UK’s MHRA Innovation Passport Awarded to Theragnostics for Glioblastoma Multiforme Therapeutic Agent
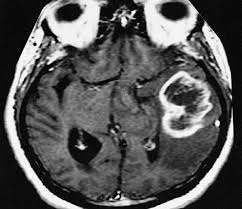 Theragnostics’ novel I-123 PARPi (THG-009) therapeutic agent has been awarded an Innovation Passport by the UK Medicines and Healthcare products Regulatory Agency (MHRA) for the treatment of primary and recurrent Glioblastoma Multiforme (GBM). This is provided under the MHRA's Innovative Licensing and Access Pathway (ILAP) which was launched in January 2021 to accelerate development and access to innovative medicines in the UK.
Theragnostics’ novel I-123 PARPi (THG-009) therapeutic agent has been awarded an Innovation Passport by the UK Medicines and Healthcare products Regulatory Agency (MHRA) for the treatment of primary and recurrent Glioblastoma Multiforme (GBM). This is provided under the MHRA's Innovative Licensing and Access Pathway (ILAP) which was launched in January 2021 to accelerate development and access to innovative medicines in the UK.
Theragnostics is developing I-123 PARPi for the treatment of primary and recurrent glioblastoma. Glioblastoma has a high rate of recurrence in patients of approximately 90% and has a poor overall prognosis of survival of less than two years in most patients. There have been no new agents approved for this indication in the last 15 years. Theragnostics' early non-clinical data have demonstrated promising results which support progression of I-123 PARPi into a Phase 1 clinical study in recurrent GBM.
The Innovation Passport is awarded to companies developing therapeutics for conditions that are life-threatening or seriously debilitating and has the potential to offer significant benefits to patients. It acts as the gateway to enter the ILAP and is a new medicine designation. The next step in the pathway is the preparation of a target development profile (TDP) document by the MHRA, National Institute for Health and care Excellence (NICE) and the Scottish Medicines Consortium (SMC). The TDP is a 'living document' which sets out the regulatory and development milestones, finds potential pitfalls and creates a roadmap to achieving early patient access in the UK. The TDP can aid pharmaceutical companies with clinical trial design and evidence generation.
"The Innovation Passport will allow us to work closely with the MHRA and its partner agencies to chart out a roadmap for regulatory and key development milestones with the primary goal of achieving early patient access for the treatment of glioblastoma," said Dr. Greg Mullen, Chief Executive Officer of Theragnostics.