Diffuse chest wall calcifications after post mastectomy radiotherapy for breast cancer
Images
CASE SUMMARY
A 56-year-old female with stage IIIA ER+/PR+/Her2- infiltrating ductal carcinoma of the right breast underwent total mastectomy and axillary lymph node dissection, adjuvant AC-T, radiotherapy (50 Gy in 25 fractions to the chest wall and supraclavicular nodes with a 10 Gy in 5 fraction electron scar boost), and hormonal therapy. Thirteen years later, she presented with a 4-month history of an enlarging right chest wall ulcer with multiple firm subcutaneous nodules around the scar. She denied recent trauma, although was found to have a subclavian arterial thrombosis, managed by thrombectomy and stent placement and resulting in partial regression of the ulcer. A chest computed tomography (CT) scan was completed following her clinic visit. A conservative excisional biopsy was negative for malignancy. She thus underwent a wide resection including the lesion, skin and underlying rib followed by reconstruction, confirming the diagnosis.
IMAGING FINDINGS
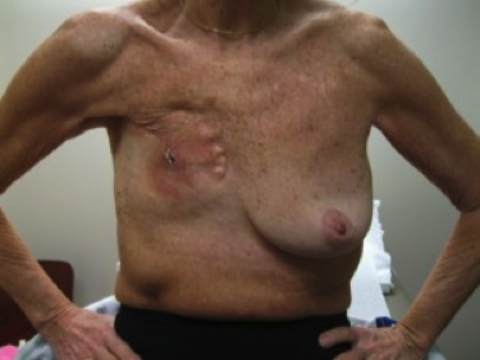
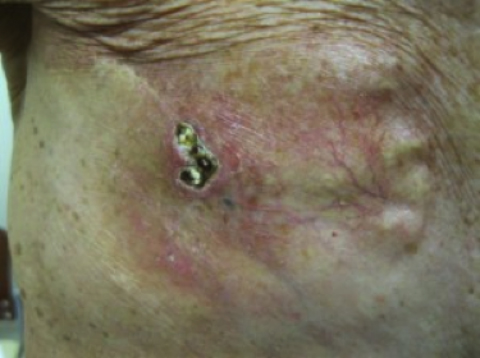
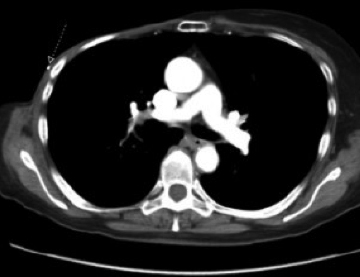
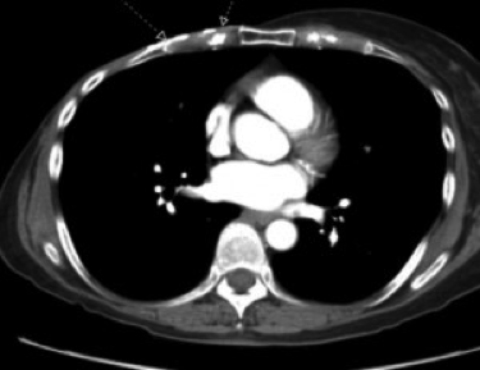
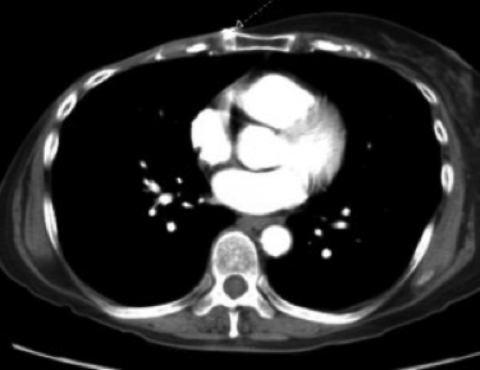
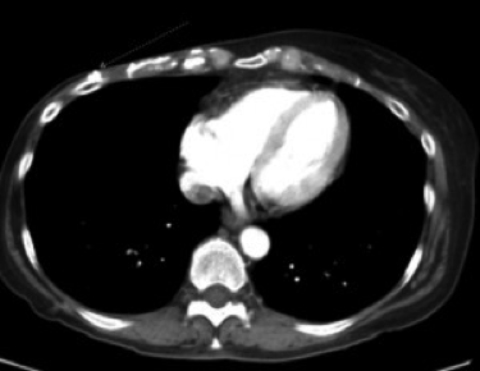
Physical examination revealed right chest wall skin atrophy with scattered telangectasias and multiple small subcutaneous nodules near a scabbed lesion along the right chest wall scar (Figure 1). Chest CT revealed multiple hyperdense, discrete subcentimeter nodules in the chest wall subcutaneous tissue (Figure 2). No lymphadenopathy or metastatic disease was seen.
DISCUSSION
Pathologic evaluation of resected tissue revealed dense fibrosis and foci of necrosis, granulation tissue and calcifications without any malignancy. Thus, the final diagnosis was chest wall ulceration and diffuse subcutaneous calcifications as a result of late-radiation related changes. In all, increasing awareness has been brought forth regarding delayed radiation effects. Although such occurrences are now less common after breast and chest wall radiotherapy due to higher energy photons and greater dose homogeneity, late radiation changes still occur in this population where expected cancer-related survival is prolonged. Identifying radiation-related changes can be challenging but, as seen in this case, the clinical presentation may prove most helpful. Late skin complications of radiotherapy include pigmentation changes, skin atrophy, fibrosis, telangiectasia, necrosis and ulceration. Such changes often progress slowly and manifest over the span of months to years, with more rapid development in cases of an associated vascular or traumatic event.
