Radiation Therapy for Dupuytren Disease: A Systematic Review of Clinical Outcomes and Adverse Effects
Applied Radiation Oncology
Published: March 12, 2026
1 Department of Radiation Oncology, University of Minnesota Medical School, Minneapolis, MN
* Corresponding author: Leslie Chang (lechang@umn.edu)
Abstract
Keywords
Dupuytren, palmar, radiation, irradiation, fibroproliferative disorder
Introduction
Dupuytren disease (DD), a benign fibroproliferative disorder, affects an estimated 12% to 29% of individuals aged 55 to 75 years in Western countries. 1 It is more common and severe in men around the age of 50, with women typically developing a severe onset later on in life.
DD presents initially in a singular hand, with 80% of affected individuals eventually having features of DD in both hands. 2 Symptoms of fibrosis can occur in any location but are most common in the middle of the palm, progressing to the 4th and 5th fingers. Once one or more nodules form in the palm, they are subsequently followed by cords that develop into the distinctive digital contractures. 3 About 25% of people with DD experience discomfort or pain, with inflammation, tenderness, burning, or itching often associated with affected joints and areas of skin. 2
The main cause of DD is not yet known. However, the development of fibrosis is associated with inflammation, and several proinflammatory cytokines have been associated with DD, including the elevated expression of TGF-β1, α-SMA, and canonical Wnt signaling. 4 DD is also associated with environmental factors such as smoking, alcohol consumption, aging, trauma, and repetitive use of the hand in various physical occupations. 3,5,6 There is ongoing evaluation of genetic risk factors, with a recent meta-analysis of 6 genome-wide association studies identifying approximately 85 single-nucleotide polymorphisms in 56 loci, implicating the hedgehog and Notch signaling pathways in DD disease etiology. 7 DD is associated with other disorders of the connective tissue, including Garrod pads, Ledderhose disease (plantar fibromatosis), and Peyronie disease in males. 2
The early stages of this disease, characterized by nodule formation without contracture, currently lack standardized clinical criteria or treatment options. Surgical intervention, the mainstay of DD treatment, is considered when the patient’s finger has lost 30° of metacarpophalangeal or interphalangeal extension of the joint. 8 Surgery is not typically advocated for early stages of the disease unless it is accompanied by persistent pain or discomfort, as the early proliferative stage with high cellularity has been associated with higher rates of recurrence. 9 Other treatment options range from minimally invasive to extensive surgical approaches. Collagenase clostridium histolyticum (Xiaflex) offers a nonoperative option through the enzymatic weakening of the cord. However, as a nonspecific collagenase, its mechanism can affect adjacent collagen-containing tissues and contribute to local adverse effects. Steroid injections such as triamcinolone acetonide are injected directly into nodules. 10 Percutaneous needle aponeurotomy (needling) involves mechanical separation of the pathologic cords using a fine needle, allowing digital extension without incision. Surgical options include fasciectomy, which entails partial or complete removal of the fibrotic tissue. While complete fasciectomy is associated with lower long-term recurrence compared with partial fasciectomy, this benefit is often outweighed by substantially greater acute morbidity and late complications.
Recurrence rates also differ based on treatment modality. When treated with needling, the 5-year recurrence rate of DD is reported to be 65%. Recurrence rate is 75% with collagenase injections and 10% to 75% for treatment with fasciectomy. More invasive surgery is thought to increase the risk of acute and late toxicity. 11 Furthermore, disease reactivation can occur following invasive interventions, such as collagenase injection, fasciotomy, and fasciectomy, contributing to the challenge of durable disease control.
In European countries such as Germany, radiation therapy (RT) is considered a preferred treatment option for patients with earlier stages of DD not meeting the criteria for surgical interventions. This approach has been attracting attention from the United States through the use of social media. 12 Recent emerging studies, including DEPART, a randomized controlled trial, have begun to evaluate the potential benefit of RT in the management of early stage DD. 13 However, the majority of available evidence assessing outcomes in patients receiving RT for DD consists of retrospective analyses and single-institution prospective studies. Controversy exists regarding the field design and standard of approach for these studies, including the treatment area, dose, and fractionation recommended for the course of treatment. Although various RT techniques have been described, split-course RT is a commonly reported strategy; however, considerable variation in dose and fractionation exists across studies. This systematic review evaluated the use of RT in DD, in an effort to provide an updated evidence base for this technique in DD treatment.
Methods
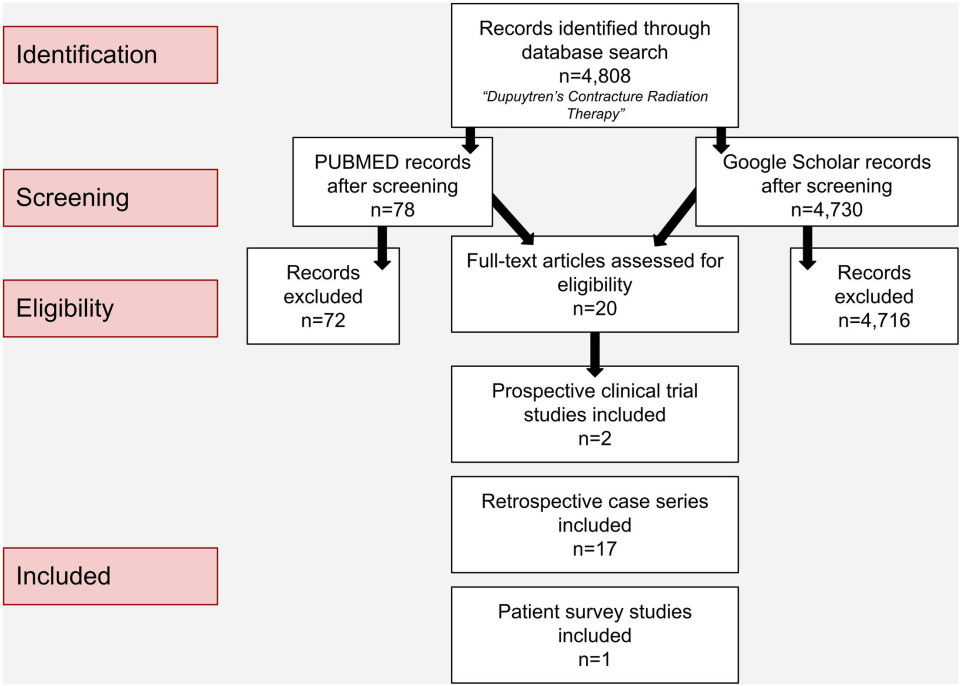
Using a method of screening based on Participants, Intervention, Comparison, Outcomes, and Study design, 4808 articles were initially considered for eligibility of analysis via search engines PubMed and Google Scholar throughout September 6, 2025, to November 6, 2025. This was performed using the free-text phrase Dupuytren’s Contracture Radiation Therapy, entered as a single search string without conjunctions. Additional records were then identified through backward citation chaining from key studies and reviews, independently screened, and assessed against this review’s prespecified criteria.
