Eradicating Cancer Stem Cells Using High Linear Energy Transfer Radiation Therapy Part 1: Physics and Radiobiology
Applied Radiation Oncology — Vol. 14, Issue 2, pp. 1-8
DOI: 10.37549/ARO-D-25-00003P1
Published: June 1, 2025
1 Department of Radiation Oncology, Mayo Clinic Florida/Mayo Clinic Comprehensive Cancer Center, Jacksonville, FL
2 Department of Radiation Oncology, Mayo Clinic Rochester/Mayo Clinic Comprehensive Cancer Center, Rochester, MN
3 Department of Radiation Medicine, Roswell Park Comprehensive Cancer Center, Buffalo, NY
4 Department of Radiation Oncology, Charité – Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany
5 Charité – Universitätsmedizin Berlin, Berlin, Germany
6 German Cancer Consortium (DKTK) and German Cancer Research Center (DKFZ), Heidelberg, Germany
7 Department of Radiology and Precision Health Program, Michigan State University, Lansing, MI
8 Department of Radiation Oncology, Ohio State University Comprehensive Cancer Center, Columbus, OH
9 Department of Radiation Medicine, Northwell Cancer Institute, New Hyde Park, NY
10 Biophysics Department, GSI Helmholtzzentrum für Schwerionenforschung, Darmstadt, Germany
* Corresponding author: Daniel M. Koffler (koffler.daniel@mayo.edu)
Abstract
Keywords
LET, linear energy transfer, particle therapy, carbon ion therapy, heavy particle therapy, radiobiology, radiation physics, spatial fractionation, ion therapy
Introduction
Heavy particle therapy (HPT), including carbon ion radiation therapy (CIRT), exhibits high linear energy transfer (LET) and offers a distinct mode of energy deposition compared with conventional photon therapy as well as proton therapy. Instead of diffusing ionization events over wide spatial geometries, HPT delivers dense ionization along the beam pathway, producing clustered and complex damage to cancer-cell DNA that is exponentially more lethal to these directly targeted cells than low LET radiation. The direct effects of HPT on cancer-cell DNA are also responsible for enhanced immune activation and clinically validated abscopal responses, suggesting a greater role for HPT in augmenting systemic disease control than conventional radiation therapy.
In particular, HPT shows promise in the treatment of glioblastoma (GBM) and pancreatic ductal adenocarcinoma (PDAC), both of which are marked by recalcitrant cancer stem cell (CSC) biology that is substantially responsible for their resistance, recurrence profiles, and overall grim clinical outcomes. Preclinical data indicate that high LET radiation can overcome CSC resistance mechanisms in general, including in GBM and PDAC, by halting or reversing stemness-promoting processes such as epithelial-mesenchymal transition (EMT) that low-LET radiation tends to induce.
New approaches such as LET painting and spatial fractionation enable delivery of high LET radiation selectively to high-biological-risk tumor subvolumes. Such techniques are necessary for the clinical implementation of HPT that is both safe and effective at improving outcomes in resistant cancers, as the same augmented lethality that is welcome when delivered to cancer cells poses a greater danger to nearby healthy tissue than low LET exposure. Additionally, biologically selective LET deposition may enhance the immunogenic/abscopal properties of HPT.
Here, we review the current status and approaching frontiers of high LET radiation therapy with a focus on its application to the often underappreciated, pressing clinical problem of CSC biology, as well as clinical and logistical barriers to the expansion of HPT beyond the niches it currently occupies.
Basic Science of Heavy Particle Therapy and Suitability in Treatment-Resistant Cancers
Physics and Biology of Heavy Particles: Local Structure Defines Radiobiology and Trumps Dose
LET can be defined as average ionizing energy transferred to medium per length of radiation track, typically in keV/µm. It is a dynamic property of charged particles over the course of a beam path, increasing with mass and decreasing with kinetic energy.1-4 More specifically, LET is proportional to , where Zeff is effective charge and is velocity.5 The relationship of these properties with medium stopping power defines the distinct Bragg peak depth dose profile for all charged particles.1,2
Clinical Relevance of LET: Protons Vs Heavy Ions
By convention, “low LET” charged particles have locally restricted averaged LETΔ ≤ 10 keV/µm and “high LET” charged particles have LETΔ ≥ 20 keV/µm, respectively, in the Spread Out Bragg Peak (SOBP).6-8 The category of HPT applies to all charged particles satisfying this definition, which in practice means all charged particles that have greater mass than protons. The conventional exclusion of protons from HPT reflects the proportional relationship between LET and , that is, even the smallest possible increase in effective charge—from 1 (protons) to 2 (helium ions)—results in a 4-fold increase in LET. Carbon ions, the most common heavy charged particles in clinical use, have a +6 charge and an LET that is 36 times greater than that of protons.
