Surface mold brachytherapy: A means to achieve therapeutic skin irradiation in a case of synchronous bilateral breast cancer wit
Images

Case summary
A 38-year-old female, gravida 2 para 2, presented with a 8 to 10-month history of progressive firmness and nipple retraction of her right breast with multiple areas of dimpling and skin redness bilaterally that started as she was breast-feeding. Upon examination, patchy skin erythema with multiple nodular lesions scattered over both the breasts were noted. On palpation, the right breast was nearly completely firm and immobile. No appreciable masses were palpated in the left breast. Bilateral palpable axillary and supraclavicular lymphadenopathy was noted.
Imaging findings
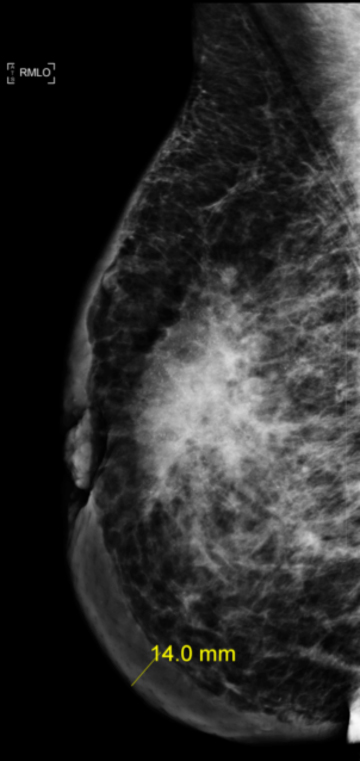
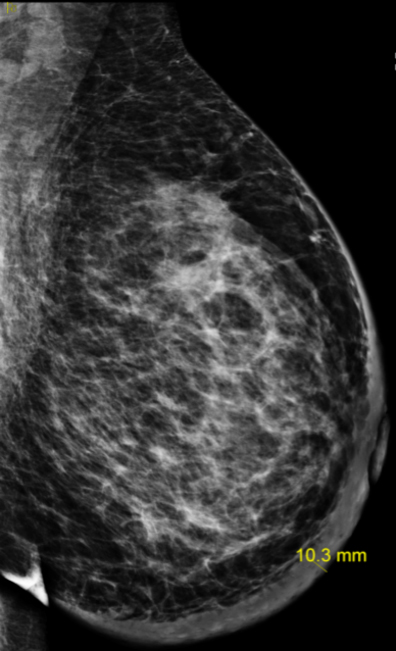
Diagnostic mammogram findings are as shown in Figure 1. An ultrasound-guided biopsy of the breast mass as well as bilateral axillary lymph nodes confirmed malignancy. Systemic staging work-up with laboratory evaluations, bone-scan and a computed tomography (CT) scan showed no evidence of distant metastatic disease. The final diagnosis was stage IIIC (cT4b cN3a cM0), bilateral breast invasive ductal carcinoma, grade-2, estrogen/progesterone receptor positive, HER-2/neu amplified.