Eligibility Criteria and Study Selection
Studies were included if they evaluated the use of RT for treating DD in human subjects. Inclusion criteria consisted of clear physician or patient-reported treatment outcomes, including disease progression and deterioration, adverse effects, and the need for salvage procedures. Randomized and nonrandomized controlled clinical trials, prospective and retrospective case series, case studies, and conference abstracts were eligible for inclusion. No restrictions were applied to the language and publication year of studies. Conversely, exclusion criteria consisted of studies focusing solely on surgical interventions (such as fasciectomy) without any use of RT, as well as studies with inadequate specificity regarding RT usage. All identified records (n = 4808) underwent title and abstract screening, which eliminated 4788 records due to either irrelevance or lack of specificity on RT techniques. Ultimately, 20 studies met all criteria for inclusion (Figure 1).
Risk-of-Bias Assessment
Approximately 85% of the qualifying studies were retrospective with limited follow-up (FU) and limited description of FU assessment, increasing susceptibility to selection and recall bias and therefore limiting the ability to draw causal inferences. Furthermore, inconsistent and unspecified FU timing across studies limited comparability and increased the chance of selective reporting bias, specifically toward any studies with one inferred FU event. Both of the prospective studies were conducted by the same research group in Germany with a focus on the effectiveness of RT for early stage DD. The large number of retrospective case studies included introduces a higher risk of bias, given the less structured methodologies inherent to retrospective study designs. Adverse events such as erythema or dryness were well reported in most studies; however, minimal discussion was given in relation to long-term risks. Reliance on only 2 primary search engines could also have led to the omission of studies not indexed in these databases.
Results
A total of 4808 records were identified through the utilization of research database screenings. Of those, 20 studies met the inclusion criteria and were included in the final analysis: 2 prospective clinical studies, 14,15 17 retrospective cohort/case series studies, 16-32 and 1 cross-sectional patient-reported survey study. 33 Among the 17 retrospective studies, 2 were written by the same author. 21,22 Among all included studies, 6 were in German 16,24,26,29-31 and 1 in Italian. 20 When needed, an image-capturing feature of Google Translate (Google LLC, Mountain View, CA) was used to extract the necessary data from photocopied versions of the documents. Data were compiled with all included studies and respective reported outcomes (Table 1).
| Study | Total Cohort (Hands) | Article Type | Disease Stage Class | Radiation Therapy Regimen | Dose | Adjunct Therapies | Follow-Up | Number of Follow-Up Appointments | Definition of Observed Progression/Method of outcomes | Regressed Outcomes | Stable Outcomes | Progressed Outcomes | Salvage Procedures | Side Effects |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prospective clinical studies | ||||||||||||||
| Seegenschmiedt et al 14 | 198 (95 at 30 Gy; 103 at 21 Gy) | Randomized clinical study (preliminary results) | Tubiana et al stage ≤ II | 5 × 3 Gy over 2 series 8 wk apart vs 7 × 3 Gy in one series | 30 Gy (95 hands), 30 Gy (95 hands) | History of treatment with local excision/partial fasciectomy (25), steroid injection (6), NSAIDs (13), vitamin E (25), other drugs (15), unspecified (12) | >1 y in all patients | Assessed at 3 and 12 mo | Compared photocopied imaging before and after treatment. Progression defined by patient subjective response and physician evaluation of nodules and cord. | Total: 54.5%, 30 Gy: 55.8%, 21 Gy: 53.4% | Total: 37.4%, 30 Gy: 36.8%, 21 Gy: 37.9% | Total: 8.1% 30 Gy: 7.4% 21 Gy: 8.7% | Surgery: 4 | Redness/dryness sites n: 76 (38%), extensive erythema in sites n: 12 (6%), dry desquamation n: 10, moist desquamation n: 3, pronounced swelling n: 3, chronic side effects n: 26 at 3 mo. FU, n: 9 (5%) at 12 mo. FU, alteration of heat and pain sensation n: 8. |