Limitations of Proton Therapy in Resistant Disease
Low Zeff sustains a photon-like low LET close to ~0.5-1 keV/µm throughout the proton beam range, including most of the SOBP.7-13 Clinical proton irradiation, like photon irradiation, is sparsely ionizing and ill-suited to radioresistant targets. Rapid deceleration in the final few microns of the proton SOBP causes LET to rise as high as 80 keV/µm due to its inverse proportionality to However, in standard proton beam therapy (PBT), the need for good coverage of large, 3-D targets requires a broad SOBP, rendering the high LET distal edge clinically insignificant in terms of dose contribution (Figure 1).7,8,14
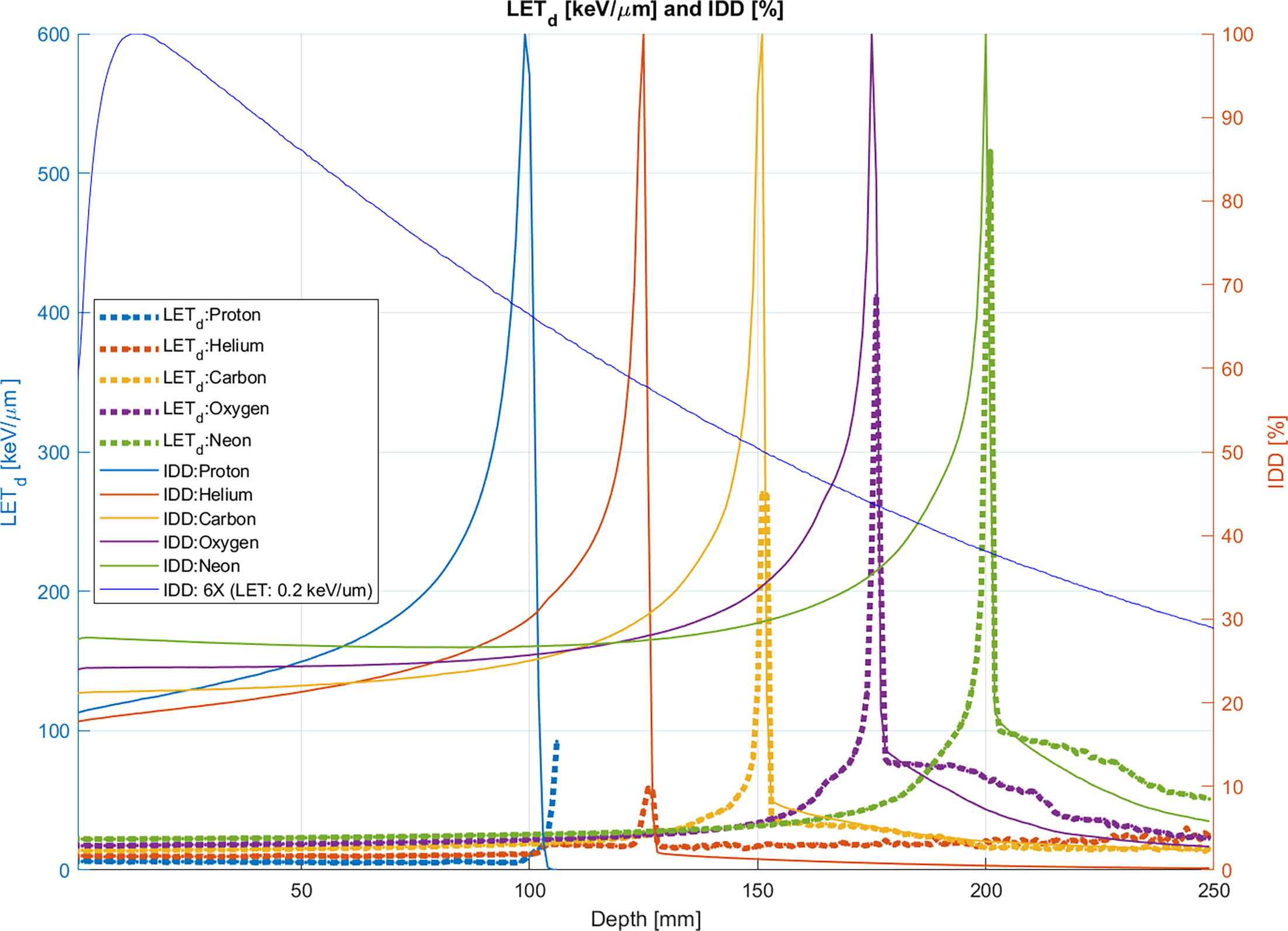
Despite efforts to improve PBT, the fundamental differences between proton beams and heavier ions render them physically and radiobiologically distinct therapeutic modalities.10,15-18 From a purely dosimetric perspective, HPT offers advantages over PBT, including more robust optimization, smaller spot sizes, reduced lateral and longitudinal scatter, and steeper distal fall-off, though this is tempered by the presence of fragmentation tails beginning with particles as massive as carbon ions.1,2,19 Along with its physical advantages, HPT may affect the clinical outcome of aggressive cancers such as PDAC and GBM, specifically with respect to the CSC biology driving them, due to its radiobiological properties.
Biological Effects of LET at the Molecular Level
Tracks of low LET radiation in biological matter are characterized by sparse, diffuse ionization events.1,14,20-22 Each Gray of photon and proton radiation at therapeutic acceleration energies induces approximately 30-40 lethal double-stranded breaks (DSBs) at wide distances, facilitating easy repair compared with 1000 sublethal, single-stranded breaks (SSBs) and a greater number of nonlethal lesions to base pairs.6,9,23-25 DNA damage from photon and proton therapy arises primarily through indirect ionization of reactive oxygen species (ROS) generated through water radiolysis, rather than through direct interaction with beam energy or δ-rays.26-28 This indirect ionization requires cellular oxygen to “fix” lesions, which explains the relative radioresistance of hypoxic tissues to photon and proton radiation and the similarity of their oxygen enhancement ratios (OERs), both approaching the maximum value of 3.23,26-32 Sensitivity to photon and proton radiation is also modulated by the cell cycle. It is diminished during the G1 and S phases of the standard cell cycle when DSBs are more isolated due to chromatin condensation. Cell cycle radiosensitivity reaches its lowest point in the G0 phase, associated with stem-like cell states, when DNA repair mechanisms are at their most robust.23,33-36
Conventional radiation therapy (RT) is susceptible to synergistic cellular and microenvironmental resistance mechanisms caused by the structural dispersion of photon and proton beam energy, leading to entropy directly or through an intermediary of clonogenically insignificant isolated interactions. At the extreme end of contumacious CSC biology in PDAC and GBM, limited successes and serial failures of radical dose escalation of conventional RT may be caused by the microenvironmental CSC niche acting effectively as an energy sink. Higher doses in proximity to critical organs-at-risk (OARs) and/or exceeding volumetric thresholds threaten to narrow to the point of futility or even invert the therapeutic ratio, thereby allowing CSCs to escape any genuine clonogenic peril posed by conventional radiation.37 LET escalation, conversely, obviates the futility of dose escalation and holds promise as a clinical solution to the CSC problem.