Differential diagnosis
Malignancy, chronic mastitis, abscess, eczema, hidradenitis suppurativa, idiopathic granulomatous mastitis
Discussion
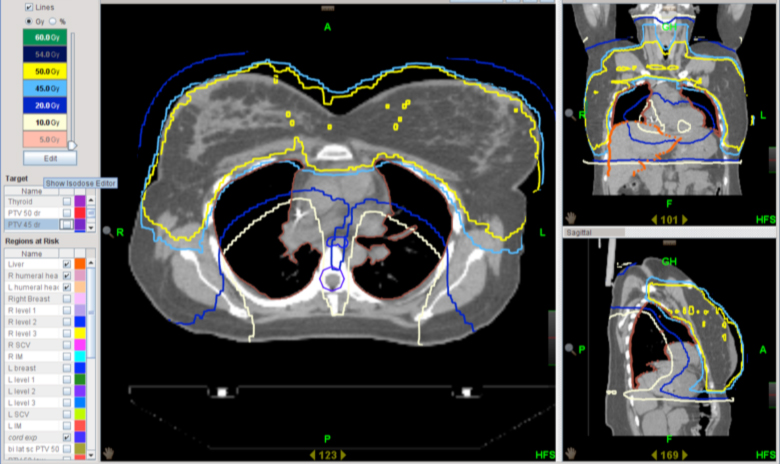
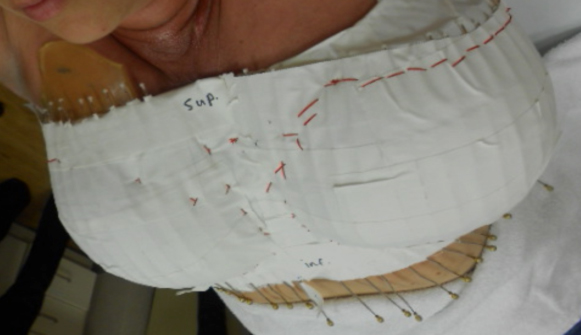
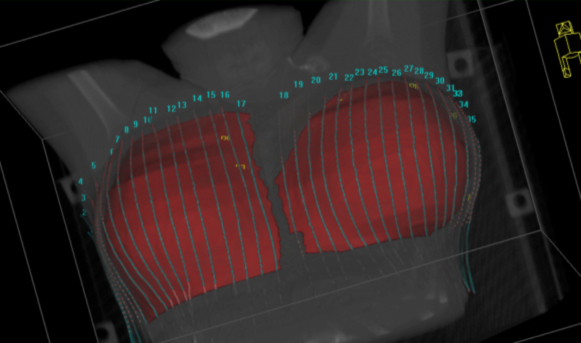
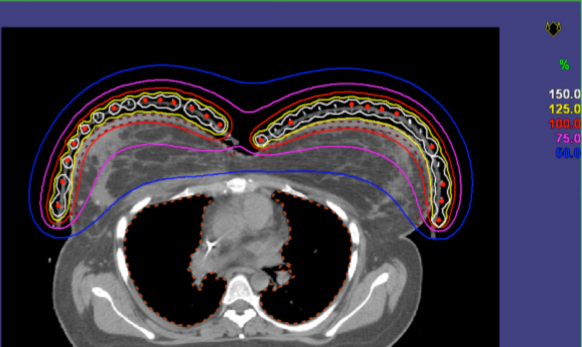
In order to facilitate bilateral mastectomy, neoadjuvant chemotherapy with dose-dense adriamycin-cyclophosphamide followed by paclitaxel-herceptin was completed. A partial response, especially over the cutaneous lesions, still precluded bilateral mastectomy.Hence, radiotherapy (RT) targeting bilateral breasts (including the skin) and the axillary, internal mammary, and supraclavicular nodes was planned. To minimize irradiation of the underlying lungs and heart, intensity-modulated radiotherapy (IMRT) technique was employed for the initial phase treatment (Figure 2). Bolus was used, and thermoluminescent dosimeter measurements were obtained to confirm appropriate skin dose (1.8 to 2 Gy per fraction). Limited by radiation tolerance doses for the lungs and heart, the final boost to the skin was planned using surface mold-based, Iridum-192 high-dose-rate (HDR) brachytherapy (Figures 3 and 4). This was treated alongside 3-dimensional (3D) conformal fields for bilateral supraclavicular nodes. Brisk acute grade-2 radiation dermatitis with nonconfluent moist desquamation was seen at one-week post-RT with good resolution by one-month. Successful bilateral mastectomy was subsequently performed with the pathological assessment revealing only minute foci (< 1%) of residual carcinoma in bilateral breasts, and one of 3 right axillary lymph nodes and zero of 7 left axillary lymph nodes were positive for residual carcinoma. Significant treatment effects with areas of mucin and fibrous changes were noted in the breast and nodal tissue. Ten months post-RT and surgery, the patient has developed lymphedema of the right upper extremity, but no symptomatic radiation pneumonitis or other late radiation toxicities.
