Clinical Evidence of Combining Radiopharmaceutical Therapy With Immune Checkpoint Inhibitors
Applied Radiation Oncology — Vol. 14, Issue 1
Published: March 1, 2025
1 Department of Medical Physics, University of Wisconsin-Madison, Madison, WI
2 Department of Cancer Biology, University of Wisconsin-Madison, Madison, WI
3 Department of Radiology, University of Wisconsin-Madison, Madison, WI
4 Carbone Cancer Center, University of Wisconsin-Madison, Madison, WI
5 Department of Human Oncology, University of Wisconsin-Madison, Madison, WI
6 William S. Middleton Memorial Veterans Hospital, Madison, WI
* Corresponding author: Quaovi H. Sodji (qsodji@humonc.wisc.edu)
Abstract
Keywords
metastatic cancer, radiopharmaceutical therapy, β-particle emitters, α-particle emitters, immune checkpoint inhibitors
Introduction
Metastatic disease accounts for approximately 90% of cancer-related deaths.1-6 Unfortunately, effective therapeutic strategies remain limited despite tremendous advances in cancer research.7 Radiopharmaceutical therapy (RPT) represents a groundbreaking approach to treating metastatic disease by delivering targeted radiation to tumors throughout the body.8,9 Leveraging pharmaceuticals that selectively bind to cancer cells or accumulate through physiological mechanisms, RPT provides a precise and effective treatment modality. Remarkably, RPT has demonstrated significant therapeutic efficacy with minimal toxicity in several cancer types.8 As the role of RPT in metastatic disease management is on the rise, its combination with immune checkpoint inhibitors (ICIs) holds the potential to enhance clinical responses beyond that achievable by either monotherapy alone.
For over a century, radiation therapy, including external beam radiation therapy (EBRT) and RPT, has shown dual benefits: tumor eradication and immune activation.10 Radiation triggers cancer cells to release damage-associated molecular patterns (DAMPs), activating the cGAS-STING pathway, which induces type I interferons and the release of cytokines that recruit immune cells.11,12 The growing promise of RPT in treating metastatic cancer, coupled with emerging insights into the immunogenic effects of radiation, has spurred preclinical and clinical studies exploring the combination of RPT and immunotherapy, such as ICIs. This review explores clinical trials investigating the combination of RPT with ICI, highlighting key clinical findings, potential challenges, and future directions in this emerging field.
Radiopharmaceutical Therapy
RPT has emerged as a promising systemic therapy, enabling radiation delivery to both local and metastatic lesions while sparing healthy tissues (Figure 1A).8,13 Unlike EBRT, which delivers radiation to all tissues in the radiation field, including malignant and adjacent normal tissues, RPT uses tumor-targeting biomolecules (eg, antibodies, peptides, or small molecules) linked to a radionuclide to form a “radiopharmaceutical” that preferentially targets cancer cells (Figure 1). The radiopharmaceutical binds selectively to receptors overexpressed on tumor cells, thus delivering radiation to the tumor while minimizing damage to surrounding tissues.14 This molecularly targeted approach makes RPT particularly effective for treating metastatic and microscopic tumors,8,15-17 where EBRT’s utility is often limited. The efficacy of RPT depends on the targeting molecule’s properties, the radionuclide’s physical characteristics, and tumor characteristics such as receptor expression, size, and tumor type. Additional factors, such as the administrated activity, tumor uptake, and pharmacokinetics, also impact the treatment outcome.18 Therefore, carefully considering these factors is crucial for RPT’s clinical efficacy and safety. The approvals of several radiopharmaceuticals, such as [223Ra]Ra-dichloride (Xofigo) and [177Lu]Lu-PSMA-167 (Pluvicto) for metastatic castration-resistant prostate cancer (mCRPC) and [177Lu]Lu-DOTATATE (Lutathera) for gastroenteropancreatic neuroendocrine tumors (GEP-NETs), have sparked a new excitement in the field.19-21 RPT faces challenges like suboptimal targeting, radioresistance, and limited immune stimulation, hindering tumor eradication.22-24 Combining RPT with systemic therapies like ICIs may overcome these limitations and improve outcomes.
