Rectal Dosimetry of Different Rectal Displacement Devices for Prostate External Beam Radiation Therapy: A Multi-Institutional Retrospective Cohort Study
Applied Radiation Oncology
Published: March 5, 2026
1 University of California, Santa Barbara, Santa Barbara, CA, USA
2 University of Nevada, Reno School of Medicine, Reno, NV, USA
3 Northern Nevada Radiation Oncology, Reno, NV, USA
4 Radiation Oncology Department, CUF Porto Institute, Porto, Portugal
* Corresponding author: Michael Kos (mkos321@gmail.com)
Abstract
Keywords
prostate, cancer, spacer, polyethylene glycol gel, inflatable balloon, PEG, IB, implant, radiation therapy
Introduction
According to the American Cancer Society, approximately 1 in 8 men are diagnosed with prostate cancer in their lifetime, with incidence increasing with age. 1 With an estimated 1.2 million cases reported in 2018, prostate cancer is the second-most common neoplasm worldwide, second to lung cancer. 1 In the United States, the overall survival rate as judged by Surveillance, Epidemiology and End Results (SEER) data for all stages is generally good, about 98% at 5 years. 2 The disease is reportedly responsible for 34,500 deaths annually in the United States, or 5.5% of US cancer deaths vs 6.7% worldwide cancer deaths. 1
Treatment of localized disease generally consists of surgery or radiation, with advanced treatment comprising hormonal therapy, chemotherapy, and radiation. Quality of life (QOL) varies by stage and treatment, but most men experience a lower QOL due to worsening genitourinary function; that is, urinary incontinence and erectile dysfunction. 3,4
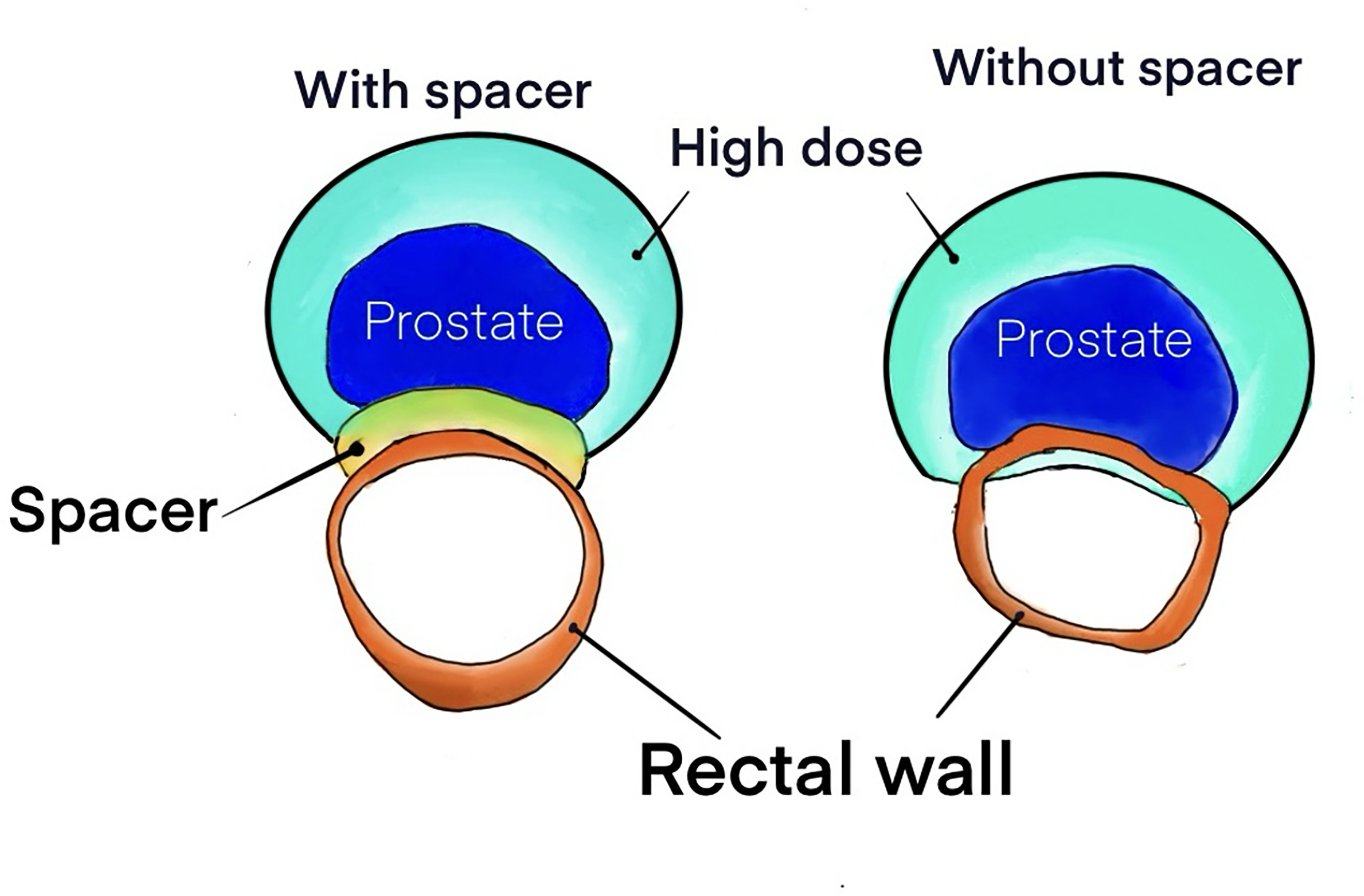
Radiation therapy (RT) is a primary treatment modality for prostate cancer; dose escalation improves local control but is limited by the potential for complications in nearby organs at risk. RT may produce more favorable genitourinary outcomes compared to surgical options, but bowel complications such as increased urgency and/or frequency, diarrhea, and rectal bleeding are much more frequent. 3-5 These complications usually improve within 6 months to a year of treatment initiation, according to prior studies, but they are associated with higher incidences of long-term gastrointestinal issues. 4,5 Rectal bleeding is a common side effect of prostate RT. 6,7 If prevented or minimized, this can greatly improve QOL in the short and long term. Attenuating the radiation dosage to the rectum has been shown to reduce side effects associated with excess radiation and has been made possible by recent advances in target localization technology. 