| Seegenschmiedt et al 15 | 863 (293 at 21 Gy; 404 at 30 Gy); 166 control | Nonrandomized control trial | (1) 21 Gy/30 Gy: N: 195/199 N/I: 50/53 I: 43/47 II-IV: 5/4 | 5 × 3 Gy over 2 series 10-12 wk apart; or 7 × 3 Gy in one series; or none at all (control) | 30 Gy vs 21 Gy | History of treatment with local excision/partial fasciectomy (65), steroid injection (36), NSAIDs (28), vitamin E (45), other drugs (14), unspecified (16) | Median 104 mo, range 61-163 mo | The clinical evaluation (treatment side effect and efficacy) was performed at 3 and 12 moand at last FU after RT | Compared photocopied imaging before and after treatment. Progression defined by patient subjective response and physician evaluation of nodules and cord. | Not reported | Not reported | 21 Gy/30 Gy/control N: 7/3.5/34 N/I: 42/30/67 I: 58/49/87.5 II-IV: 80/75/100 Total: 22/16/52 | Surgery 21 Gy: 12% 30 Gy: 8% Control: 30% Salvage RT 21 Gy: 5.5% 30 Gy: 6% Control: 20% | Redness/dryness cases n:151 (25%) extensive erythema/moist desquamation/pronounced local swelling cases n:16 (2%), chronic side effects at last FU in 83 sites (14%). |
| Retrospective case series | ||||||||||||||
| Finney 21 | 25 | Case series | (2) Stage 1: 3 Stage 2: 4 Stage 3: 18 | 3000 r Ra-Moulage | 3000 r | None | 2-10 y | Not directly stated | Subjective improvement of softening nodules. Objective increase in movement was used. No cases were reported of worsening contracture. | Overall: 75% Stage 1: 100% Stage 2: 75% Stage 3: 72% | Overall: 25% Stage 1: 0% Stage 2: 25% Stage 3: 28% | None | Unknown | Skin dryness, occasional slight erythema. |
| Finney 22 | 43: 25 gamma radiation (presented in 1953 article); 18 × RT | Case series | (2) In X-RT group: Stage 1: 3 Stage 2: 0 Stage 3: 13 Stage 4: 2 | 1-3 × 1000 r Ra-Moulage | 1500-3000 r | None | >18 mo | Not directly stated | Regression was defined as a lessening of the feeling of tightness in the palm and fingers. | X-RT group only: 77.8% Stage 1: 100% Stage 2: n/a Stage 3: 76.9% Stage 4: 50.0% | X-RT group only: 22.2% Stage 1: 0 Stage 2: n/a Stage 3: 23.1% Stage 4: 50.0% | None | Unknown | Skin dryness, immediate paraesthesia. |
| Wasserburger 30 | 146 patients | Case series | (*) I: 69 II: 46 III: 31 | 1-3 × 1.000 r , Ra-Moulage | 1.000-3.000 rad | None | 2-19 y | Not directly stated | The success is defined as restorative full extension and full abilities of the fingers with no more detectable fixed skin areas. | Overall: 69.8% I: 89.8% II: 56.5% III: 32.2% | Unknown | Unknown | Surgical intervention: 8 | Not reported. |
| Corsi 20 | 13 | Case series | (3) All in the early stages of the disease | 600-800 r of penetrating RT cycle then 2200-2500 r of plesiotherapy | Variable | Vitamin E | 6 mo to 3 y | Not directly stated Multiple FUs advised for some patients | Subjective increased elasticity, smoother skin, and decreased nodules. Improved extension of the 3-4 metacarpals was also assessed as a measurement of improvement. | 77% | 23% | 0 | Surgical referral: 1 | Cutaneous criteria reactions in some, also epidermolysis in some instances. The epidermolysis quickly resolved soon after. |
| Lukacs et al 28 | 36 | Case series | (*) Stage 1: 32 Stage 2: 4 | (4 Gy twice a week) repeated every 2 mo | 24-40 Gy | None | ≤5 y | Not directly stated | Regression was defined as a definite softening of nodular lesions and improvement of contractures of the palms. | Stage 1: 81% , Stage 2: 75% , Total: 81% | Stage 1: 19% Stage 2: 25% Total: 19% | None | Surgical referral: 3 | Not reported. |
| Vogt/Hoschau 29 | 109 patients | Case series | (3)I: 98 II: 4 III/IV: 7 | (2 × 4 Gy) × 4 every 2 mo | 32 Gy | None | >3 y | Not directly stated | No disease progression is considered a success. Initial symptoms of pain, redness, nodule formation, increased thickness, and stretching limitation were evaluated for inclusion. | 1:21% Il: 25% III/IV: - Total: 20% | 1:74% Il: 50% III/V: 86% Total: 74% | 1: 4% Il: 25% III/IV: 14% Total: 6% | Unknown | Not reported. |
| Koehler 26 | 33 | Case series | (3) All stage I | 10 × 2 Gy 3-5 times a week (2.0 single dose daily or 3 times up to total dose of about 20 Gy) | 20 Gy | None | 1-3 y | Not directly stated | Progression was defined as an increase in hardening/contractures. Improvements were subjective visual results and patient reports. | Total: 21% | Total: 61% | Total: 18% | Surgical intervention: 3 Salvage RT: 3 | Not reported. |
| Herbst/Regler 24 | 33 patients (46 hands) | Case series | (4) N: 16 I: 30 II: 3 III: 1 IV: 1 (mainly treated early stages from some patients being excluded due to extended contracture and developed recurrence noted) | 3-14 × 3 Gy (< 42 Gy) | 9-42 Gy TD | None | >1.5 y | Not directly stated | Imaging evaluation of nodules and cords. | 39/46 (85%) | 6/46 (13%) findings/symptoms remained unchanged without demonstrable progression up until time of result evaluation | (1/46) 2% | Surgery (one stage IV case due to therapy not being followed) | RT dermatitis n: 13 patients (39%). Some cases observed acute dermatitis with skin redness/subcutaneous emphysema. |
