jama.280.11.969.
- Charlson M, Pompei P, Ales K, MacKenzie C. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-383. doi:doi:10.1016/0021-9681(87)90171-8.
- Grimm P, Blasko J, Ragde H. Ultrasound-guided transperineal implantation of Iodine-125 and palladium-103 for the treatment of early stage prostate cancer. Atlas Urol Clin of North Am. 1994;2:113-125.
- Nath R, Anderson L, Luxton G. Dosimetry of interstitial brachytherapy sources: recommendations of the AAPM radiation therapy committee task group No.43. Med Phys. 1995;22(2):209-234. doi:doi:10.1118/1.597458.
- Roach M, Hanks G, Thames H. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO consensus conference. Int J Radiat Oncol Biol Phys. 2006;65(4):965-974. doi:doi:10.1016/j.ijrobp.2006.04.029.
- LENT SOMA scales for all anatomic sites. Int J Radiat Oncol Biol Phys. 1995;31(5):1049-1091. doi:doi:10.1016/0360-3016(95)90159-0.
- Kaplan E, Meier P. Non-parametric estimation from incomplete observations. J Am Stat Assoc. 1958;53(282):457-481. doi:doi:10.1080/01621459.1958.10501452.
- Cox D. Regression models and life tables. J R Stat Soc Series B Stat Methodol. 1972;34(2):187-202. doi:doi:10.1111/j.2517-6161.1972.tb00899.x.
- Rosenbaum P, Rubin D. Reducing bias in observation studies using subclassification on the propensity score. J Am Stat Assoc. 1984;79(387):516-524. doi:doi:10.1080/01621459.1984.10478078.
- Roach M, Moughan J, Lawton C. Sequence of hormonal therapy and radiotherapy field size in unfavourable, localized prostate cancer (NRG/RTOG 9413): long-term results of a randomized, phase 3 trial. Lancet Oncol. 2018;19(11):1504-1515. doi:doi:10.1016/S1470-2045(18)30528-X.
- Blanchard P, Faivre L, Lesaunier F. Outcome according to elective pelvic radiation therapy in patients with high-risk localized prostate cancer: a secondary analysis of the GETUG 12 phase 3 randomized trial. Int J Radiat Oncol Biol Phys. 2016;94(1):85-92. doi:doi:10.1016/j.ijrobp.2015.09.020.
- Murthy V, Maitre P, Kannan S. Prostate-only versus whole-pelvic radiation therapy in high-risk and very-high risk prostate cancer (POP-RT): outcomes from phase III randomized controlled trial. J Clin Oncol. 2021;39(11):1234-1242. doi:doi:10.1200/JCO.20.03282.
- Ciezki J. High-risk prostate cancer in the modern era: does a single standard of care exist. Int J Radiat Oncol Biol Phys. 2013;87(3):440-442. doi:doi:10.1016/j.ijrobp.2013.06.006.
- Michalski J, Winter K, Prestidge B. Effect of brachytherapy with external beam radiation therapy versus brachytherapy alone for intermediate-risk prostate cancer: NRG oncology RTOG 0232 randomized clinical trial. J Clin Oncol. 2023;41(24):4035-4044. doi:doi:10.1200/JCO.22.01856.
- Morris W, Tyldesley S, Rodda S. Androgen suppression combined with elective nodal and dose escalated radiation therapy (the ASCENDE-RT trial): an analysis of survival endpoints for a randomized trial comparing a low-dose-rate brachytherapy boost to a dose-escalated external beam boost for high- and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2017;98(2):275-285. doi:doi:10.1016/j.ijrobp.2016.11.026.
- Rodda S, Morris W, Hamm J, Duncan G. ASCENDE-RT: an analysis of treatment-related morbidity for a randomized trial comparing a low-dose-rate brachytherapy boost with a dose-escalated external beam boost for high- and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2017;98(3):581-589. doi:doi:10.1016/j.ijrobp.2017.02.027.
- Merrick G, Wallner K, Galbreath R, Butler W, Adamovich E. Is supplemental external beam radiation therapy essential to maximize brachytherapy outcomes in patients with unfavorable intermediate-risk disease?. Brachytherapy. 2016;15(1):79-84. doi:doi:10.1016/j.brachy.2015.09.011.
- Yang D, Muralidhar V, Nguyen P. Lack of benefit from the addition of external beam radiation therapy to brachytherapy for Intermediate- and high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2017;99(4):904-911. doi:doi:10.1016/j.ijrobp.2017.07.034.
- Ciezki J, Weller M, Reddy C. A comparison between low-dose-rate brachytherapy with or without androgen deprivation, external beam radiation therapy with or without androgen deprivation, and radical prostatectomy with or without adjuvant or salvage radiation therapy for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2017;97(5):962-975. doi:doi:10.1016/j.ijrobp.2016.12.014.
- Cookson M, Aus G, Burnett A. Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American urological association prostate guidelines for localized prostate cancer update panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol. 2007;177(2):540-545. doi:doi:10.1016/j.juro.2006.10.097.
- Goy B, Soper M, Chang T. Treatment results of brachytherapy vs external beam radiation therapy for intermediate-risk prostate cancer with 10-year follow up. Brachytherapy. 2016;15(6):687-694. doi:doi:10.1016/j.brachy.2016.06.015.
- Goy B, Burchette R. Ten-year complication outcomes of radical prostatectomy vs external beam radiation therapy vs brachytherapy for 1,503 patients with intermediate risk prostate cancer. Brachytherapy. 2021;20(6):1083-1089. doi:doi:10.1016/j.brachy.2021.04.004.
- Schad M, Kowalchuk R, Beriwal S, Showalter T. How might financial pressures have impacted brachytherapy? A proposed narrative to explain the declines in cervical and prostate brachytherapy utilization. Brachytherapy. 2019;18(6):780-786. doi:doi:10.1016/j.brachy.2019.07.001.
- Petereit D, Frank S, Viswanathan A. Brachytherapy: where has it gone?. J Clin Oncol. 2015;33(9):980-982. doi:doi:10.1200/JCO.2014.59.8128.
- Yashar C. Stormy “fourcast” for brachytherapy. Int J Radiat Oncol Biol Phys. 2015;93(4):733-736. doi:doi:10.1016/j.ijrobp.2015.06.022.
Disclosures
The authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere.
Citation
. Unfavorable Intermediate- and High-Risk Prostate Cancer Treated With Predominantly Brachytherapy Alone With Long-Term Follow-Up. Applied Radiation Oncology. 2023;12(4):20-28. doi:10.37549/ARO-D-23-00023.