5 Radiation dose to the rectum can be reduced by physically displacing the rectum from high-dose regions and optimizing dose distributions to reduce rectal dose-volume histogram (DVH) endpoints. 8 See Figure 1 for an example of how a PEG spacer displaces the rectum. Various rectal displacement devices (RDDs) are used to increase the space between the rectum and prostate. The 2 main types are polyethylene glycol (PEG) gel and an inflatable balloon (IB) placed between the prostate and rectum. PEG (Boston Scientific, USA) polymerizes to form a semi-solid gel. The BioProtect Balloon System (BioProtect, Israel) uses a balloon inflated with sterile water. Both technologies have been shown to reduce rectal dose. 9-12
Rectal implants continue to reduce late QOL impacts of prostate RT at 3 years post-treatment (9.2% vs 2.0% incidence of grades > 1 for control and spacer, respectively, and 5.7% vs 0% for grades > 2). 13 Another device that has entered the market inserts a hyaluronic acid gel that has been shown effective in a randomized controlled trial. 14
In this study, we analyzed the dose-sparing capabilities of 2 types of RDD: PEG implants and IB implants. By measuring the received dose to the rectal wall for both implant-treated patients and control patients, we present the mean received dose and dose reduction for both implants. We compare PEG and IB implants and discuss relevant clinical differences between the 2 modalities.
Materials and Methods
Patient Selection
Patients with early-/intermediate-stage prostate cancer undergoing external RT treatment between 2013 and 2020 with a spacer were enrolled in a 2-institution, retrospective cohort study. Patients who had previously undergone RT, prostatectomy, or pelvic lymph node radiation were excluded. Seminal vesicle involvement was not an exclusion factor. Controls received no RDD and were selected from consecutive cases in the same time frame when spacer modalities were not as readily available. Many of the IB patient data were collected from an international center owing to the IB’s approval for use in the European Union (EU) as opposed to its pending approval in the United States.
A total of 283 patients, mean age 73 years (range 50-90 years), were analyzed. A total of 99 US patients were implanted with PEG, 92 patients (17 US patients, 75 EU patients) were implanted with IB, and 92 US patients were not implanted with either rectal spacer.