| Weinzierl et al 31 | 34 patients | Case series | (3) All stage I | 5 × 3 Gy, 6 wk apart | 15-33 Gy | None | Mean 7 y | Not directly stated | Iselin grading, decreased extension, thickening and hardening of nodules/cords. | 9% | 41% | 50% | Surgery: 4 | Skin dryness/occasional scaling of volar skin n: 32% patients. |
| Keilholz et al 25 | 142 (57 with >5-year FU) | Case series | (1) Total/5 year N: 82/28 N/I: 17/10 I: 30/17 II: 12/1 III: 1/1 | 5 × 3 Gy over 2 series 6 wk apart | 30 Gy | None | Median 6 y, range 1-12 y | Not directly stated | In addition to patient-reported outcomes, functional changes and flexion deformity of fingers were measured using a protractor. The size of nodules and cords was directly measured and their consistency palpated. | 3 mo: 7% N: 2% N/I: 29% I: 10% II/III: 0% 5 year: 11% | 3 mo: 92% N: 98% N/I: 71% I: 83% II/III: 100% 5 year: 67% | 3 mo: 1% N: 0% N/I: 0% I: 6.7% II/III: 0% 5 year: 23% | Surgery: 6 None in 5-yr f/u group | Acute mild skin reactions/erythema/dry desquamation with burning and itching n: 61 (43%), radiodermatitis/edema n: 14 cases (10%). Other complaints were dry skin, mild skin atrophy, slight fibrosis, and occasional telangiectasia. |
| Adamietz et al 16 | 176 | Case series | (1) 0: 5 N: 76 N/I: 15 I: 65 II: 12 III: 3 | 5 × 3 Gy over 2 series 6-8 wk apart | 30 Gy | None | Median 10 y, range 7-18 y | Not directly stated | Tubiana stage, size, and appearance of nodules and cords. | N: 15% I: 6% II: 0% III: 0% Total: 10% | N: 66% I: 29% II: 17% III: 0% Total: 49% | (in + out of field) N: 19% I: 65% II: 83% III: 100% Total: 41% | Surgery: 4 Salvage RT: 10 | Anhidrosis/strong scaling n: 44 hands (25%), skin atrophy n: 15 palmar areas (8.5%). |
| Betz et al 18 | 208 | Case series | (1) N: 115 N/I: 33 I: 50 II: 7 III: 2 IV: 1 | 5 × 3 Gy over 2 series 6-8 wk apart | 30 Gy | Prior treatments with recurrence leading to RTX referral: Surgery (8), steroid injection (1) | Median 13 y, range 2-25 y | Not directly stated | The size and number of nodules or cords were directly measured and their consistency palpated. | N: 6% N/I: 30% I: 6% II: 0% III: 0% IV: 0% Total: 10% | N: 81% N/I: 40% I: 32% II: 14% III: 0% IV: 0% Total: 59% | (in + out of field) N: 13% N/I: 30% I: 62% II: 86% III: 100% IV: 100% Total: 31% | Surgery (39 patients, 42 cases) | Minor long-term radiogenic skin/subcutaneous changes n: 66 cases (32%), dry skin/desquamation n: 47 cases (23%), erythema n: 5 (2%), skin atrophy n: 14 cases (7%). |
| Grenfell/Borg 23 | 4 | Case series | (1) N: 2, I: 2 | 5 × 3 Gy over 2 series 6 wk apart | 30 Gy | None | Median 35 mo, range 34-42 mo | Not directly stated | Objective reduction in size of the target lesion(s) or subjective improvement in pain or discomfort. | 100% | Not reported | Not reported | Not reported | Mild local erythema and edema, minimal fatigue. |
| Zirbs et al 32 | 297 | Retrospective patient survey case series | (*) Symptom duration: < 20 mo: 56 > 20 mo: 61 | 2 × 4 Gy over 4 wk, with 8 wk apart between treatments | 32 Gy | Prior treatment with surgery (18), needle fasciotomy (8), local steroid (3), oral vitamins (1), homeopathic cream and massage (1), shock-wave therapy (1), magnetic field therapy (1), NSAIDs (1), hand gymnastics (1), other injections (1) | Median 40 mo, range 6-115 mo | Not directly stated | Patients measured with a visual analog scale. | 45% by patient report, 21.6% of nodules regressed (92/426 nodules) | 35% by patient report | - | Not reported | Dryness n: 82 patients (39.8%), erythema n: 42 patients (20.4%), desquamation n: 8 patients (3.8%), chronic side effects (more than 4 weeks) n: 71 (34%). |
| Banks et al 17 | 10 patients, 8 hands irradiated (11 feet also irradiated) | Case series | (*) Not reported | 5-6 × 3 Gy over 2 series 3 mo apart | 30-36 Gy | None | Mean 12.1 mo, range 0.9-30.5 mo | Not directly stated | Reduction in pain or deformity of lesions. | 71% (but not distinguished from plantar lesions) | 24% (but not distinguished from plantar lesions) | 5% (but not distinguished from plantar lesions) | Not during study period | Not reported. |
| Latusek et al 27 | 117 patients (number of hands unknown) | Case series | (*) “In the early stages of DD” 61.5 % had contractures | 7 × 3 Gy in one series | 21 Gy | Prior treatment with laser therapy (1%), surgery (13%), ultrasound (1%), local steroid injection (1%) | Mean 4.8 (SD ±6.11) mo, range 1-34 mo | Not directly stated | Improvement defined as a decrease in the size of nodules, reduction of contracture, or the improvement of manual function. | 57.50% | 35% | 7.50% | Not mentioned | No skin problems n: 87.5% subjects, palmar dryness n: 2.5%, erythema n: 7.5%, superficial epidermal exfoliation n: 2.5%. |