Interestingly, the patient presented here developed a subclavian artery thrombosis with no prior coronary or peripheral artery disease. Studies have implicated breast or chest wall radiotherapy to such thrombotic events, including within the arteries of the heart.1 Partial improvement of her ulceration after re-vascularization of the subclavian artery may implicate her thrombosis as the inciting event for such late radiation changes.
In general, calcification is often regarded as a component of the healing process, generated as a physiologic defense designed to contain, wall off and stabilize injured or damaged tissue. This process of inflammation causing coronary artery calcification has been well-documented.2 The pathogenesis of radiation-induced dystrophic calcification is obscure, but it may be caused by persistent inflammation, leading to phosphate binding to denatured proteins, or from a dysregulation of intracellular Ca2+ concentration of mitochondria in dying cells.3 In addition, mineralizing micro-angiopathy is also considered to play a role in the development of calcifications. This calcification often develops in irradiated tissue secondary to the leakage of plasma fluid from damaged vessels, and regional ischemia resulting from impairment in the microcirculation secondary to the mineralizing micro-angiopathy.4
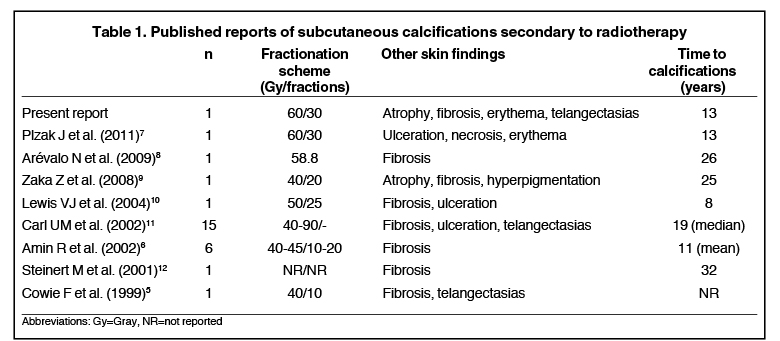
The presence of subcutaneous calcifications has been shown previously in case reports to be a sign of delayed radiation changes. Based on a literature review, a total of 28 cases of subcutaneous calcifications has been reported following radiotherapy for oncologic conditions, all exceeding doses of 40 Gy (Table 1).5-12 The first reported case by Cowie et al. demonstrated calcifications along the match line between the chest wall and supraclavicular fields, implying a relationship to dose hot spots.5 These findings were also seen in a case series by Amin et al., again implying formation along the match line.6 Nonetheless, in all the reported series, additional skin sequelae were seen and, thus, emphasize the importance of long-term follow-up and documentation of clinical findings.
CONCLUSION
This case, along with a small body of literature, demonstrates the finding of subcutaneous calcifications as a delayed toxicity from radiotherapy. Determining whether chest wall or breast skin changes are a result of radiotherapy or cancer recurrence remains challenging. Presence of multiple skin findings presenting years following radiotherapy, particularly in light of an inciting traumatic or vascular event, may help guide management. Aggressive biopsy or surgical intervention should be pursued cautiously if post-radiotherapy toxicity is suspected, as wound-healing can be problematic.
REFERENCES
- Marks LB, Yu X, Prosnitz RG, et al. The incidence and functional consequences of RT-associated cardiac perfusion defects. Int J Radiat Oncol Biol Phys. Sep 1 2005;63(1):214-223.
- Li JJ, Zhu CG, Yu B, et al. The role of inflammation in coronary artery calcification. Ageing Res Rev. Dec 2007;6(4):263-270.
- Enoch S, Kupitz S, Miller DR, Harding KG. Dystrophic calcification as a cause for non healing leg ulcers. Int Wound J. Jun 2005;2(2):142-147.
- Suzuki S, Nishio S, Takata K, et al. Radiation-induced brain calcification: paradoxical high signal intensity in T1-weighted MR images. Acta Neurochir (Wien). 2000;142(7):801-804.
- Cowie F, Jones R. Subcutaneous calcification as a late effect of orthovoltage chest wall irradiation. Clin Oncol (R Coll Radiol). 1999;11(3):196-197.
- Amin R, Hamilton-Wood C, Silver D. Subcutaneous calcification following chest wall and breast irradiation: a late complication. Br J Radiol. Mar 2002;75(891):279-282.
- Plzak J, Kalitova P, Urbanova M, Betka J. Subcutaneous calcification in the pectoralis major flap: a late complication of radiotherapy. Br J Radiol. Nov 2011;84(1007):e221-223.
- Arévalo N, Cabeza B, Ferreirós J, Bustos A. Heterotopic calcification of the chest wall: An unusual complication of radiation therapy. Euro J of Radiol Extra. 2009;70(2):e53-e55.
- Zaka Z, Fodor J, Udvarhelyi N, et al. Subcutaneous calcification as a delayed complication of radiotherapy: a case report and review of the literature. Pathol Oncol Res. Dec 2008;14(4):485-488.
- Lewis VJ, Holt PJ. Subcutaneous calcification following high-dose radiotherapy. Br J Dermatol. May 2004;150(5):1049-1050.
- Carl UM, Hartmann KA. Heterotopic calcification as a late radiation effect: report of 15 cases. Br J Radiol. May 2002;75(893):460-463.
- Steinert M, Gottlober P, Gall H, Peter RU. [Subcutaneous calcifications after radiotherapy]. Hautarzt. Jun 2001;52(6):518-521.