As shown in Table 1, the patient groups demonstrated no significant differences with respect to age, mean prescribed dose, and mean rectal volume, with standard error reported for all mean values. Patient demographic and fractionation data were tested for statistical significance using the Kruskal-Wallis test.
| Total | PEG | IB | Control | P value | |
|---|---|---|---|---|---|
| Demographic data | |||||
| Age at treatment (years) | 73.2±6.6 | 72.1±5.8 | 73.7±7.0 | 73.8±7.0 | 0.13 |
| Prostate volume (cc) | 75.2±32.4 | 65.8±27.9 | 94.7±33.9 | 65.9±26.4 | <.001 |
| Rectum volume (cc) | 68.5±26.0 | 60.7±22.0 | 74.5±26.3 | 70.8±28.0 | 0.15 |
| Treatment data | |||||
| Prescribed dose (Gy) | 77.6±5.4 | 75.4±5.5 | 77.1±6.2 | 80.3±2.7 | 0.39 |
| Number of fractions | 39.1±7.5 | 36.4±8.5 | 37.2±6.5 | 43.9±4.2 | <.001 |
| Dose/fraction (Gy) | 2.0±0.3 | 2.2±0.4 | 2.1±0.3 | 1.8±0.2 | <.001 |
Data Collection
In all cases, CT scans were collected from patients after spacer placement and before treatment. Radiation exposure to the rectum was calculated based on previously recorded volumetric and dosimetry data for all patients. Control patients consisted of those treated prior to common use of PEG, with data collected in the same fashion. The primary endpoints were rectal dosimetry in D0.1cc, D1cc, and D2cc, where D0.1cc refers to the dose given to the hottest 0.1 cc of the rectum.
Treatment Planning
Target volume margins were defined with planning target volume (PTV) expansions of 5 mm applied in all directions from the clinical target volume (CTV) except posteriorly, where a reduced 3 mm margin was implemented for the boost phase to minimize rectal dose exposure.
Fiducial marker systems differed between institutions: electromagnetic transponders (Calypso) were used at the US center; radiopaque gold seed markers were employed at the EU center. These differences in marker composition did impact target treatment planning. The PTV to CTV expansions were different for US vs international patients. For international patients, the expansion was 5 mm in the X, Y, and Z axes. For US patients, the expansion was 3.0 mm posteriorly for the initial volumes and then 3.0 mm in the X, Y, and Z axes for the boost component. Given that the majority of gel implants were in the United States, this strongly favored the gel implant dosimetry.
Equivalent Dose in 2 Gy Fractions
See Table 1 for prescribed dose and fractionation schemes. To compare patients with different treatment fractionation schemes, all received doses were converted to equivalent dose in 2 Gy fractions (EQD2) using the following formula:
where D is the total prescribed dose in Gy and d is the dose per fraction in Gy. 15 For this study, an α/β of 3 was used for late rectal toxicity endpoints. 16