| Ciernik et al 19 | 5 | Case series | (1) I: 2 III: 1 IV: 2 | Anhidrosis n: 44 hands (25%), skin atrophy n: 15 palmar areas (8.5%) | 20 Gy | Palmar aponeurectomy | Mean 14.1 mo, range 6-30 mo | Annual FUs | Joint extension 6 mo after RT | 100% | 0 | 0 | Not during study period | Not reported. |
| Patient survey studies | ||||||||||||||
| Schuster et al 33 | 51 sites in the hand (15 sites in the foot) | Retrospective patient survey case series | (1) N or N/I | 7 × 3 Gy in one series or 5 × 3 Gy over 2 series 6-8 wk apart | 21 Gy or 30 Gy | Prior treatment with surgery (4), steroid injections (2) | Median 31 mo, range 1-61 mo | Not directly stated | Progression was defined as new or worsening clinical disease (eg, nodule growth, cord development, contracture) or worsening symptoms (pain, itching/burning, or pressure) as described by patients. | Symptomatic improvement 70-81% pain/itching/burning, 95% pressure, 64% mobility, overall 93% some symptomatic benefit | - | In-field: 30% In-field after re-irRT: 21% | Surgery: 2 Collagenase: 1 Needle aponeurotomy: 2 Re-irRT: 4 Massage: 1 | Acute toxicity n: 13 patients (39%), dryness/erythema/dry desquamation/edema/tenderness n: 89 sites, late toxicities n: 10 patients (30%). |
| Future prospective trials | ||||||||||||||
| DEPART Trial, Burgess et al (2018-not specified) 34 | 60 patients | Prospective randomized clinical trials |
2 Arms:
Arm1: < 10° contracture Arm2: (*) Not reported | 10 × 3 Gy over 2 series 4-12 wk apart | 30 Gy | Patients with planned LF, PNF, or CI of flexion contracture(s) were eligible for the adjuvant component | Minimum 5 y | Independently reviewed at 6 mo, 12 mo, annually up to 60 mo and then biannually up to 104 mo | QuickDASH, URAM, and a VAS for pain. CTCAE v4.03 toxicity recording. | Improved 36 mo metrics: QuickDASH: 17.83 Obs vs 10.93 LDRT (P = .013) URAM: 4.8 Obs vs 3.1 LDRT (P = .056) VAS: 2.1 Obs vs 1.0 LDRT (P = .001) Long-term data not yet published | Not yet published | Not yet published | Not yet published. | Acute toxicity n: 39 patients (65%), dermatitis RT n: 14 patients (23%), reduced sweating n: 12 patients (20%), pruritus/skin pain n: 4 (6.7%) Reduced sweating, although mild, tended to persist. |
| University of Minnesota Trial (2019-2026) 35 | 50 patients | Prospective observational pilot study | (*) Not reported | 5 daily treatments of 300 cGy × 10 for a total of 3000 cGy over 2 series 6-8 wk apart | 30 Gy | Limited fasciectomy, PNA, and CCH injection | 2 y | 6 wk after completion of all RT and 1 and 2 y after | Using the 5-item Southampton Dupuytren’s Scoring System questionnaire. Scores range from 0 (no problem) to 4 (severe problem), with total scores ranging from a sum of 0 to 20. High scores indicate greater impairment due to the disease. | Not yet published | Not yet published | Not yet published | Not yet published. | Not yet published. |
Treatment Protocols
Each of the 20 studies varied in their RT technique, dose, approach with adjuvant therapy, FU time/frequency, duration, and outcomes. Disease progression and regression were highly inconsistent. Many reports did not specify the criteria used to classify clinical disease improvement or progression, with several studies omitting quantitative measures entirely. A wide variety of disease stages were included; however, a majority of patients were characterized as early stage DD, typically regarded as stages 0 to 1. A majority (68%) of patients among all studies were listed to either be in the early stages of DD to the study’s discretion or less than or equal to stage 1 in the respective classification spectra. Several disease classification spectra were utilized (Table 2), including that of Tubiana et al (1966) with modifications, 14-16 18,19,23,25,33 as well as Shaw (1951), 21,22 Iselin (1967), 20,26,29,31 and Millesi (1980s). 24 There were 5 studies with unknown staging systems. 17,27,28,30,32 Several different RT modalities were used, including electrons, superficial orthovoltage or radium moulage, external beam RT, and brachytherapy. Regardless of the disease stage, most studies used doses commonly up to 30 Gy or 3000 r, and different fractionation schedules ranged from 9 to 42 Gy in total dose across studies. No dosimetric analysis was reported in the retrospective studies to confirm dose distribution. The FU period ranged from 0.9 months to 19 years. Due to the varying medians and reporting format of FU periods within the studies, a pooled mean was difficult to obtain. However, a majority of studies (60%) explicitly reported waiting at least 1 year post treatment for initial FU.