LET-Induced DNA Complexity and Repair Disruption
High LET radiation therapy differs from conventional RT at all physical doses. At the 100-101 nm scale of single DNA molecules, increasing ionization density brings a higher overall yield of DSB, a higher yield of clustered DSB—that is, 2 or more DSB produced by a single radiation track up to ~25 bp or 1-2 helical turns apart on a single DNA molecule—and a higher probability of direct rather than ROS-dependent indirect damage.2,23,38,39 Whereas sparse DSB promote anti-apoptotic pathways, particularly in p53-mutated stem-associated phenotypes typical of high-grade cancers, accumulation of clustered DSB reverses this pattern, suppressing anti-apoptotic BCL family factors prominently, and enabling non-p53-mediated apoptosis where the p53 pathway has been lost.40-44 Experimental studies show that sufficiently high LET can efficiently induce apoptosis irrespective of p53 status.45
Clustered DSBs in proximity to each other create complex, multi-track lesions causing deletions upwards of 1000 bp in size, compared with <100 bp typical for low LET modalities. DNA repair mechanisms become less efficient and more error-prone, specifically by exhaustion of the high-fidelity homologous recombination mechanism forcing reliance on low-fidelity non-homologous end joining; SSB and base damages along the same track as DSB augment lethality as shown by XRCC1 recruitment at DSB foci.1,38,46-48 High LET lesions also slow down the DNA repair process, shown by more durable γH2AX phosphorylation, inducing failed repair of potentially lethal lesions and sublethal lesions made lethal by second hits.49
Chromosomal Catastrophe and Multiple Cell Death Pathways
Frequent geometric variation of DSB promotes lethal mutations within chromosomes and, along with the fragmentation of large intact segments >1000 bp, exposes lesioned strands on different chromosomes to each other, inducing aberrant, nonviable chromosomal rearrangements such as rings, dicentrics, and micronuclei.1,46,50-52 The volume and variability of lesion geometry, chromosomal rearrangement, and iteratively misrepaired DSB trigger cell death by mechanisms within and beyond the apoptotic spectrum, including autophagy, senescence, necroptosis, and necrosis.48,51,53-60 Notably, impairment and delay of DNA repair cause cells with accumulated unrepaired DSBs escaping apoptosis and G2/M checkpoint arrest to form micronuclei upon entry into mitosis.61-63
In contrast to the stemness-promoting EMT often induced by conventional low LET irradiation, high LET irradiation tends to reverse EMT. Low LET promotes diffuse ROS and stimulates HIF complex stabilization with downstream EMT products, whereas high LET causes HIF destabilization and downregulation of its products.64,65 Both high and low LET can produce ROS efficiently, yet the expected signaling pathways triggered by ROS, such as PI3K/Akt and EGFR, are suppressed under high LET irradiation.66-70 Due to the density of indirect and direct ionization, structured propagation of ROS and oxidative stress occurs only locally along the track, evading the spatial distribution element required to trigger the usual signaling pathways.71-73
Multiscale Damage: From DNA to Tissue Architecture
High ionization density of heavy particles leads to intrinsically more lethal DNA lesions, overwhelms multiple DNA repair pathways, sensitizes hypoxic cells through various mechanisms, and activates cell death pathways that counteract resistance. These processes propagate over increasingly large structural scales. Clustered DSBs from separate tracks can aggregate into more complex lesions affecting structures from the level of nucleosomes (101 nm), chromatin loops (102 nm), entire chromosomes (103 nm), and multiple chromosomes across whole nuclei (104 nm), up to the architectural scale of tissues, including extranuclear organelles, neighboring cells, ECM, and stroma (105 nm).
At sufficiently high ionization density, a single radiation track can cause complex damage. Additionally, the extension of single-track clustered lesions across the chromatin ultrastructure multiplies the formation of lethal chromosomal aberrations and radiosensitizes resistant cell cycle phases as the shielding effect of chromatin packing and coiling is lost.47,51,74
Cytosolic DNA Exposure, Innate Immunity, and Abscopal Effects
As LET rises, progressively complex DNA damage leads to the formation of increasingly small, cleaved DNA fragments (<40 bp) capable of diffusing into the cytosol through intermittent nuclear membrane disruption. In parallel, persistent unrepaired or aberrantly repaired chromosomal lesions can rupture into the cytosol from unstable micronuclei—sometimes undergoing explosive fragmentation, as seen in chromothripsis.61,62,75-77 Neoantigenic recognition in the extranuclear environment by damage-associated molecular pattern-sensing mechanisms triggers cascades of pro-immune cytokines stimulating MHC-1 upregulation and surface expression, TH1 > TH2 and M1 > M2 polarizations, dendritic cell maturation and phagocytosis, CD4 and CD8 lymphocyte infiltration, antitumoral natural killer (NK) cell priming, etc, culminating in immune cell death through both innate and adaptive immunity.1,3,78-84 These immune effects may extend beyond the local tumor microenvironment to produce abscopal effects—immune-mediated regression of distant, untreated lesions.85-100
These features help explain the unique efficacy of high LET irradiation in targeting CSCs, as demonstrated in basic research. In both in vitro and in vivo settings, alone and in combination with amplifying systemic agents, CIRT has been shown to reduce or eliminate the clonogenically viable subset of CD44-marked PDAC-CSCs.101-103 In the GBM setting, HPT has shown even more robust findings in multiply reproduced culture and xenograft experiments. Both CIRT and high LET neutron therapy eradicate GBM-CSCs expressing the major CSC marker CD133 and shift the transcriptional profile of GBM cells away from pathways associated with stemness, invasion, angiogenesis, hypoxia, chemoradioresistance, and proliferation, including specific suppression of major stemness/EMT signaling pathways, including Akt and EGFR.67,68,104-109 CSC sensitivity to CIRT has been shown to extend to multiple cancers in individual studies and systematic reviews.110-113
Toward Optimal LET for CSC Eradication
Within the typical LET range of the carbon ion SOBP of roughly 40-80 keV/µm, the OER is reduced but not fully abrogated, meaning some CSC-associated radioresistance mechanisms may persist. To achieve complete hypoxia-independent CSC sterilization, more massive ions such as oxygen, nitrogen, or neon—capable of generating LET ≥ 100 keV/µm within SOBP—may be required. However, practical application of ions with effective charge numbers (Zeff) > 10 is likely limited by unmitigable toxicity to normal tissue.1,29,72,114-119
Conclusion
The physical and biological underpinnings of HPT, especially its ability to target CSCs via high LET-induced damage, provide a compelling foundation for its clinical application in aggressive and treatment-resistant tumors. GBM and PDAC, relatively common cancers with stubbornly poor prognoses, are auspicious use cases for CSC-directed therapy. In Part 2, we apply the foregoing principles to advanced techniques such as LET painting, biologically optimized spatial fractionation, and multi-ion therapy. These strategies are necessary to allow for the safe delivery of HPT, enabling the realization of its biological potential. We further explore the logistical barriers to wide implementation of HPT and prospects for overcoming them.