Statistical and Data Analysis
All statistical analyses were conducted in Python 3.10 using SciPy and pandas libraries. Prior to comparative analysis, normality of dosimetric data was assessed using Shapiro-Wilk and Jarque-Bera tests for all DVH parameters across treatment groups. For D0.1cc and D1cc, all groups showed deviation from normality (P < .05); for D2cc, 1 of 3 groups approximated normality. Levene test was used to assess homogeneity of variance across groups, revealing variance heterogeneity across all DVH parameters (all P < .05).
Given the non-normal distributions and variance heterogeneity, nonparametric methods were employed for robust inference. Kruskal-Wallis H-tests were used for omnibus testing of group differences across all DVH parameters, followed by Mann-Whitney U tests for post hoc pairwise comparisons with Bonferroni correction (α = .0167) to control family-wise error rate across 3 pairwise comparisons. 17
The percent difference between cohort groups was calculated using the symmetric formula:
Results
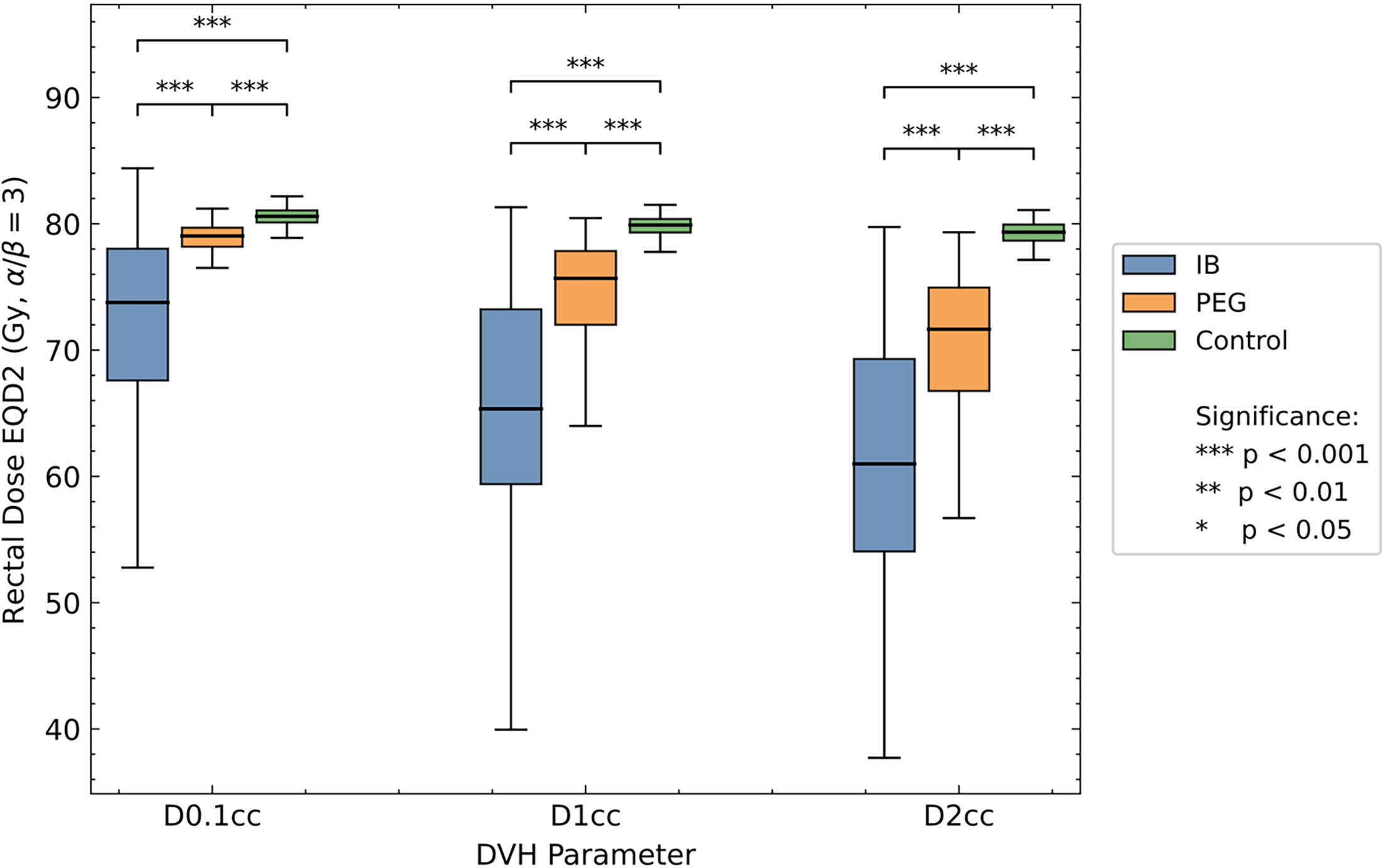
For each patient, D0.1cc, D1cc, and D2cc were collected and analyzed, with all doses converted to EQD2. See Table 2 for analyzed dosimetry data for all patient groups.
| Dose-Volume Histogram Parameter | IB | PEG | Control | P value |
|---|---|---|---|---|
| D0.1cc (Gy) | 71.7±8.1 | 78.1±3.5 | 80.4±1.2 | <.001 |
| D1cc (Gy) | 65.2±9.5 | 73.7±6.0 | 79.5±2.1 | <.001 |
| D2cc (Gy) | 61.2±9.9 | 69.9±7.1 | 78.7±2.7 | <.001 |
Kruskal-Wallis tests revealed highly significant differences among treatment groups across all DVH parameters (all P < .001), with large effect sizes (η²H range: 0.68-0.72, all indicating large effects per conventional thresholds). All pairwise effect sizes indicated large effects (Cohen d = 1.01-1.07; threshold for large effects d > 0.8).
See Figure 2 for a comparison of rectal DVH data from all groups. See Figure 3 for percent difference data between cohorts. For the IB implant, we found a mean received D0.1cc of 71.7 Gy (SD = 8.1 Gy) and dose reduction of 11.5% compared to the control cohort (P < .001), and dose reduction of 8.5% compared to patients receiving the PEG implant (P < .001). We found a mean received D1cc of 65.2 Gy (SD = 9.5 Gy) and dose reduction of 19.7% compared to control patients (P < .001), and dose reduction of 12.2% compared to patients receiving the PEG implant (P < .001). We found a mean received D2cc of 61.2 Gy (SD = 9.9 Gy) and dose reduction of 25% compared to the control cohort (P < .001), and dose reduction of 13.2% compared to patients receiving the PEG implant (P < .001).
