Low dose rate brachytherapy for prostate cancer at the Cleveland Clinic: A technical review
Images

Prostate cancer is the most common cancer in American men. The American Cancer Society estimates about 238,590 new cases of prostate cancer will be diagnosed in 2013. According to the most recent data, including all stages of prostate cancer, the relative 5-year survival rate is over 99%.1 In 1995, only 2% of new cases were treated with brachytherapy; today that number is about 30%.
Brachytherapy (from the Greek word brachys, which means near) is the use of ionizing radiation material that is placed inside of or near to the tumor. The interstitial brachytherapy of the prostate began in 1917 using radium needles guided by a finger in the rectum.2 All prostate brachytherapy is currently performed under transrectal ultrasound (TRUS) guidance using techniques pioneered by Holm in Denmark and advanced by a group of investigators based in Seattle.3-7
In this procedure, 125I, 103Pd, or 131Cs brachytherapy sources are implanted through needles placed through the perineum and guided by a template under TRUS guidance. Prostate cancer is graded according to the tumor-node-metastasis (TNM) staging system along with the prostate specific antigen (PSA) and Gleason sum score (GSS). Early stage prostate cancer is classified into 3 risk groups by the National Comprehensive Cancer Network (NCCN).8
- Low-risk: T-stage ≤2b, GSS ≤6, PSA ≤10.
- Intermediate-risk: T-stage ≤2b and a) GSS=7 or b) PSA >10 but ≤20.
- High-risk: T-stage = 2c or GSS ≥8 or PSA >20. Brachytherapy can be used alone to treat low-risk disease and may be used as a boost for external beam radiotherapy (EBRT) to treat high-risk disease. For intermediate risk prostate cancer, the choice is subject to the study by Radiotherapy Oncology Group (RTOG 0232, 2003).9 As opposed to the external beam radiation, prostate brachytherapy is performed in a single treatment, and it can reduce the complications to the surrounding tissues, such as the rectum.
Patient selection
Our practice at the Cleveland Clinic offers several choices to the prostate cancer patient. These include observation (watchful waiting),surgery, and radiation therapy (external beam or permanent seed implant). The implant program was started in 1996, and the number of patients treated now exceeds 4000. The vast majority of these were as monotherapy. Patients typically arrive in the early morning and are discharged in the afternoon from the ambulatory surgical center where the implants are performed. More than 90% of the implants were done on low or intermediate risk patients.
Isotopes
Our choice of isotope is 125I primarily because its relatively long half-life permits banking and storage of seeds for several weeks. Seeds are ordered weekly with reference strength of 0.499 U. Approximately one-quarter of the seeds ordered are loose; the rest are linked instrands with a 1-cm spacing. We have over the years ordered sources from several vendors. Currently we use the Source Tech Medical model STM1251 125I source (STM 1251) obtained from Bard Medical. STM-1251 consists of a cylindrical gold rod (0.180 mm diameter)inside of a 3.81 mm long aluminum wire with a diameter of 0.510 mm. The overall source length is 4.50 mm and the active length is 3.80mm. Dosimetric data for STM1251 are published in AAPM TG-43 update.10 For our practice, the seeds arrive sterilized with a 10% third party assay. The 125I seeds are loaded by special procedure therapists into needles using a loading block and a pair of tweezers at the time of implantation.
Ultrasound volume study
The patient is induced into general anesthesia and placed in the dorsal lithotomy position so that pubic arch interference is minimized. The TRUS probe provides both transverse and longitudinal images. It is mounted in a probe carrier along with a manually operated stepping device (5-mm step size). Attached to the carrier is a template with holes to accommodate 18-gauge needles arranged in a 13 by 13 matrix at 5-mm spacing. The grid pattern can be displayed on the monitor screen of the US unit. Transverse images starting 1-cm superior to the prostate base and proceeding through the gland to 1-cm past the apex are acquired using a video capture device attached to the planning computer. Geiger-Mueller (GM) detector and ionization chamber are available to check the room and patients for lost seeds and to measure the exposure at the skin surface and 1 m away.