References
- Tinganelli W, Durante M. Carbon ion radiobiology. Cancers (Basel). 2020;12(10). doi:10.3390/cancers12103022.
- Durante M, Loeffler J. Charged particles in radiation oncology. Nat Rev Clin Oncol. 2010;7(1):37-43. doi:10.1038/nrclinonc.2009.183.
- Helm A, Fournier C. High-LET charged particles: radiobiology and application for new approaches in radiotherapy. Strahlenther Onkol. 2023;199(12):1225-1241. doi:10.1007/s00066-023-02158-7.
- Tommasino F, Scifoni E, Durante M. New ions for therapy. Int J Part Ther. 2016;2(3):428-438. doi:10.14338/IJPT-15-00027.1.
- Johnson D, Chen Y, Ahmad S. Dose and linear energy transfer distributions of primary and secondary particles in carbon ion radiation therapy: a monte carlo simulation study in water. J Med Phys. 2015;40(4):214-219. doi:10.4103/0971-6203.170785.
- Brahme A. Recent advances in light ion radiation therapy. Int J Radiat Oncol. 2004;58(2):603-616. doi:10.1016/j.ijrobp.2003.09.034.
- Kempe J, Gudowska I, Brahme A. Depth absorbed dose and LET distributions of therapeutic 1H, 4He, 7Li, and 12C beams. Med Phys. 2007;34(1):183-192. doi:10.1118/1.2400621.
- Kantemiris I, Karaiskos P, Papagiannis P, Angelopoulos A. Dose and dose averaged LET comparison of 1H. Med Phys. 2011;38(12):6585-6591. doi:10.1118/1.3662911.
- Tommasino F, Durante M. Proton radiobiology. Cancers (Basel). 2015;7(1):353-381. doi:10.3390/cancers7010353.
- Smith E, Winterhalter C, Underwood T. A monte carlo study of different LET definitions and calculation parameters for proton beam therapy. Biomed Phys Eng Express. 2021;8(1). doi:10.1088/2057-1976/ac3f50.
- Murshed H, Murshed H. Fundamentals of Radiation Oncology. 2019:57-87. doi:10.1016/B978-0-12-814128-1.00003-9.
- Podgoršak E. Radiation Oncology Physics: A Handbook for Teachers and Students. 2005.
- Orton C. Radiation Dosimetry: Physical and Biological Aspects. 1986. doi:10.1007/978-1-4899-0571-0.
- Byun H, Han M, Yang K. Physical and biological characteristics of particle therapy for oncologists. Cancer Res Treat. 2021;53(3):611-620. doi:10.4143/crt.2021.066.
- Giovannini G, Böhlen T, Cabal G. Variable RBE in proton therapy: comparison of different model predictions and their influence on clinical-like scenarios. Radiat Oncol. 2016;11(1). doi:10.1186/s13014-016-0642-6.
- McNamara A, Willers H, Paganetti H. Modelling variable proton relative biological effectiveness for treatment planning. Br J Radiol. 2020;93(1107). doi:10.1259/bjr.20190334.
- Underwood T, McNamara A, Appelt A. A systematic review of clinical studies on variable proton Relative Biological Effectiveness (RBE). Radiother Oncol. 2022;175:79-92. doi:10.1016/j.radonc.2022.08.014.
- McMahon S, Paganetti H, Prise K. LET-weighted doses effectively reduce biological variability in proton radiotherapy planning. Phys Med Biol. 2018;63(22). doi:10.1088/1361-6560/aae8a5.
- Knäusl B, Fuchs H, Dieckmann K, Georg D. Can particle beam therapy be improved using helium ions? - a planning study focusing on pediatric patients. Acta Oncol. 2016;55(6):751-759. doi:10.3109/0284186X.2015.1125016.
- Durante M, Debus J, Loeffler J. Physics and biomedical challenges of cancer therapy with accelerated heavy ions. Nat Rev Phys. 2021;3(12):777-790. doi:10.1038/s42254-021-00368-5.
- Schlaff C, Krauze A, Belard A, O’Connell J, Camphausen K. Bringing the heavy: carbon ion therapy in the radiobiological and clinical context. Radiat Oncol. 2014;9(1):88. doi:10.1186/1748-717X-9-88.
- Weber U, Kraft G. Comparison of carbon ions versus protons. Cancer J. 2009;15(4):325-332. doi:10.1097/PPO.0b013e3181b01935.
- Hill M. Radiation track structure: how the spatial distribution of energy deposition drives biological response. Clin Oncol. 2020;32(2):75-83. doi:10.1016/j.clon.2019.08.006.