Radiotherapy plays a critical role in management of breast cancer. CT scan-based 3D-conformal radiotherapy (3D-CRT) planning techniques are utilized to limit dose to normal underlying lung and heart (in left-sided malignancies).1 The challenge of RT planning is increased even further in the setting of bilateral breast cancer (BBC), especially if regional lymphatics need to be irradiated.2 In an old series reported fromMassachusetts General Hospital in 1981, 3 out of 15 long-term survivors with synchronous or metachronous BBC developed medial subcutaneous fibrosis/necrosis after having received comprehensive bilateral breast and lymphatic irradiation planned in the conventional 2-dimensional(2D)-era.3 The authors described various techniques for match-line management in that era. Investigators from the University of Pennsylvania reported their initial experience in 55 patients with BBC who received whole-breast RT (WBRT) after breast conserving surgery (BCS) using conventionally planned tangential portals.4 One-quarter of the patients had a midline field overlap of up to 4 cm. No patient developed match-line fibrosis with 4% patients developing pneumonitis. Yamauchi et al described 17 patients with synchronous or metachronous BBC who were treated with BCS followed by WBRT (50 Gy in 25 fractions) using tangential portals with midline matched fields designed using CT-based planning.5 Treatments were well tolerated, with only one patient developing moderate midline subcutaneous fibrosis. No cases of significant pneumonitis were reported. Sharma et al described electron arc therapy to treat bilateral chest walls in the postmastectomy setting.6 This technique provides adequate skin doses, reduces the risk of field overlap in the midline from conventional tangential fields, and also effectively reduces dose to underlying lung and heart. However, treating regional lymphatics can still require matched photon fields. In addition, the authors noted that meticulous treatment planning with strict quality assurance is required for electron dosimetry.
More recently, IMRT-based radiation planning and delivery techniques are used to additionally reduce normal tissue irradiation.7 In the setting of BBC, IMRT offers the advantage of treating a complex target of bilateral breasts/chest walls and locoregional lymphatics, while eliminating the challenges associated with field matching and minimizing the dose to the organs at risk. Nicolini et al described a dosimetric comparison between fixed-field IMRT and volumetric-modulated arc therapy (VMAT) for treating bilateral breasts and showed several dosimetric improvements with the VMAT technique.8 Lee et al attempted to enhance IMRT planning even further by comparing dose-volume-based IMRT planning versus generalized equivalent uniform dose-based optimization planning and demonstrated superiority in dosimetric outcomes by pursuing the latter approach.2
In our case, for the initial phase of RT (50 Gy in 25 fractions) we utilized Tomotherapy-based IMRT technique. Nonetheless, considering the large volume of tissue requiring irradiation, the volume of bilateral lungs receiving 30 Gy (V30 Gy) was 18%, while V20 Gy and V5 Gy was 28% and 65% respectively, and the mean bilateral lung dose was 14.7 Gy. The heart V25 Gy, V20 Gy, V10 Gy and mean doses were 23%, 35%, 75%, and 18.5 Gy, respectively. To maintain a risk for symptomatic pneumonitis of ≤ 20%, Quantitative Analysis of Normal Tissue Effects in the Clinic (QUANTEC) guidelines currently recommend limiting the bilateral lung dose to V20 Gy ≤ 30%,with mean dose ≤ 20 Gy.9 Cardiac dose is limited to mean < 26 Gy and V30 Gy < 46% to reduce the risk for pericarditis, while V25 Gy should be < 10% to keep the risk for long-term cardiac mortality < 1%.