| Classification Systems | ||||
|---|---|---|---|---|
| (1) Tubiana (1966) with modifications | (2) Shaw (1951) | (3) Iselin (1967) | (4) Millesi (1980s) | |
| Grade/stage | N: Nodules, cords, or skin retraction in palmar fascia without finger contracture | 1a: Nodule in palmar fascia but not including the skin without finger contracture 1b: Nodule in the skin but not involving the palmar fascia and no finger contracture | 0: Small subcutaneous nodules without contracture | Grade 0: No contracture; normal extension |
| *N/I: Nodules, cords, or skin retraction with extension deficit of 1˚ to 5˚ | 2: Nodule in fascia involving skin without finger contracture | 1: Nodules or cords in palm or up to proximal phalanx with MCPJ contracture | Grade I: MCP or PIP joint contracture ≤45° | |
| I: Nodules, cords, or skin retraction with extension deficit of 6˚ to 45˚ | 3: Nodule in fascia involving skin with finger contracture | 2: Nodules or cords extend up to the middle phalanx with MCPJ and PIPJ contracture | Grade II: Contracture > 45° but ≤90° | |
| II: Nodules, cords, or skin retraction with extension deficit of 46-90˚ | 4: All 3 prior stages with secondary changes to tendons/joints | 3: Nodules or cords extend to the distal phalanx and contracture involving all 3 phalanges | Grade III: Contracture > 90° | |
| III: Nodules, cords, or skin retraction with extension deficit of 91-135˚ | 4: Severe contracture of the PIPJ resulting in hyperextension at the DIPJ (boutonniere deformity) | Grade IV: Severe deformity and contracture of multiple fingers, functional impairment | ||
| IV: Nodules, cords, or skin retraction with extension deficit > 135˚ | ||||
Outcomes of Radiation
When evaluating the outcomes of all qualifying studies, 13 out of the 20 (65%) reported the regression of patient symptoms as the most frequent outcome. The percentage of patients with this outcome ranged from 45% to 100%. with a mean of 74.5%. 14,17,19-24 27,28,30,32 The Schuster et al study was omitted from this mean calculation due to patient outcome being subjective, with unclear FU duration in survey modality. Regardless, the results obtained post treatment from the Schuster patient survey showed 70% to 81% symptomatic improvement. 33 The Seegenschmiedt et al 2012 study did not report total regression outcomes, but focused instead on the disease progression for patients undergoing RT. In the results, the control group reported clinical evidence of progression in 52% of patients, compared with 22% in the cohort of patients receiving 21 Gy, and 16% in the cohort of patients receiving 30 Gy. There was no statistically significant difference in disease progression based on the dose of RT used (21 Gy vs 30 Gy). However, disease progression increased among untreated control patients compared with irradiated patients (P < .001). 15 5 studies reported a lack of disease progression in radiated patients as the most common outcome. 16,18,25,26,29 Only one study found significant progression of disease after RT following an initial FU examination 7 years after RT. 31
The criteria for establishing disease regression, stability, or progression varied from study to study. Most studies characterized improvement as some form of relief from symptoms, such as the decrease of nodules or cords, reduction of pain or deformity, increased joint extension, and smoothness of the skin. Additionally, the percentage of irradiated patients reported to need subsequent surgical intervention post-therapy ranged from 2% to 20%. 14-16,18,24-26 30,31,33 A need for surgical referral was reported in 2 of the 20 studies, 20,28 and 8 were not reported during the study period. 17,19,21-23 27,29,32
Adverse Events
When analyzing adverse effects and toxicities from RT, common outcomes included skin redness, erythema, dryness, desquamation, and atrophy. 14,15,18,21,23-25 27,31-33 The ongoing DEPART trial has provided new prospective data on the toxicity and persistent effects of RT. A total of 404 hands—202 randomized to observation, and 202 to low-dose RT (LDRT)—were followed for a median period of 36 months. On FU, 162 hands experienced at least one toxicity, with the vast majority (96.6%) noted as grade 1. No grade 3 events were observed. Furthermore, of the 14 grade 2 toxicities, only one (reduced sweating) persisted up to 24 months following treatment. 13 Quality-of-life outcomes using the Quick Disabilities of Arm, Shoulder, and Hand measure demonstrated improvement in the LDRT arm (10.9 vs 17.8 P = .013). Future results will provide a more comprehensive assessment of longer-term disease control and toxicity.
Discussion
In this systematic review, we analyzed 20 studies consisting of prospective, retrospective, and cross-sectional survey studies evaluating the effects of using RT for DD. Although different staging systems were used depending on the study’s design, a majority (68%) of treated patients were reported to either be in the early stages of DD (to the study’s discretion) or ≤ stage 1 in the respective classification system. Overall, 65% of patients in the 20 studies reported the regression of symptoms as the most common outcome. It is hypothesized there is a therapeutic window for RT in patients with early stage DD, as early stage patients have decreased progression (Tubiana stage N/I progression after RT was 30% compared with 75% in stage 2-4). 14 As RT preferentially affects rapidly proliferating tissues, RT would be more efficacious in the early disease state of high cellularity and mitosis than in later-stage disease characterized by low cellularity and high collagen concentration. Among the 13 studies deemed successful, 2 reported a 100% regression outcome. 19,23 Both studies comparatively share a smaller cohort size of ≤5 irradiated hands, but Ciernik’s 2021 study included a wider array of disease stages in its patient demographics (Table 1). 19 Despite the overwhelming unanimity of results, the study’s small cohort sizes limit the ability to draw widespread conclusions.
Only one study reported disease progression as the most common outcome following RT. In the retrospective analysis by Weinzierl et al, nearly all irradiated hands demonstrated progression as their primary outcome during long-term FU, which the authors attributed to the approximate 7-year period in which patients were not monitored between completion of RT and the first FU assessment. Importantly, the observed pattern and rate of progression did not differ meaningfully from the natural history of early DD, suggesting that RT in this cohort did not alter long-term disease behavior. 31
It is important to recognize that not all patients with early stage DD experience progression when left untreated. A meaningful proportion can remain clinically stable for years. As the Seegenschmiedt cohort demonstrates, approximately 50% of untreated patients were reported to show no progression of symptoms during extended FU. 15 Given this variability, RT should not be considered for patients with stable and asymptomatic disease. Instead, it is best to make treatment decisions based on documented, active disease progression characterized by clinical criteria and worsening changes in hand grip strength, finger span or finger-raise measurements, or contracture deformity. 36 This will ensure that RT is only offered when biologically and clinically justified.
Chronic side effects were less commonly reported with a calculated average of 22.8% in patients, falling between an encompassing range of 13% to 30% experiencing persistent changes after treatment. Common long-term effects consisted of symptoms such as altered heat sensation, pain, or other acute effects. The timing for FU evaluation of chronic symptoms varied or was not explicitly stated. In the UK, the National Institute for Health and Care Excellence cautions there is limited evidence for the safety of RT in patients with early DD, with a discussion of theoretical risk on the development of RT-induced cancer in the long-term effects. 37 However, no cases of long-term cancer caused from RT of DD have been reported.