- Hall E, Giaccia A. Radiobiology for the Radiologist. 2019.
- Kinner A, Wu W, Staudt C, Iliakis G. Gamma-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res. 2008;36(17):5678-5694. doi:10.1093/nar/gkn550.
- Desouky O, Ding N, Zhou G. Targeted and non-targeted effects of ionizing radiation. J Radiat Res Appl Sci. 2015;8(2):247-254. doi:10.1016/j.jrras.2015.03.003.
- Lee K, Lee J, Park J, Huh T, Lee Y. Low energy proton beam induces tumor cell apoptosis through reactive oxygen species and activation of caspases. Exp Mol Med. 2008;40(1):118-129. doi:10.3858/emm.2008.40.1.118.
- Rakotomalala A, Escande A, Furlan A, Meignan S, Lartigau E. Hypoxia in solid tumors: how low oxygenation impacts the “six Rs” of radiotherapy. Front Endocrinol. 2021;12. doi:10.3389/fendo.2021.742215.
- Scifoni E, Tinganelli W, Weyrather W. Including oxygen enhancement ratio in ion beam treatment planning: model implementation and experimental verification. Phys Med Biol. 2013;58(11):3871-3895. doi:10.1088/0031-9155/58/11/3871.
- Stewart R, Streitmatter S, Argento D. Rapid MCNP simulation of DNA Double Strand Break (DSB) Relative Biological Effectiveness (RBE) for photons, neutrons, and light ions. Phys Med Biol. 2015;60(21):8249-8274. doi:10.1088/0031-9155/60/21/8249.
- Bouleftour W, Rowinski E, Louati S. A review of the role of hypoxia in radioresistance in cancer therapy. Med Sci Monit. 2021;27. doi:10.12659/MSM.934116.
- Crabtree H, Cramer W, Murray J. The action of radium on cancer cells. II—some factors determining the susceptibility of cancer cells to radium. Proc R Soc Lond B. 1933;113(782):238-250. doi:10.1098/rspb.1933.0044.
- Pawlik T, Keyomarsi K. Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys. 2004;59(4):928-942. doi:10.1016/j.ijrobp.2004.03.005.
- Krude T, Keller C. Chromatin assembly during s phase: contributions from histone deposition, DNA replication and the cell division cycle. Cell Mol Life Sci. 2001;58(5-6):665-672. doi:10.1007/pl00000890.
- Ma Y, Kanakousaki K, Buttitta L. How the cell cycle impacts chromatin architecture and influences cell fate. Front Genet. 2015;6. doi:10.3389/fgene.2015.00019.
- Zhao L, Wu D, Mi D, Sun Y. Radiosensitivity and relative biological effectiveness based on a generalized target model. J Radiat Res. 2017;58(1):8-16. doi:10.1093/jrr/rrw062.
- Chan J, Lee S, Fraass B. Survival and failure patterns of high-grade gliomas after three-dimensional conformal radiotherapy. J Clin Oncol. 2002;20(6):1635-1642. doi:10.1200/JCO.2002.20.6.1635.
- Liu W, Wu R, Guo J. High turnover and rescue effect of XRCC1 in response to heavy charged particle radiation. Biophys J. 2022;121(8):1493-1501. doi:10.1016/j.bpj.2022.03.011.
- Hirayama R, Ito A, Tomita M. Contributions of direct and indirect actions in cell killing by high-LET radiations. Radiat Res. 2009;171(2):212-218. doi:10.1667/RR1490.1.
- Lips J, Kaina B. DNA double-strand breaks trigger apoptosis in p53-deficient fibroblasts. Carcinogenesis. 2001;22(4):579-585. doi:10.1093/carcin/22.4.579.
- Roos W, Kaina B. DNA damage-induced cell death by apoptosis. Trends Mol Med. 2006;12(9):440-450. doi:10.1016/j.molmed.2006.07.007.
- Zhang Y, Dube C, Gibert M. The p53 pathway in glioblastoma. Cancers. 2018;10(9):297. doi:10.3390/cancers10090297.
- Weissmueller S, Manchado E, Saborowski M. Mutant p53 drives pancreatic cancer metastasis through cell-autonomous PDGF receptor β signaling. Cell. 2014;157(2):382-394. doi:10.1016/j.cell.2014.01.066.
- Li S, Huang H, Xing M. Carbon ion induces cell death and G2/m arrest through pRb/E2F1Chk2/Cdc2 signaling pathway in x-ray resistant B16F10 melanoma cells. Dose Response. 2022;20(2). doi:10.1177/15593258221092364.
- Takahashi A, Matsumoto H, Furusawa Y. Apoptosis induced by high-LET radiations is not affected by cellular p53 gene status. Int J Radiat Biol. 2005;81(8):581-586. doi:10.1080/09553000500280484.
- Asaithamby A, Hu B, Chen D. Unrepaired clustered DNA lesions induce chromosome breakage in human cells. Proc Natl Acad Sci USA. 2011;108(20):8293-8298. doi:10.1073/pnas.1016045108.
- Mladenova V, Mladenov E, Stuschke M, Iliakis G. DNA damage clustering after ionizing radiation and consequences in the processing of chromatin breaks. Molecules. 2022;27(5):1540. doi:10.3390/molecules27051540.
- Hagiwara Y, Oike T, Niimi A. Clustered DNA double-strand break formation and the repair pathway following heavy-ion irradiation. Journal of Radiation Research. 2019;60(1):69-79. doi:10.1093/jrr/rry096.
- Lopez Perez R, Nicolay N, Wolf J. DNA damage response of clinical carbon ion versus photon radiation in human glioblastoma cells. Radiother Oncol J Eur Soc Ther Radiol Oncol. 2019;133:77-86. doi:10.1016/j.radonc.2018.12.028.
- Ward J. The complexity of DNA damage: relevance to biological consequences. Int J Radiat Biol. 1994;66(5):427-432. doi:10.1080/09553009414551401.