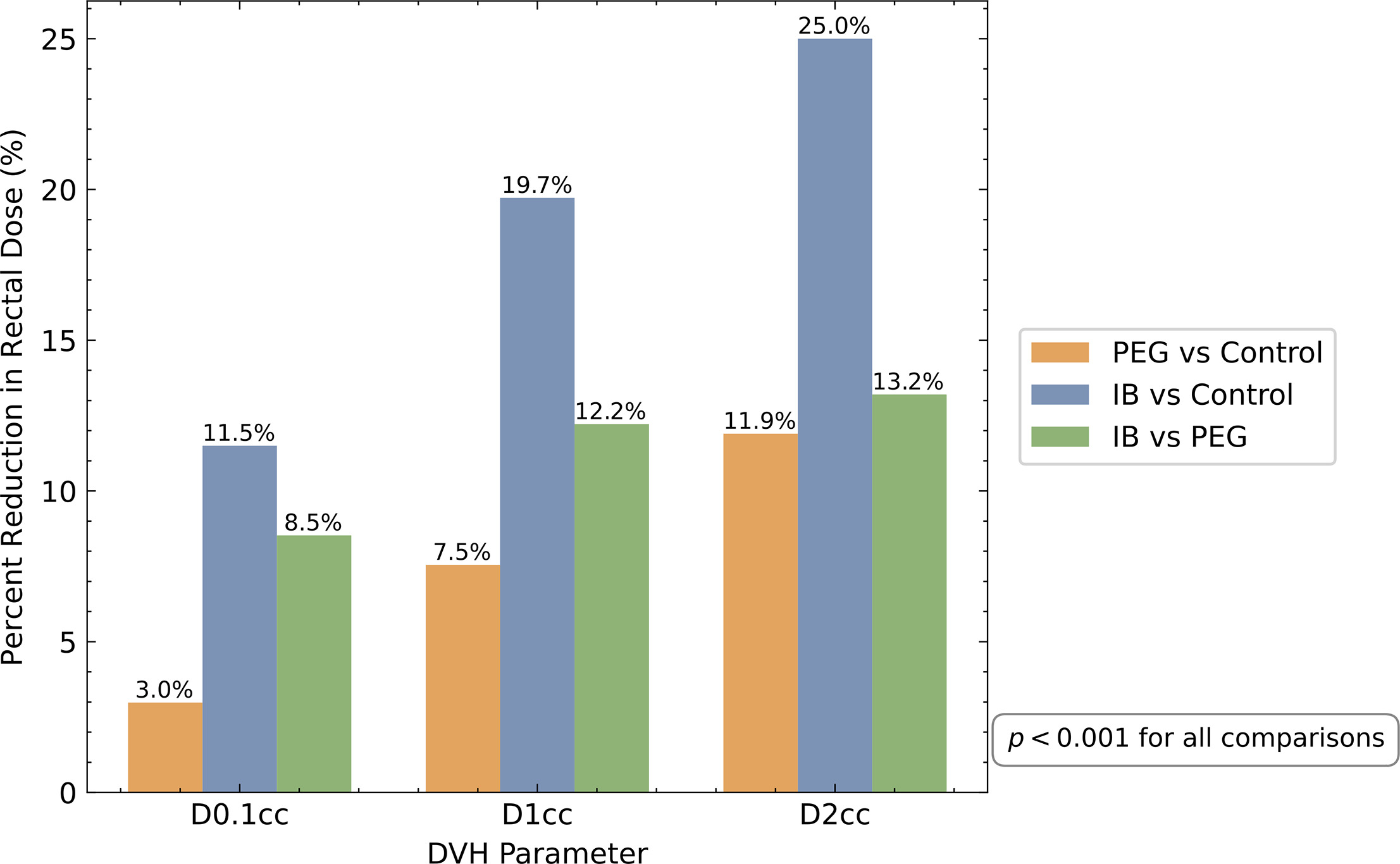
For the PEG implant, we found a mean received D0.1cc of 78.1 Gy (SD = 3.5 Gy) and dose reduction of 3% compared to the control cohort (P < .001). We found a mean received D1cc of 73.7 Gy (SD = 6.0 Gy) and dose reduction of 7.5% compared to the control cohort (P < .001). We found a mean received D2cc of 69.9 Gy (SD = 7.1 Gy) and dose reduction of 11.9% compared to the control cohort (P < .001).
Discussion
This multi-institutional retrospective cohort study compared rectal dose-sparing capabilities of 2 RDD technologies in 283 patients undergoing prostate external beam RT. Our findings demonstrate that PEG gel and IB implants significantly reduce radiation dose to the rectum compared to controls, with the IB device providing superior dose reduction across all DVH parameters.