A high risk of bias presents an additional challenge when interpreting the results. 8 In the 2 prospective clinical studies conducted by the same author, methods and outcomes were reported comprehensively, in contrast to the majority of retrospective studies, which frequently lacked methodological detail and specificity. 14,15 Patient-reported outcome measures were often subjective and dependent on individual patient perception, rendering them more susceptible to response bias. 33 Furthermore, substantial heterogeneity was observed across the included studies with respect to patient demographics, treatment parameters, adjunctive therapies, study endpoints, and FU duration. Consequently, drawing precise conclusions from the currently available published data remains challenging.
Several prospective studies on RT in DD are underway. The ongoing DEPART randomized clinical trial will provide prospective randomized data on RT efficacy both as definitive treatment and as adjuvant treatment after needle aponeurotomy. It will also explore the adverse effects of DD following RT as described previously. 34 Furthermore, the University of Minnesota’s ongoing Post-contracture Release RT for DD study has been evaluating efficacy for postoperative RT through an observational prospective pilot study format (NCT04122313). 35 Similarly, the Dartmouth-Hitchcock Medical Center has an ongoing study evaluating RT for Dupuytren’s Contracture Following Non-Surgical Release (NCT06330545). 38
Taken together, the available evidence across studies and existing trials suggests that RT may be most beneficial in earlier, proliferative stages of DD, when nodules are active, but contractures are minimal (< 10 degrees). RT should be recommended when there is documented active progression in patients, ideally captured using standardized clinical criteria (changes in hand grip strength, finger span or finger-raise measurements, or contracture deformity). If RT is used, current data support moderate-dose regimens (30 Gy in 10) fractions in a split-course modality (6-12 weeks break between 5 fraction courses) or 21 Gy in 7 fractions without break. A split-course approach is thought to best balance both risk and therapeutic effects. Patients who are unable to receive the split course regimen could be considered for the 21 Gy in a 7 fractions regimen, although there is a slightly increased risk of contracture progression. 15 Treatment planning should include fields delineated based on the fascial thickening seen on CT planning and palpation of nodules/cords plus margins. 36
Using CT-based planning techniques, prospective studies and practice guidelines recommend the application of bolus and coverage of the involved palmar fascia with the >90% isodose line. 13,36 However, the evidence base remains limited by unspecified outcome measures and inconsistent reporting of progression, regression, and long-term control. To improve the field, future prospective, standardized trials are needed, using uniform definitions of disease activity, consistent functional outcome measures, and detailed long-term FU to determine whether RT alters the natural history of early DD. The development of core outcome sets and universal consensus based on clinically meaningful endpoints will be essential for future progress.
Conclusion
In summary, the use of early stage RT as a noninvasive method to treat DD holds promising potential as an alternative to invasive surgery. However, debates over the variability of study design, FU duration, and outcome reports limit the strength of existing evidence in current literature. To better assess long-term efficacy for prophylactic therapy and treatment, future studies should adopt a standardized protocol, consisting of larger and controlled patient populations, to produce consistent and high-quality results. The future of these results will be essential to guide the next steps in clinical decision-making.
References
- Lanting R, Broekstra D, Werker P, van den Heuvel E. A systematic review and meta-analysis on the prevalence of Dupuytren disease in the general population of western countries. Plast Reconstr Surg. 2014;133(3):593-603. doi:10.1097/01.prs.0000438455.37604.0f.
- Mansur H, Oliveira E, Gonçalves C. Epidemiological analysis of patients with Dupuytren’s disease. Rev Bras Ortop. 2018;53(1):10-14. doi:10.1016/j.rboe.2017.12.003.
- Salari N, Heydari M, Hassanabadi M. The worldwide prevalence of the Dupuytren disease: a comprehensive systematic review and meta-analysis. J Orthop Surg Res. 2020;15(1):495. doi:10.1186/s13018-020-01999-7.
- Verjee L, Verhoekx J, Chan J. Unraveling the signaling pathways promoting fibrosis in Dupuytren’s disease reveals TNF as a therapeutic target. Proc Natl Acad Sci USA. 2013;110(10). doi:10.1073/pnas.1301100110.
- Eckerdal D, Nivestam A, Dahlin L. Surgical treatment of Dupuytren’s disease – outcome and health economy in relation to smoking and diabetes. BMC Musculoskelet Disord. 2014;15:1176. doi:10.1186/1471-2474-15-1176.
- Michou L, Lermusiaux J, Teyssedou J. Genetics of Dupuytren’s disease. Joint Bone Spine. 2012;79(1):7-12. doi:10.1016/j.jbspin.2011.05.027.
- Riesmeijer S, Kamali Z, Ng M. A genome-wide association meta-analysis implicates hedgehog and notch signaling in Dupuytren’s disease. Nat Commun. 2024;15(1):199. doi:10.1038/s41467-023-44451-0.
- Ball C, Izadi D, Verjee L, Chan J, Nanchahal J. Systematic review of non-surgical treatments for early Dupuytren’s disease. BMC Musculoskelet Disord. 2016;17(1). doi:10.1186/s12891-016-1200-y.
- Balaguer T, David S, Ihrai T. Histological staging and Dupuytren’s disease recurrence or extension after surgical treatment: a retrospective study of 124 patients. J Hand Surg Eur Vol. 2009;34(4):493-496. doi:10.1177/1753193409103729.
- Trojian T, Chu S. Dupuytren’s disease: diagnosis and treatment. Am Fam Physician. 2007;76(1):86-89.
- Ruettermann M, Hermann R, Khatib-Chahidi K, Werker P. Dupuytren’s disease-etiology and treatment. Dtsch Arztebl Int. 2021;118(46):781-788. doi:10.3238/arztebl.m2021.0325.
- Dupuytren’s online forum.