- Wilkinson B, Hill M, Parsons J. The cellular response to complex DNA damage induced by ionising radiation. Int J Mol Sci. 2023;24(5). doi:10.3390/ijms24054920.
- Singleton B, Griffin C, Thacker J. Clustered DNA damage leads to complex genetic changes in irradiated human cells. Cancer Res. 2002;62(21):6263-6269.
- Borges H, Linden R, Wang J. DNA damage-induced cell death. Cell Res. 2008;18(1):17-26. doi:10.1038/cr.2007.110.
- Wang J. DNA damage and apoptosis. Cell Death Differ. 2001;8(11):1047-1048. doi:10.1038/sj.cdd.4400938.
- Jinno-Oue A, Shimizu N, Hamada N. Irradiation with carbon ion beams induces apoptosis, autophagy, and cellular senescence in a human glioma-derived cell line. Int J Radiat Oncol. 2010;76(1):229-241. doi:10.1016/j.ijrobp.2009.08.054.
- Sai S, Koto M, Yamada S. Basic and translational research on carbon-ion radiobiology. Am J Cancer Res. 2023;13(1):1-24.
- Wang X, Chen X, Li G. Application of carbon ion and its sensitizing agent in cancer therapy: a systematic review. Front Oncol. 2021;11:708724. doi:10.3389/fonc.2021.708724.
- Koom W, Sai S, Suzuki M. Superior effect of the combination of carbon-ion beam irradiation and 5-fluorouracil on colorectal cancer stem cells in vitro and in vivo. Onco Targets Ther. 2020;13:12625-12635. doi:10.2147/OTT.S276035.
- Chaurasia M, Bhatt A, Das A, Dwarakanath B, Sharma K. Radiation-induced autophagy: mechanisms and consequences. Free Radic Res. 2016;50(3):273-290. doi:10.3109/10715762.2015.1129534.
- Bao C, Sun Y, Dwarakanath B. Carbon ion triggered immunogenic necroptosis of nasopharyngeal carcinoma cells involving necroptotic inhibitor BCL-x. J Cancer. 2021;12(5):1520-1530. doi:10.7150/jca.46316.
- Roberts C, Morgan G, Holt P. A critical comparison of the micronucleus yield from high and low LET irradiation of plateau-phase cell populations. Mutat Res. 1986;160(3):237-242. doi:10.1016/0027-5107(86)90133-8.
- Durante M, Formenti S. Radiation-induced chromosomal aberrations and immunotherapy: micronuclei, cytosolic DNA, and interferon-production pathway. Front Oncol. 2018;8. doi:10.3389/fonc.2018.00192.
- Akudugu J, Slabbert J, Roos W, Böhm L. Micronucleus response of human glioblastoma and neuroblastoma cells toward low-LET photon and high-LET p(66)/be neutron irradiation. Am J Clin Oncol. 2003;26(3). doi:10.1097/01.COC.0000072500.18954.2D.
- Subtil F, Wilhelm J, Bill V. Carbon ion radiotherapy of human lung cancer attenuates HIF-1 signaling and acts with considerably enhanced therapeutic efficiency. FASEB J. 2014;28(3):1412-1421. doi:10.1096/fj.13-242230.
- Wozny A, Lauret A, Battiston-Montagne P. Differential pattern of HIF-1α expression in HNSCC cancer stem cells after carbon ion or photon irradiation: one molecular explanation of the oxygen effect. Br J Cancer. 2017;116(10):1340-1349. doi:10.1038/bjc.2017.100.
- Konings K, Vandevoorde C, Baselet B, Baatout S, Moreels M. Combination therapy with charged particles and molecular targeting: a promising avenue to overcome radioresistance. Front Oncol. 2020;10. doi:10.3389/fonc.2020.00128.
- Stahler C, Roth J, Cordes N, Taucher-Scholz G, Mueller-Klieser W. Impact of carbon ion irradiation on epidermal growth factor receptor signaling and glioma cell migration in comparison to conventional photon irradiation. Int J Radiat Biol. 2013;89(6):454-461. doi:10.3109/09553002.2013.766769.
- Jin X, Li F, Zheng X. Carbon ions induce autophagy effectively through stimulating the unfolded protein response and subsequent inhibiting akt phosphorylation in tumor cells. Sci Rep. 2015;5. doi:10.1038/srep13815.
- Shi X, Zhang Y, Zheng J, Pan J. Reactive oxygen species in cancer stem cells. Antioxid Redox Signal. 2012;16(11):1215-1228. doi:10.1089/ars.2012.4529.
- Checa J, Aran J. Reactive oxygen species: drivers of physiological and pathological processes. J Inflamm Res. 2020;13:1057-1073. doi:10.2147/JIR.S275595.
- Tsao D, Kalogerinis P, Tabrizi I. Induction and processing of oxidative clustered DNA lesions in 56fe-ion-irradiated human monocytes. Radiat Res. 2007;168(1):87-97. doi:10.1667/RR0865.1.
- Stewart R, Yu V, Georgakilas A. Effects of radiation quality and oxygen on clustered DNA lesions and cell death. Radiat Res. 2011;176(5):587-602. doi:10.1667/rr2663.1.
- Wozny A, Rodriguez-Lafrasse C. The “stealth-bomber” paradigm for deciphering the tumour response to carbon-ion irradiation. Br J Cancer. 2023;128(8):1429-1438. doi:10.1038/s41416-022-02117-6.
- Lorat Y, Timm S, Jakob B, Taucher-Scholz G, Rübe C. Clustered double-strand breaks in heterochromatin perturb DNA repair after high linear energy transfer irradiation. Radiother Oncol J Eur Soc Ther Radiol Oncol. 2016;121(1):154-161. doi:10.1016/j.radonc.2016.08.028.