Clinical Significance of Dosimetric Findings
The IB implant achieved substantial dose reductions of 11.5%, 19.7%, and 25% for D0.1cc, D1cc, and D2cc, respectively, compared to controls (all P < .001). These reductions translate to mean doses of 71.7 Gy, 65.2 Gy, and 61.2 Gy (EQD2) for the respective parameters. The PEG implant, while also providing statistically significant dose reduction (3%, 7.5%, and 11.9% for D0.1cc, D1cc, and D2cc; all P < .001), demonstrated more modest dose-sparing, with mean values of 78.1 Gy, 73.7 Gy, and 69.9 Gy.
Direct comparison between devices revealed the IB implant reduced doses by 8.5%, 12.2%, and 13.2% relative to PEG across all 3 parameters (all P < .001).
These findings agree with prior studies showing that both implant technologies significantly reduce radiation dose to the rectum. 9,10 A previous study also agrees that the IB implant is superior to PEG in reducing the rectal dose. 18
The progressively greater dose-sparing effect observed at larger volumes (D2cc > D1 cc > D.1 cc) for the IB device likely reflects superior spatial symmetry. Well-positioned IB implants typically create 1.7 to 2.0 cm separation between rectum and prostate. Suboptimal PEG symmetry may result in incomplete coverage, where small rectal volumes receive higher doses despite larger volumes benefiting from displacement.
Product Comparison
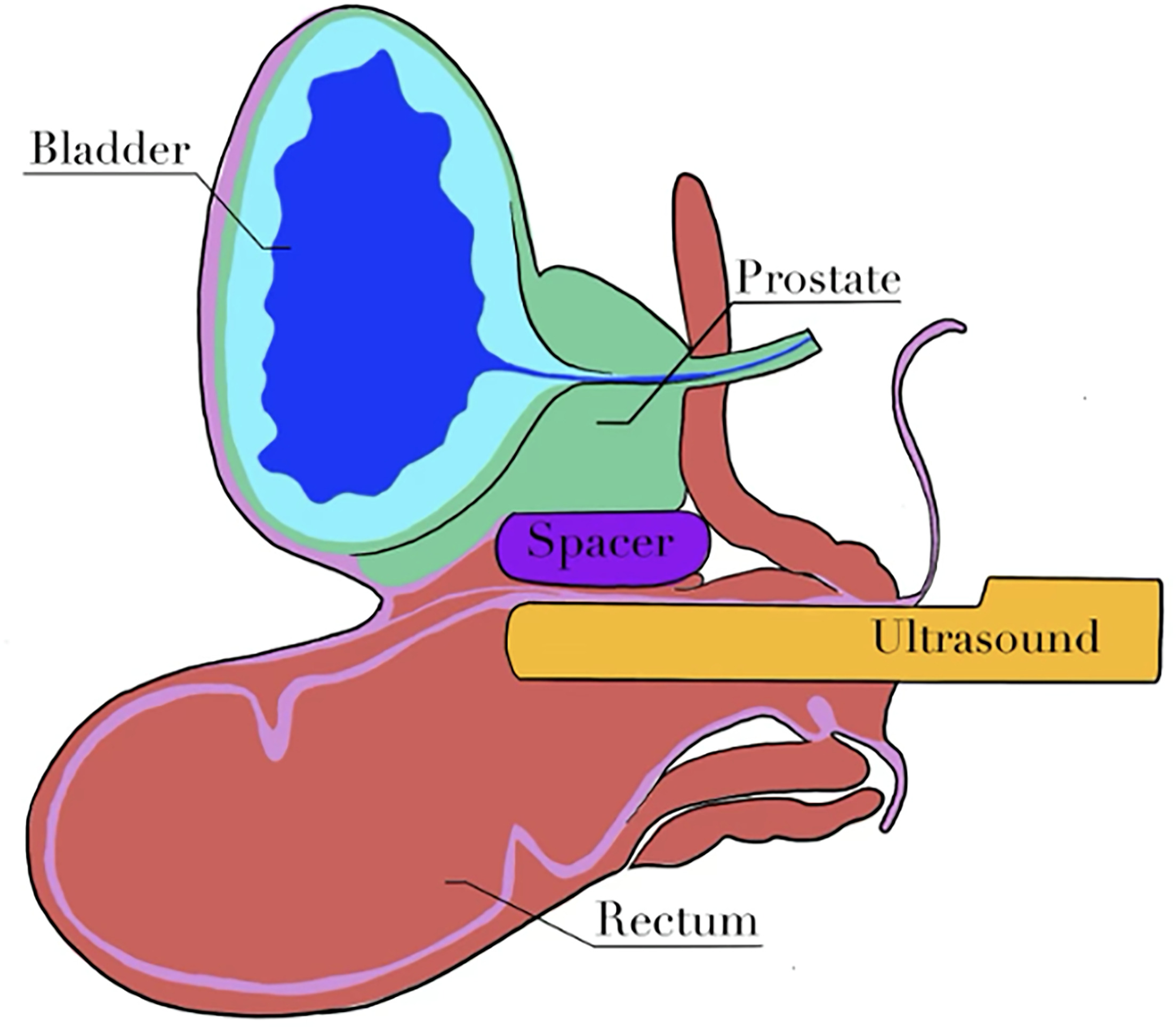
Beyond these dosimetric distinctions, the 2 devices also differ substantially in their insertion techniques and procedural requirements, see Table 3. Insertion procedures of the PEG and IB are relatively quick, typically performed after beacon placement using transrectal ultrasound-guided needle or instrument into the perirectal space. See Figure 4 for a depiction of the ultrasound-guided implantation procedure. PEG uses hydrodissection followed by a 10 to 12 mL gel injection. IB requires blunt dissection, insertion of the apparatus and balloon, then filled with 16 to 20 mL sterile water. The IB device tip is blunt, requiring more force but is less likely to perforate the rectum. 19 The IB allows opportunities to inflate, deflate, and reposition the balloon, which can improve symmetry. Poor spacer symmetry results in less rectal dose reduction but still offers significant reduction. 20 The separation between the rectum and prostate is typically 1.7 to 2.0 cm with the IB implant. More bleeding usually occurs with IB (typically < 5 mL), but lidocaine administration nearly eliminates pain. The balloon is easily visible on MRI, obviating the need for contrast.
| Characteristic | IB | PEG |
|---|---|---|
| Implant size (cc) | 16-20 | 10-12 |
| Imaging modalities where implant is visible | MRI, CT | MRI, CT when contrast is added |