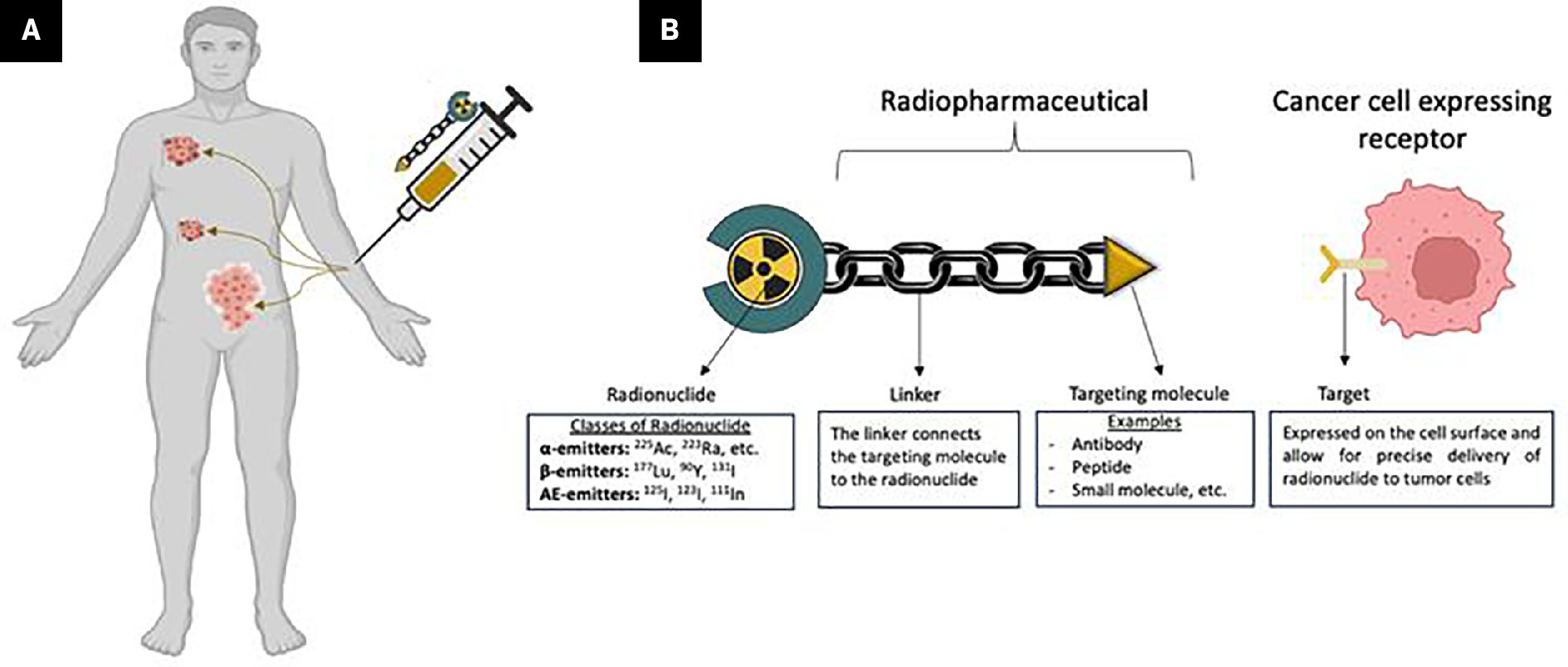
Targeting Molecules
In RPT, antibodies, peptides, or small molecules are designed to bind selectively to tumor-specific receptors or antigens, ensuring precise delivery of radiation to cancer cells while sparing healthy tissues.25
Antibodies
Their high specificity and potentially strong binding affinity make antibodies ideal for targeting tumor-associated antigens and delivering radiation to cancer cells.26,27 Effective antibodies target antigens that are highly expressed on tumors but minimally expressed or absent in healthy tissue. However, antibody size can limit tumor microenvironment (TME) penetration28 and prolong circulation,29 increasing off-target toxicities. Smaller monoclonal antibody (mAb) fragments like single-chain variable fragments partially retain target binding capacity while improving TME penetration. The US Food and Drug Administration/European Medical Agency (FDA/EMA)-approved examples of antibody-based radiopharmaceuticals include Zevalin ([90Y]Y-ibritumomab tiuxetan)30 and Bexxar ([131I]I-tositumomab),31 which target the CD20 protein on the surface of B-cells expressed by non-Hodgkin lymphoma.