Treatment planning
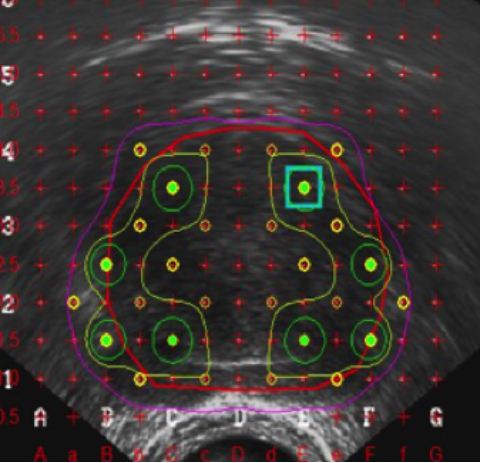
VariSeed 8.0.2 (Varian Medical Systems, Charlottesville, VA) is the treatment planning system currently used. AAPM TG43 dose calculation formalism is implemented in the VariSeed treatment planning system to calculate the dose. The source is modeled as a point source with an anisotropy factor varying with distance. At the onset of our implant program, patients were planned in advance and sources ordered specifically for that patient. However, we soon moved to operating room (OR) based planning, which had particular advantages for both patient and procedure efficiency.11 Once the axial images obtained at 5-mm intervals are registered in the planning system, bladder, rectum,and CTV (prostate) are contoured. A Foley catheter is used by some physicians to visualize the urethra. The planning strategy is a modified peripheral loading with a dose margin of 3 to 5 mm (Figure 1). Partial coverage of the seminal vesicles is usually part of the plan. Loose seeds are implanted near the interface between the prostate and the rectum to keep the rectal dose below the prescription dose. The centralregion of the CTV is kept to <120% of the prescription dose (144 Gy). A typical plan will have a V100 of the CTV of nearly 100%, a D90 of the CTV between 100 and 120%, a V150 of the CTV of approximately 50%, and a V200 of the CTV of 15% or less. Seeds in strands are used for the periphery, while the central region is loaded with loose seeds permitting easy retrieval from the urethra or bladder if necessary.
Needle loading and implantation
Once the plan is ready and reviewed by the authorized user physician, a worksheet that includes the number and distributions of needles and seeds is printed and used by the therapists to load the seeds into the needles in the RO under sterile conditions. The loaded needles are placed in a sterile box having a lid that corresponds to the template (Figure 2). This box is fully loaded before any seeds are implanted in the patient. The implantation is performed by the radiation oncologist under ultrasound guidance starting with the most anterior row of the template.
Quality assurance
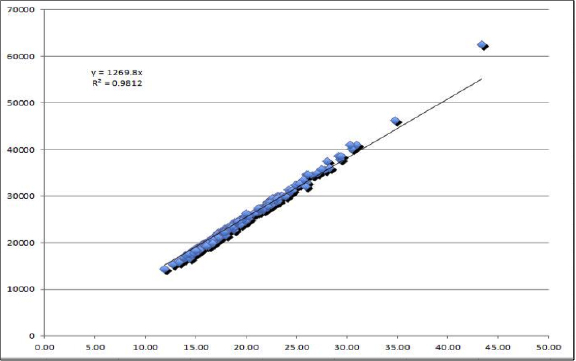
Pursuant to the regulations for medical use of radioactive materials (Ohio is an agreement state), a third party assay of 10% of the seeds is provided. The specific patient plan is checked by the physicist using a home-grown nomogram (Figure 3). Radioactive material inventory control is maintained by seed counts at various stages of each procedure: seeds brought from storage and seeds placed on the loading table before and after the needles have been loaded. Any discrepancy is resolved before the procedure is allowed to continue. A GM detector is kept in the OR to locate misplaced seeds and to survey each needle after its seeds have been implanted. Also available is a sensitive exposure meter for room and patient surveys to conduct radiation survey following the procedure. The radiation survey includes the floor, waste, and all used applicators. This meter is used to measure the exposure rate at the surface and at 1 m from the implanted patient for documentation.The patient survey is reviewed by the medical physicist to confirm that the prostate seed implant satisfies all pertinent federal and state regulations regarding the release of patients with radioactive sources, NCRP commentary No. 11. Patients are transported to a recovery area and then usually released a few hours after the procedure with a set of instructions for home use and a card that is to be carried for several months indicating the nature of the implant (isotope and activity). A post-implant radiograph is taken in the recovery area permitting assessment of the implant.