- Gekara N. DNA damage-induced immune response: micronuclei provide key platform. J Cell Biol. 2017;216(10):2999-3001. doi:10.1083/jcb.201708069.
- Pang D, Winters T, Jung M. Radiation-generated short DNA fragments may perturb non-homologous end-joining and induce genomic instability. J Radiat Res. 2011;52(3):309-319. doi:10.1269/jrr.10147.
- Pang D, Chasovskikh S, Rodgers J, Dritschilo A. Short DNA fragments are a hallmark of heavy charged-particle irradiation and may underlie their greater therapeutic efficacy. Front Oncol. 2016;6. doi:10.3389/fonc.2016.00130.
- Huang Y, Dong Y, Zhao J. Comparison of the effects of photon, proton and carbon-ion radiation on the ecto-calreticulin exposure in various tumor cell lines. Ann Transl Med. 2019;7(20). doi:10.21037/atm.2019.09.128.
- Yoshimoto Y, Oike T, Okonogi N. Carbon-ion beams induce production of an immune mediator protein, high mobility group box 1, at levels comparable with x-ray irradiation. J Radiat Res. 2015;56(3):509-514. doi:10.1093/jrr/rrv007.
- Ebner D, Tinganelli W, Helm A. The immunoregulatory potential of particle radiation in cancer therapy. Front Immunol. 2017;8. doi:10.3389/fimmu.2017.00099.
- Zhou H, Tu C, Yang P. Carbon ion radiotherapy triggers immunogenic cell death and sensitizes melanoma to anti-PD-1 therapy in mice. Oncoimmunology. 2022;11(1). doi:10.1080/2162402X.2022.2057892.
- Onishi M, Okonogi N, Oike T. High linear energy transfer carbon-ion irradiation increases the release of the immune mediator high mobility group box 1 from human cancer cells. J Radiat Res. 2018;59(5):541-546. doi:10.1093/jrr/rry049.
- Guo Y, Shen R, Wang F. Carbon ion irradiation induces DNA damage in melanoma and optimizes the tumor microenvironment based on the cgas–STING pathway. J Cancer Res Clin Oncol. 2023;149(9):6315-6328. doi:10.1007/s00432-023-04577-6.
- Pompos A, Durante M, Choy H. Heavy ions in cancer therapy. JAMA Oncol. 2016;2(12):1539-1540. doi:10.1001/jamaoncol.2016.2646.
- Matsunaga A, Ueda Y, Yamada S. Carbon-ion beam treatment induces systemic antitumor immunity against murine squamous cell carcinoma. Cancer. 2010;116(15):3740-3748. doi:10.1002/cncr.25134.
- Lussier D, Alspach E, Ward J. Radiation-induced neoantigens broaden the immunotherapeutic window of cancers with low mutational loads. Proc Natl Acad Sci U S A. 2021;118(24). doi:10.1073/pnas.2102611118.
- Ando K, Fujita H, Hosoi A. Intravenous dendritic cell administration enhances suppression of lung metastasis induced by carbon-ion irradiation. J Radiat Res. 2017;58(4):446-455. doi:10.1093/jrr/rrx005.
- Ogata T, Teshima T, Kagawa K. Particle irradiation suppresses metastatic potential of cancer cells. Cancer Res. 2005;65(1):113-120.
- Akino Y, Teshima T, Kihara A. Carbon-ion beam irradiation effectively suppresses migration and invasion of human non-small-cell lung cancer cells. Int J Radiat Oncol Biol Phys. 2009;75(2):475-481. doi:10.1016/j.ijrobp.2008.12.090.
- Takahashi Y, Yasui T, Minami K. Carbon ion irradiation enhances the antitumor efficacy of dual immune checkpoint blockade therapy both for local and distant sites in murine osteosarcoma. Oncotarget. 2019;10(6):633-646. doi:10.18632/oncotarget.26551.
- Fujita M, Imadome K, Shoji Y. Carbon-ion irradiation suppresses migration and invasiveness of human pancreatic carcinoma cells miapaca-2 via rac1 and rhoa degradation. Int J Radiat Oncol Biol Phys. 2015;93(1):173-180. doi:10.1016/j.ijrobp.2015.05.009.
- Harding S, Benci J, Irianto J. Mitotic progression following DNA damage enables pattern recognition within micronuclei. Nature. 2017;548(7668):466-470. doi:10.1038/nature23470.
- Katsuki S, Takahashi Y, Tamari K. Radiation therapy enhances systemic antitumor efficacy in PD-L1 therapy regardless of sequence of radiation in murine osteosarcoma. PLOS ONE. 2022;17(7). doi:10.1371/journal.pone.0271205.
- Zhou H, Yang P, Li H. Carbon ion radiotherapy boosts anti-tumour immune responses by inhibiting myeloid-derived suppressor cells in melanoma-bearing mice. Cell Death Discov. 2021;7(1):332. doi:10.1038/s41420-021-00731-6.
- Iijima M, Okonogi N, Nakajima N. Significance of PD-L1 expression in carbon-ion radiotherapy for uterine cervical adeno/adenosquamous carcinoma. J Gynecol Oncol. 2020;31(2). doi:10.3802/jgo.2020.31.e19.
- Hartmann L, Schröter P, Osen W. Photon versus carbon ion irradiation: immunomodulatory effects exerted on murine tumor cell lines. Sci Rep. 2020;10(1). doi:10.1038/s41598-020-78577-8.
- Du J, Kageyama S, Hirata H. Comparative analysis of the immune responses in cancer cells irradiated with x-ray, proton and carbon-ion beams. Biochem Biophys Res Commun. 2021;585:55-60. doi:10.1016/j.bbrc.2021.11.004.
- Wang J, Dai Z, Miao Y. Carbon ion (12C6+) irradiation induces the expression of klrk1 in lung cancer and optimizes the tumor microenvironment based on the NKG2D/NKG2D-ls pathway. Cancer Letters. 2021;521:178-195. doi:10.1016/j.canlet.2021.09.003.