Peptides
Peptides are versatile for RPT owing to rapid TME penetration, high uptake, and quick clearance from nontarget tissues, offering optimized pharmacokinetics. Their relatively higher stability enables chemical modifications and radiolabeling, making them versatile agents in nuclear medicine. A notable example of a peptide-based FDA/EMA-approved radiopharmaceutical is Lutathera ([177Lu]Lu-DOTA-TATE), indicated for the treatment of somatostatin receptor 2-positive gastroenteropancreatic neuroendocrine tumors.32,33
Small Molecules
Small molecule-based radioligands offer advantages as radiopharmaceuticals due to their efficient TME penetration and rapid clearance from systemic circulation, reducing off-target effects and toxicity. Although less specific than antibodies or peptides, small molecules effectively target cancer-associated antigens, such as the prostate-specific membrane antigen (PSMA) in prostate cancer. A notable example is the FDA-approved [177Lu]Lu-PSMA-167 (Pluvicto) for mCRPC,34 showcasing radioligand therapy’s potential in precision oncology.
Each targeting molecule in RPT offers a unique balance of strengths and limitations, with selection guided by tumor traits, precision, clearance, and off-target risks. This enables personalized and effective cancer therapy.
Radionuclides
A wide range of radionuclides is available for RPT, and selecting the appropriate one is crucial, as it directly influences treatment safety and efficacy. This choice is guided by factors such as physical half-life, availability, cost, radiochemical methods, and radiation properties, including energy level, type (α, β, or Auger electrons), linear energy transfer (LET), and penetration range (Figure 2 and Table 1).
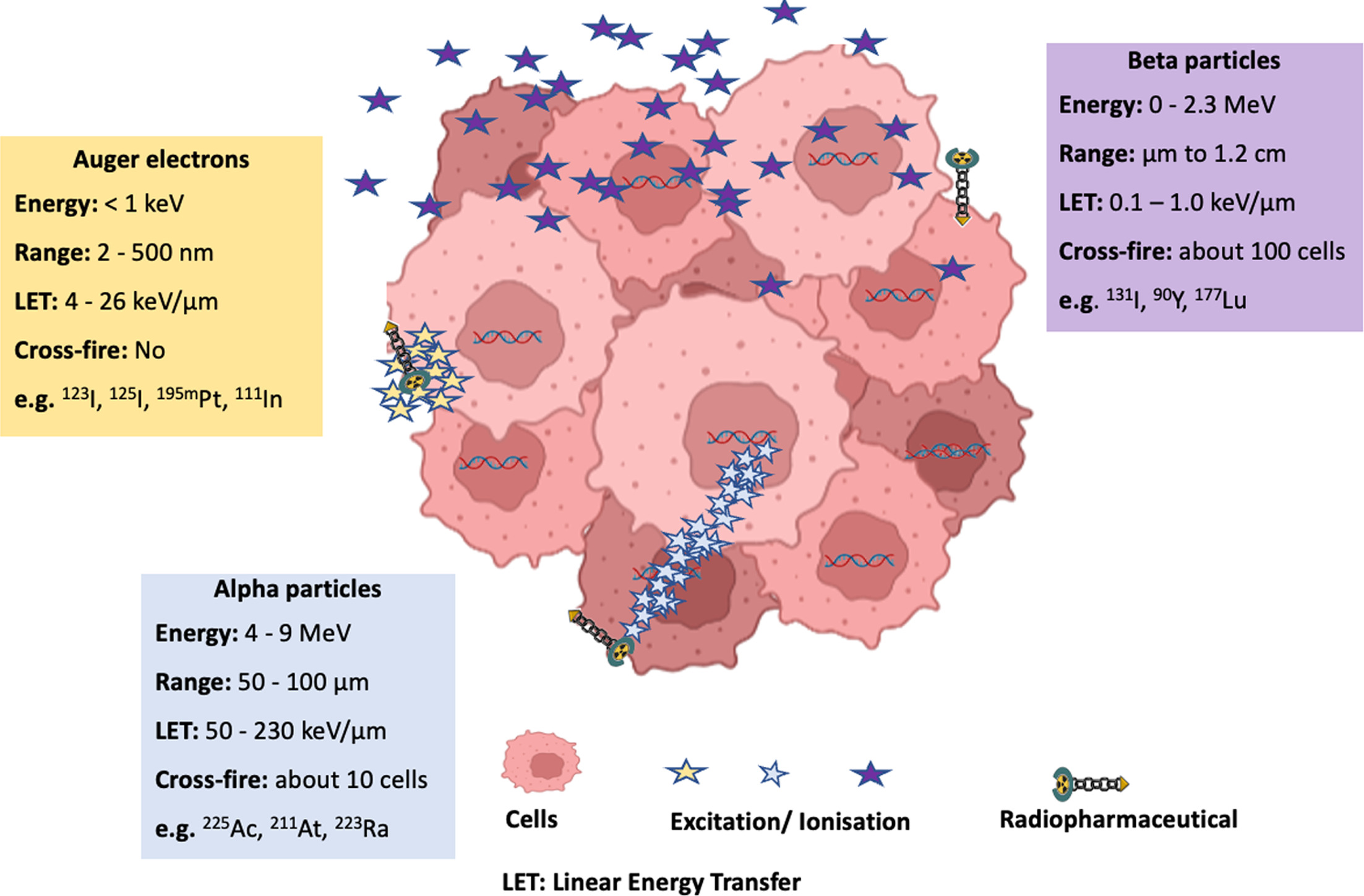
| Particle Emitted | Energy | Range in Tissue | LET (keV/µm) | Key DNA Damage Characteristics | Example of Radionuclides |
|---|---|---|---|---|---|
| β-particles | 0-2.3 MeV | μm to1.2 cm | 0.1-1.0 | Most single-strand breaks and some double-strand breaks but is easily repairable. (lower lethal damage efficiency) | 131I, 90Y, 177Lu |
| α-particles | 5-9 MeV | 50-100 µm | 50-230 | Mostly clustered double-strand breaks, making them complex and difficult to repair. (higher lethal damage efficiency) | 225Ac, 211At, 223Ra |