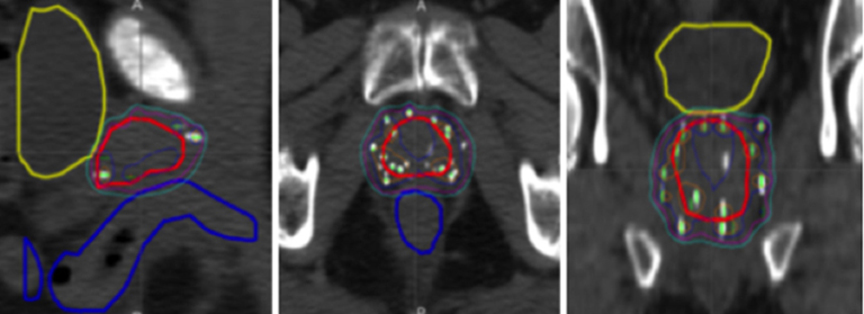
Patients are requested to return to the clinic for a post-implant CT scan typically one month after the implantation. A quantitative dose analysis is carried out using dose-volume histograms (DVH) for the prostate, rectum, and bladder. The post-implantation dosimetry provides information about the implant quality and aims to improve the team techniques (Figure 4). Comparison of V100 and D90values between the pre-plan and the post-plan is done to ensure that differences between the pre-plan and post-plan are <20%.
Conclusion
The procedures used in our clinic for prostate seed implants have proven to be efficient (up to 6 patients in one day). These procedures likely work best with large practices where a seed bank can be maintained.
References
- ACS Cancer Facts and Figures 2013. American Cancer Society. http://www.cancer.org/research/cancerfactsfigures/cancerfactsfigures/cancer-facts-figures-2013. Accessed February 20, 2014.
- Barringer BS. Radium in the treatment of carcinoma of the bladder and prostate. JAMA. 1917; 68:1227–1230.
- Whitmore WF. Interstitial radiation therapy for carcinoma of the prostate. Prostate. 1980;1: 157–168.
- Holm HH, Juul N, Pedersen JF, et al. Transperineal 125iodine seed implantation in prostatic cancer guided by transrectal ultrasonography. J Urol. 1983;130:283–286.
- Blasko JC, Grimm PD, Ragde H. Brachytherapy and organ preservation in the management of carcinoma of the prostate. Semin Radiat Oncol. 1993;3:240-249.
- Johansson JE, Holmberg L, Johansson S, et al. Fifteen-year survival in prostate cancer. A prospective, population-based study in Sweden. JAMA. 1997;277:467–471.
- Ragde H, Blasko JC, Grimm PD, et al. Interstitial iodine-125 radiation without adjuvant therapy in the treatment of clinically localized prostate carcinoma. Cancer. 1997; 80:442-453.
- NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer V.1.2007. National Comprehensive Cancer Network. http://www.nccn.org/professionals/physician_gls/PDF/ prostate.pdf V (2007).
- Nath R, Bice W, Butler WM, et al. AAPM recommendations on dose prescription and reporting methods for permanent interstitial Brachytherapy for Prostate Cancer. AAPM report No. 137. Med Phys. 2009;36(11):5310-5322.
- Rivard MJ, Butler WM, DeWerd LA, et al. Supplement to the 2004 update of the AAPM Task Group No. 43 Report. Med. Phys. 2007;34:2187-2205.
- Wilkinson DA, Lee EJ, Ciezki JP, Mohan DS, et al. Dosimetric comparison of pre-planned and or-planned prostate seed brachytherapy. Int J Radiat Oncol Biol Phys. 2000;48(4):1241-1244.