- Moncharmont C, Guy J, Wozny A. Carbon ion irradiation withstands cancer stem cells’ migration/invasion process in Head and Neck Squamous Cell Carcinoma (HNSCC). Oncotarget. 2016;7(30):47738-47749. doi:10.18632/oncotarget.10281.
- Tubin S, Yan W, Mourad W, Fossati P, Khan M. The future of radiation-induced abscopal response: beyond conventional radiotherapy approaches. Future Oncol. 2020;16(16):1137-1151. doi:10.2217/fon-2020-0063.
- Sai S, Kim E, Koom W. Carbon-ion beam irradiation and the mir-200c mimic effectively eradicate pancreatic cancer stem cells under in vitro and in vivo conditions. OncoTargets Ther. 2021;14:4749-4760. doi:10.2147/OTT.S311567.
- Sai S, Wakai T, Vares G. Combination of carbon ion beam and gemcitabine causes irreparable DNA damage and death of radioresistant pancreatic cancer stem-like cells in vitro and in vivo. Oncotarget. 2015;6(8):5517-5535. doi:10.18632/oncotarget.3584.
- Oonishi K, Cui X, Hirakawa H. Different effects of carbon ion beams and x-rays on clonogenic survival and DNA repair in human pancreatic cancer stem-like cells. Radiother Oncol. 2012;105(2):258-265. doi:10.1016/j.radonc.2012.08.009.
- Baek S, Ishii H, Tamari K. Cancer stem cells: the potential of carbon ion beam radiation and new radiosensitizers (review). Oncol Rep. 2015;34(5):2233-2237. doi:10.3892/or.2015.4236.
- Chiblak S, Tang Z, Lemke D. Carbon irradiation overcomes glioma radioresistance by eradicating stem cells and forming an antiangiogenic and immunopermissive niche. JCI Insight. 2019;4(2). doi:10.1172/jci.insight.123837.
- Chiblak S, Tang Z, Campos B. Radiosensitivity of patient-derived glioma stem cell 3-dimensional cultures to photon, proton, and carbon irradiation. Int J Radiat Oncol. 2016;95(1):112-119. doi:10.1016/j.ijrobp.2015.06.015.
- Hirota Y, Masunaga S, Kondo N. High linear-energy-transfer radiation can overcome radioresistance of glioma stem-like cells to low linear-energy-transfer radiation. J Radiat Res. 2014;55(1):75-83. doi:10.1093/jrr/rrt095.
- Ciamarone F, Meister S, Furkel J. Abstract 1105: carbon ion radiotherapy eradicates glioblastoma via tumor immune environment reprograming. Cancer Res. 2023;83(7_supplement):1105-1105. doi:10.1158/1538-7445.AM2023-1105.
- Valable S, Gérault A, Lambert G. Impact of hypoxia on carbon ion therapy in glioblastoma cells: modulation by LET and hypoxia-dependent genes. Cancers (Basel). 2020;12(8). doi:10.3390/cancers12082019.
- Vares G, Ahire V, Sunada S. A multimodal treatment of carbon ions irradiation, mirna-34 and mtor inhibitor specifically control high-grade chondrosarcoma cancer stem cells. Radiother Oncol. 2020;150:253-261. doi:10.1016/j.radonc.2020.07.034.
- Sai S, Vares G, Kim E. Carbon ion beam combined with cisplatin effectively disrupts triple negative breast cancer stem-like cells in vitro. Mol Cancer. 2015;14(1). doi:10.1186/s12943-015-0429-7.
- Wang Q, Liu R, Zhang Q. Biological effects of cancer stem cells irradiated by charged particle: a systematic review of in vitro studies. J Cancer Res Clin Oncol. 2023;149(9):6625-6638. doi:10.1007/s00432-022-04561-6.
- Cui X, Oonishi K, Tsujii H. Effects of carbon ion beam on putative colon cancer stem cells and its comparison with x-rays. Cancer Res. 2011;71(10):3676-3687. doi:10.1158/0008-5472.CAN-10-2926.
- Lee S, Mizushima K, Kohno R. Estimating the biological effects of helium, carbon, oxygen, and neon ion beams using 3D silicon microdosimeters. Phys Med Biol. 2021;66(4). doi:10.1088/1361-6560/abd66f.
- Sokol O, Scifoni E, Tinganelli W. Oxygen beams for therapy: advanced biological treatment planning and experimental verification. Phys Med Biol. 2017;62(19):7798-7813. doi:10.1088/1361-6560/aa88a0.
- Kase Y, Kanai T, Matsumoto Y. Microdosimetric measurements and estimation of human cell survival for heavy-ion beams. Radiat Res. 2006;166(4):629-638. doi:10.1667/RR0536.1.
- Inaniwa T, Suzuki M, Hyun Lee S. Experimental validation of stochastic microdosimetric kinetic model for multi-ion therapy treatment planning with helium-, carbon-, oxygen-, and neon-ion beams. Phys Med Biol. 2020;65(4). doi:10.1088/1361-6560/ab6eba.
- Tessonnier T, Mairani A, Brons S. Experimental dosimetric comparison of 1H, 4He, 12C and 16O scanned ion beams. Phys Med Biol. 2017;62(10). doi:10.1088/1361-6560/aa6516.
- Tran L, Bolst D, Guatelli S. The relative biological effectiveness for carbon, nitrogen, and oxygen ion beams using passive and scanning techniques evaluated with fully 3D silicon microdosimeters. Med Phys. 2018;45(5):2299-2308. doi:10.1002/mp.12874.
Disclosures
The authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere.
Citation
. Eradicating Cancer Stem Cells Using High Linear Energy Transfer Radiation Therapy Part 1: Physics and Radiobiology. Applied Radiation Oncology. 2025;14(2):1-8. doi:10.37549/ARO-D-25-00003P1.