An IMRT boost plan to adequately treat bilateral breast skin resulted in intolerably high pulmonary and cardiac doses. Instead, a customized surface brachytherapy (BRT) mold was generated to allow delivery of an additional 10 Gy in 5 fractions, prescribed to a depth of7 mm beneath the skin surface of the breasts. Achieving irradiation of skin to therapeutic doses was especially critical since lack of dermal response was the primary reason for unresectability. Surface brachytherapy is not used very commonly in management of breast cancer. Older experiences have been described from the University of Heidelberg using reusable pulsed-dose-rate (PDR) skin mold brachytherapy in patients with cutaneous involvement from primary or recurrent breast cancer.10,11 Stewart et al described the utility of high-dose-rate (HDR) surface applicator-based BRT to deliver scar boost radiation in 2 patients who underwent immediate breast reconstruction postmastectomy, a scenario that is increasingly seen in the current era.12 When comparing with electron-fields, superior homogeneity, and coverage of the scar was noted.
Conclusion
Radiotherapy planning for bilateral breast cancer with cutaneous involvement can be a practical challenge. In our patient, initial comprehensive IMRT followed by skin boost delivered using surface-mold BRT safely provided therapeutic radiation doses to the skin, while minimizing the dose heterogeneity and other challenges associated with matching multiple radiation fields. This can be considered as an option for the management of this rare and complex clinical scenario.
References
- Beavis AW. Treatment planning challenges in breast irradiation: The ideal and the practical. Clin Oncol (R Coll Radiol). 2006;18:200-209.
- Lee TF, Ting HM, Chao PJ, et al. Dosimetric advantages of generalised equivalent uniform dose-based optimisation on dose-volume objectives in intensity-modulated radiotherapy planning for bilateral breast cancer. Br J Radiol. 2012;85:1499-1506.
- Kopelson G, Munzenrider JE, Doppke K, Wang CC. Bilateral breast cancer: radiation therapy results and technical considerations. Int J Radiat Oncol Biol Phys. 1981;7:335-341.
- Fung MC, Schultz DJ, Solin LJ. Early-stage bilateral breast cancer treated with breast-conserving surgery and definitive irradiation: The University of Pennsylvania experience. Int J Radiat Oncol Biol Phys. 1997;38:959-967.
- Yamauchi C, Mitsumori M, Nagata Y, et al. Bilateral breast-conserving therapy for bilateral breast cancer: Results and consideration of radiation technique. Breast Cancer. 2005;12:135-139.
- Sharma PK, Jamema SV, Kaushik K, et al. Electron arc therapy for bilateral chest wall irradiation: Treatment planning and dosimetric study. Clin Oncol (R Coll Radiol). 2011;23:216-222.
- Arthur DW, Morris MM, Vicini FA. Breast cancer: New radiation treatment options. Oncology (Williston Park). 2004;18:1621-1629; discussion 1629-1630, 1636-1638.
- Nicolini G, Clivio A, Fogliata A, Vanetti E, Cozzi L. Simultaneous integrated boost radiotherapy for bilateral breast: A treatment planning and dosimetric comparison for volumetric modulated arc and fixed field intensity modulated therapy. Radiat Oncol. 2009;4:27.
- Marks LB, Yorke ED, Jackson A, et al. Use of normal tissue complication probability models in the clinic. Int J Radiat Oncol Biol Phys. 2010;76:S10-19.
- Fritz P, Hensley FW, Berns C, Schraube P, Wannenmacher M. First experiences with superfractionated skin irradiations using large afterloading molds. Int J Radiat Oncol Biol Phys. 1996;36:147-157.
- Harms W, Krempien R, Hensley FW, Berns C, Wannenmacher M, Fritz P. Results of chest wall reirradiation using pulsed-dose-rate (PDR) brachytherapy molds for breast cancerlocal recurrences. Int J Radiat Oncol Biol Phys. 2001;49:205-210.
- Stewart AJ, O’Farrell DA, Bellon JR, et al. CT computer-optimized high-dose-rate brachytherapy with surface applicator technique for scar boost radiation after breast reconstruction surgery. Brachytherapy. 2005;4:224-229.