| Auger and conversion electrons | <1 keV | <1 µm | 4-26 | Mix of clustered double-strand breaks and single-strand breaks (lethality dependent on nuclear DNA proximity) | 123I, 125I, 195mPt, 111In |
Physical Half-Life
The time needed for half of an RPT’s radioactive atoms to decay is critical. The half-life must be amenable to the radiolabelling process, the distribution logistics of the agent, and the targeting molecule’s pharmacokinetics. While radionuclides with a short half-life, measured in hours, are preferred for imaging application, RPTs featuring short T1/2 radionuclides may lead to a significant decay before the radiopharmaceutical reaches the TME, thus reducing treatment efficacy. Conversely, a long half-life can increase radiation exposure to healthy tissue, thus increasing treatment-related side effects. Ideally, RPT radionuclides should have a half-life of 1 to 7 days for optimal balance.35
Linear Energy Transfer
The linear energy deposited by ionizing radiation per unit distance in tissue (keV/µm) significantly influences its biological effect. High LET of radiation (eg, α-particles, 50-230 keV/µm) induces dense clusters of double-strand DNA breaks (DSBs), causing irreparable DNA damage and high cytotoxicity. Intermediate LET radiation (eg, Auger electrons, 4-26 keV/µm) generates localized single-strand DNA breaks (SSBs) and DSBs, with cytotoxicity dependent on nuclear proximity due to limited penetration. Low LET radiation (eg, β-particles, 0.2 keV/µm) primarily induces SSBs and indirect damage via free radicals, which are often repairable, though clustered SSBs may result in DSBs.36,37 Radionuclides used in RPT are classified into 3 main categories based on their radiation type: β-particle emitters, α-particle emitters, and Auger/conversion electrons emitters.
β-particle emitters such as lutetium-177 (177Lu), yttrium-90 (90Y), and iodine-131 (131I), with a low LET (~0.2 keV/µm) and tissue penetration up to 12 mm, have been widely used in RPT. Owing to their deeper penetration range (several millimeters), low-LET β⁻-emitters can effectively treat heterogeneous (target expression) tumors,38,39 resulting in more effective tumor coverage,40,41 but may have lower lethal damage efficiency per unit dose.