Limitations
The limitations of this study include bias with respect to physician experience; greater experience generally improves outcomes. In this study, both physicians had little experience with IB placements, but PEG data were aggregated from one physician who had performed over 500 placements. Complication rates and symmetry after PEG placement have demonstrated improvement after the first 20 cases. 21 The IB data based on the first 13 cases likely underestimate the technology’s potential performance. Another bias relates to prostate volume differences (mean 95 cc in IB group vs 66 cc in others). Randomization would have been ideal, but both devices are rapidly evolving. Beyond PEG and perirectal balloons, hyaluronic acid spacers have demonstrated rectal dose-sparing with supportive prospective evidence. Hyaluronic acid spacers have also demonstrated rectal dose-sparing and are now commercially available. 14 Future comparison studies that include this RDD type are warranted.
Conclusion
We report the dosimetric analysis of 2 RDDs, a PEG gel implant and an IB implant, for reduction of radiation dose to the rectum during prostate cancer treatment using external beam RT. Both implants are well-tolerated, with a low but possible risk of complications. 14 Both demonstrated statistically significant reduction of received dose to the rectum compared to the absence of implants in control patients. The IB implant demonstrated superior dose reduction to the rectum than the PEG implant. RDDs are a new technology and are under continued development and improvement. Further investigation and comparison of RDDs is warranted as technology improves.
References
- Barsouk A, Padala S, Vakiti A. Epidemiology, staging and management of prostate cancer. Med Sci. 2020;8(3):28. doi:10.3390/medsci8030028.
- Rawla P. Epidemiology of prostate cancer. World J Oncol. 2019;10(2):63-89. doi:10.14740/wjon1191.
- Eton D, Lepore S. Prostate cancer and health-related quality of life: a review of the literature. Psychooncology. 2002;11(4):307-326. doi:10.1002/pon.572.
- Taylor J, Chen V, Miller R, Greenberger B. The impact of prostate cancer treatment on quality of life: a narrative review with a focus on randomized data. Res Rep Urol. 2020;12:533-546. doi:10.2147/RRU.S243088.
- Serrano N, Kalman N, Anscher M. Reducing rectal injury in men receiving prostate cancer radiation therapy: current perspectives. Cancer Manag Res. 2017;9:339-350. doi:10.2147/CMAR.S118781.
- Bae B, Kim J, Pyo H, Hong S, Park W. Long-term findings of rectal endoscopy and rectal bleeding after moderately hypofractionated, intensity-modulated radiotherapy for prostate cancer. Sci Rep. 2023;13(1):22099. doi:10.1038/s41598-023-43202-x.