- Martin J, Burgess T, McClelland B. Toxicities and quality of life following observation or radiation therapy for Dupuytren’s disease: three-year secondary analysis data from the prevention group of the international dupuytren’s evaluation of preventative or adjuvant radiation therapy (DEPART) randomized trial. Int J Radiat Oncol Biol Phys. 2026;124(3):604-613. doi:10.1016/j.ijrobp.2025.09.037.
- Seegenschmiedt M, Olschewski T, Guntrum F. Radiotherapy optimization in early-stage Dupuytren’s contracture: first results of a randomized clinical study. Int J Radiat Oncol Biol Phys. 2001;49(3):785-798. doi:10.1016/S0360-3016(00)00745-8.
- Seegenschmiedt M, Keilholz L, Wielpütz M. In: Dupuytren’s disease and related hyperproliferative disorders. 2012:349-371.
- Adamietz B, Keilholz L, Grünert J, Sauer R. Radiotherapy of early stage dupuytren disease. Long-term results after a median follow-up period of 10 years. Strahlenther Onkol. 2001;177(11):604-610. doi:10.1007/pl00002371.
- Banks J, Wolfson A, Subhawong T. T2 signal intensity as an imaging biomarker for patients with superficial fibromatoses of the hands (Dupuytren’s disease) and feet (Ledderhose disease) undergoing definitive electron beam irradiation. Skeletal Radiol. 2018;47(2):243-251. doi:10.1007/s00256-017-2792-5.
- Betz N, Ott O, Adamietz B. Radiotherapy in early-stage Dupuytren’s contracture. Long-term results after 13 years. Strahlenther Onkol. 2010;186(2):82-90. doi:10.1007/s00066-010-2063-z.
- Ciernik I, Goldschmidt P, Wösle M, Winter J. Feasibility of aponeurectomy in combination with perioperativeir high dose rate brachytherapy for Dupuytren’s disease 192. Strahlenther Onkol. 2021;197(10):903-908. doi:10.1007/s00066-021-01801-5.
- Corsi M. Radiotherapy of Dupuytren’s disease. Friuli Med. 1966;21(3):391-403.
- Finney R. Dupuytren's contracture a radiotherapeutic approach. The Lancet. 1953;262(6795):1064-1066. doi:10.1016/S0140-6736(53)90669-2.
- Finney R. Dupuytren’s contracture. Br J Radiol. 1955;28(335):610-614. doi:10.1259/0007-1285-28-335-610.
- Grenfell S, Borg M. Radiotherapy in fascial fibromatosis: a case series, literature review and considerations for treatment of early-stage disease. J Med Imaging Radiat Oncol. 2014;58(5):641-647. doi:10.1111/1754-9485.12178.
- Herbst M, Regler G. Dupuytren’s contracture. Radiotherapy in the early stages. Strahlentherapie. 1985;161(3):143-147.
- Keilholz L, Seegenschmiedt M, Sauer R. Radiotherapy for prevention of disease progression in early-stage Dupuytren’s contracture: initial and long-term results. Int J Radiat Oncol Biol Phys. 1996;36(4):891-897. doi:10.1016/s0360-3016(96)00421-x.
- Koehler A. Radiotherapy in Dupuytren’s contracture. Radiobiol Radiother. 1984;25:6.
- Latusek T, Miszczyk L, Gierlach G, Zając P. An effectiveness evaluation of the palmar fascia irradiation of patients suffering from Dupuytren’s disease. Nowotw J Oncol. 2017;67(3):162-167. doi:10.5603/NJO.2017.0027.
- Lukacs S, Braun-Falco O, Goldschmidt H. Radiotherapy of benign dermatoses: indications, practice, and results. J Dermatol Surg Oncol. 1978;4(8):620-625. doi:10.1111/j.1524-4725.1978.tb00512.x.
- Vogt H, Hochschau L. Behandlung der dupuytrenschen kontraktur. MMW Münch Med Wochenschr. 1980;122:125-130.
- Wasserburger K. Therapy of Dupuytren’s contracture. Strahlentherapie. 1956;100(4):546-560.
- Weinzierl G, Flügel M, Geldmacher J. Lack of effectiveness of alternative non-surgical treatment procedures of Dupuytren contracture. Chir Z Alle Geb Oper Medizen. 1993;64:492-494.
- Zirbs M, Anzeneder T, Bruckbauer H. Radiotherapy with soft x-rays in dupuytren’s disease - successful, well-tolerated and satisfying. J Eur Acad Dermatol Venereol. 2015;29(5):904-911. doi:10.1111/jdv.12711.
- Schuster J, Saraiya S, Tennyson N. Patient-reported outcomes after electron radiation treatment for early-stage palmar and plantar fibromatosis. Pract Radiat Oncol. 2015;5(6). doi:10.1016/j.prro.2015.06.010.
- Burgess T, Wegener E, McClelland B. Adverse events after adjuvant radiation therapy for dupuytren’s disease in the DEPART randomized trial. J Hand Surg Eur Vol. 2025;50(7):891-897. doi:10.1177/17531934251321393.
- Efficacy of post-contracture release radiation for the treatment of Dupuytren’s disease. 2025.
- Yunes M. Radiation therapy for Dupuytren contracture. Prac Radiat Oncol. 2025;15(3):248-252. doi:10.1016/j.prro.2025.01.005.
- Radiation therapy for early Dupuytren’s disease: overview.
- Warhold L. Radiation therapy for Dupuytren’s contracture following non-surgical release. 2024.
Disclosures
The authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere. The patient has provided informed consent for the publication of this case report.
Citation
. Radiation Therapy for Dupuytren Disease: A Systematic Review of Clinical Outcomes and Adverse Effects. Applied Radiation Oncology. 2026. doi:10.37549/ARO-D-25-0053.