α-particle emitters such as radium-223 (223Ra) and actinium-225 (225Ac) deliver potent therapy with high LET (50-230 keV/µm) and a short tissue range (50-100 µm); thus, they are ideal for treating micrometastases.39,40,42 Their high LET causes dense clusters of DSBs, which are difficult to repair,39 making them highly cytotoxic.43
Auger/conversion electron emitters such as iodine-123 (123I), iodine-125 (125I), and indium-111 (111In) have a very short tissue range (< 1 µm), making them effective near critical cell structures like nuclear DNA, and a medium-to-high LET (4-26 keV/µm) inducing a mix of SSBs and DSBs.16,39,42
Many β-particle and Auger emitters also emit γ rays, enabling their dual use for therapy and imaging.44 For example, γ emissions from 177Lu allow real-time visualization of radiopharmaceutical distribution, ensuring accurate targeting and dose optimization.45,46
Overall, radionuclide selection for RPT depends on properties like half-life, LET, radiation type, and tissue penetration. β-emitters appear to be better suited for larger tumors, while α-emitters target micrometastases with high cytotoxicity, and Auger emitters provide precise, localized radiation (< 1 µm) near critical structures like nuclear DNA.
Immunomodulatory Effects of RPT and Rationale for Combining RPT With ICI
The efficacy of RPT extends beyond direct cytotoxicity as it induces significant pro-inflammatory immune responses.47,48 Ionizing radiation enhances tumor immunogenicity, modulates the TME, and promotes innate and adaptive immunity.47,48 Irradiated tumor cells release DAMPs49 and express immunomodulatory molecules, recruiting antigen-presenting cells to activate T cells and drive systemic antitumor immunity.50 Potluri et al showed that [90Y]Y-NM600 modified the TME by increasing CD8+ T cell infiltration and PD-L1 expression on myeloid cells.51 In a murine study, Hernandez et al observed a reduction in immunosuppressive regulatory T cells and a notable increase in activated CD8+ T cells in EL4 murine tumors treated with [90Y]Y-NM600 compared with controls.52 Furthermore, upon rechallenging [90Y]Y-NM600-treated complete responders with EL4 cells, none developed tumors,52 suggesting the induction of a tumor-specific memory in RPT-treated mice. Emerging preclinical data suggest that targeted α-particle therapy (TAT) can also induce immunostimulatory effect.53 Lejeune et al further demonstrated that TAT triggers transcriptional and molecular signatures consistent with immunogenic cell death in preclinical syngeneic tumor models.54 Despite the reported immunomodulatory effects of RPT, its efficacy as a monotherapy often lacks durability, underscoring the compelling rationale for combining RPT with immunotherapy.55 Foundational studies have shown the synergism between RPT and immunotherapy, such as improved survival with [90Y]-anti-CEA (carcinoembryonic antigen) antibodies in combination with a CEA/TRICOM (TRICOM: 3 T-cell costimulatory molecules B7-1, ICAM-1, and LFA-3) vaccine in colon cancer models.56 This combination represents a promising strategy for achieving durable tumor control; thus, it may pave the way for enhancing patient outcomes through synergistic treatment strategies.
Clinical Trials Combining RPT With ICI
Building on preclinical evidence, several clinical trials have been initiated to evaluate the safety and efficacy of RPT-ICI combinations across cancers. Key outcomes are discussed here, underscoring the potential of these combination therapies to advance clinical treatment paradigms. Table 2 concisely summarizes these clinical trials, categorized by disease type for clarity.