- Schaldemose E, Madsen C, Zedan A. Risk factors for rectal bleeding in prostate cancer after radiotherapy with a validation of current rectal dose constraints. Acta Oncol. 2025;64. doi:10.2340/1651-226X.2025.42551.
- Susil R, McNutt T, DeWeese T, Song D. Effects of prostate-rectum separation on rectal dose from external beam radiotherapy. Int J Radiat Oncol Biol Phys. 2010;76(4):1251-1258. doi:10.1016/j.ijrobp.2009.07.1679.
- Mariados N, Sylvester J, Shah D. Hydrogel spacer prospective multicenter randomized controlled pivotal trial: dosimetric and clinical effects of perirectal spacer application in men undergoing prostate image guided intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys. 2015;92(5):971-977. doi:10.1016/j.ijrobp.2015.04.030.
- Melchert C, Gez E, Bohlen G. Interstitial biodegradable balloon for reduced rectal dose during prostate radiotherapy: results of a virtual planning investigation based on the pre- and post-implant imaging data of an international multicenter study. Radiother Oncol. 2013;106(2):210-214. doi:10.1016/j.radonc.2013.01.007.
- Quinn T, Daignault-Newton S, Bosch W. Who benefits from a prostate rectal spacer? Secondary analysis of a phase III trial. Pract Radiat Oncol. 2020;10(3):186-194. doi:10.1016/j.prro.2019.12.011.
- Song D, Dabkowski M, Costa P. Prospective, randomized controlled pivotal trial of biodegradable balloon rectal spacer for prostate radiation therapy. Int J Radiat Oncol Biol Phys. 2024;120(5):1410-1420. doi:10.1016/j.ijrobp.2024.07.2145.
- Hamstra D, Mariados N, Sylvester J. Continued benefit to rectal separation for prostate radiation therapy: final results of a phase III trial. Int J Radiat Oncol Biol Phys. 2017;97(5):976-985. doi:10.1016/j.ijrobp.2016.12.024.
- Mariados N, Orio P, Schiffman Z. Hyaluronic acid spacer for hypofractionated prostate radiation therapy: a randomized clinical trial. JAMA Oncol. 2023;9(4):511-518. doi:10.1001/jamaoncol.2022.7592.
- Voyant C, Julian D, Roustit R, Biffi K, Lantieri C. Biological effects and equivalent doses in radiotherapy: a software solution. Rep Pract Oncol Radiother. 2014;19(1):47-55. doi:10.1016/j.rpor.2013.08.004.
- Brand D, Brüningk S, Wilkins A. Estimates of alpha/beta (α/β) ratios for individual late rectal toxicity endpoints: an analysis of the chhip trial. Int J Radiat Oncol Biol Phys. 2021;110(2):596-608. doi:10.1016/j.ijrobp.2020.12.041.
- Bensken W, Ho V, Pieracci F. Basic introduction to statistics in medicine, part 2: comparing data. Surg Infect. 2021;22(6):597-603. doi:10.1089/sur.2020.430.
- Wolf F, Gaisberger C, Ziegler I. Comparison of two different rectal spacers in prostate cancer external beam radiotherapy in terms of rectal sparing and volume consistency. Radiother Oncol. 2015;116(2):221-225. doi:10.1016/j.radonc.2015.07.027.
- Schörghofer A, Drerup M, Kunit T. Rectum-spacer related acute toxicity - endoscopy results of 403 prostate cancer patients after implantation of gel or balloon spacers. Radiat Oncol. 2019;14(1):47. doi:10.1186/s13014-019-1248-6.
- Fischer-Valuck B, Chundury A, Gay H, Bosch W, Michalski J. Hydrogel spacer distribution within the perirectal space in patients undergoing radiotherapy for prostate cancer: impact of spacer symmetry on rectal dose reduction and the clinical consequences of hydrogel infiltration into the rectal wall. Pract Radiat Oncol. 2017;7(3):195-202. doi:10.1016/j.prro.2016.10.004.
- Pinkawa M, Klotz J, Djukic V. Learning curve in the application of a hydrogel spacer to protect the rectal wall during radiotherapy of localized prostate cancer. Urology. 2013;82(4):963-968. doi:10.1016/j.urology.2013.07.014.
Disclosures
Harkness, Field, Zhang, and Vale report no conflicts of interest. Costa and Kos report serving as principal investigators and clinical proctors and being paid for each procedure and clinical training during the trial.
Citation
. Rectal Dosimetry of Different Rectal Displacement Devices for Prostate External Beam Radiation Therapy: A Multi-Institutional Retrospective Cohort Study. Applied Radiation Oncology. 2026. doi:10.37549/ARO-D-25-00016.