| Disease | Trial | Phase | Disease Status | Target | RPT | ICI | Combination Sequence | Trial Status/Result | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Prostate cancer | NCT02814669 | Ib | mCRPC | Bone metastases | [223Ra]Ra: 55 kBq/kg (IV) every 28 days, 6 administrations | Atezolizumab: 840 mg (IV) every 14 days | Concurrent or staggered | Combination: greater toxicity | 57 |
| NCT03093428 | II | mCRPC | Bone metastases | [223Ra]Ra: every 4 weeks at a predetermined dose (IV) | Pembrolizumab: every 3 weeks at a predetermined dose (IV) | Concurrent | No improved efficacy | 58 | |
| NCT03658447 (PRINCE) | I | mCRPC | PSMA | [177Lu]Lu-PSMA-617: 8.5 GBq (IV), every 6 weeks, up to 6 cycles | Pembrolizumab: 200 mg every 3 weeks (IV) | Concurrent | PSA-RR: 76% No safety concerns rPFS: 11.2 months PSA-PFS: 8.2 months OS: 17.8 months | 59 | |
| Lung cancer | NCT03325816 | I/II | Extensive stage SCLC | SSTR | [177Lu]Lu -DOTA0-Tyr3-Octreotate: 3.7 or 7.4 GBq (IV), every 8 weeks, 4 cycles | Nivolumab: 240 mg every 2 weeks (IV) | Concurrent | Combination well tolerated PR: 1 out 7 patients | 60 |
| NCT03996473 | I | Metastatic NSCLC | Bone metastases | [223Ra]Ra: 55 kBq/kg (IV), every 6 weeks, up to 6 cycles | Pembrolizumab: 200 mg every 3 weeks (IV) up to 35 doses | Concurrent | Study closed | ||
| Renal cancer (ccRCC) | NCT05663710 | Ib/II | Advanced ccRCC | CAIX | [177Lu]Lu-girentuximab: 1.48 GBq/m2 (IV), every 12 weeks, up to 3 cycles | Nivolumab (dose not available) Cabozantinib: given orally | Concurrent | Ongoing | 61 |
| NCT05239533 (STARLITE 2) | II | Advanced ccRCC | CAIX | [177Lu]Lu-girentuximab: 1.8 or 2.4 GBq/m2 (IV), every 12-14 weeks, up to 3 cycles | Nivolumab: 200 mg every 2 weeks | Concurrent | Ongoing | 62 | |
| Merkel cell cancer | NCT05583708 | II | Metastatic | SSTR | [177Lu]Lu-DOTATATE: 7.4 GBq (IV), every 2 months, up to 4 doses | Pembrolizumab: 400 mg every 6 weeks (IV) | Concurrent | Temporarily suspended | |
| NCT04261855 (GoTHAM) | Ib/II | Metastatic | SSTR | [177Lu]Lu -DOTATATE: 2 administrations separated by 8-10 weeks | Avelumab: 10 mg/kg every 2 weeks for 24 months (IV) | Concurrent | Ongoing | ||
| Thyroid cancer | NCT03215095 | I | Recurrent/metastatic | rhTSH | [131I]I : 100 mCi | Durvalumab: 1500 mg IV every 4 weeks | Concurrent | Active, not recruiting | |
| Refractory neuroblastoma | NCT02914405 (MiNivAN) | I | Relapsed or refractory High risk | Norepinephrine transporter | [131I]I-meta-iodobenzylguanidine | Nivolumab: 3 mg/kg Dinutuximab (anti-GD2 monoclonal antibody): 50 or 100 mg/m2 | Concurrent | Recruiting | |
| NETs with liver metastases | NCT03457948 | II | Metastatic | SSTR | [177Lu]Lu-DOTA0-Tyr3-Octreotate | Pembrolizumab | Concurrent | Recruiting |
Prostate Cancer
A phase Ib study (NCT02814669) investigated the combination of [223Ra]RaCl₂ with atezolizumab in mCRPC patients with bone, lymph node, or visceral metastases. This combination resulted in greater toxicity than either agent alone and failed to show clinical benefit.57 Among the grade 3/4 adverse events, 34.1% were attributed to atezolizumab, while 27.3% were associated with [223Ra]RaCl₂.
A randomized phase II study (NCT03093428) evaluated [223Ra]RaCl₂ with pembrolizumab in patients with mCRPC. A recent report showed a median progression-free survival (PFS) of 6.1 months for [223Ra]RaCl₂ + pembrolizumab versus 5.7 months for [223Ra]RaCl₂ alone and a median overall survival (OS) of 16.9 months versus 16.0 months, respectively.58 While the combination was well tolerated with no unexpected toxicity, it did not demonstrate improved efficacy.
PRINCE (NCT03658447), a phase I clinical trial, evaluated the safety and efficacy of [177Lu]Lu-PSMA-617 in combination with pembrolizumab in patients with mCRPC. The prostate-specific antigen response rate (PSA-RR) was 76% compared with 46% with [177Lu]Lu-PSMA-617 alone. The median radiographic PFS, PSA-PFS, and OS were 11.2 months, 8.2 months, and 17.8 months, respectively.59 No additional safety concerns were identified with the addition of pembrolizumab, confirming the favorable safety profile of this combination.
Lung Cancer
Advanced lung cancer has also been the focus of clinical trials exploring the combination of RPT with immunotherapy. A phase I/II trial (NCT03325816) investigating nivolumab with Lutathera in patients with extensive-stage small cell lung cancer (SCLC) demonstrated a tolerable toxicity profile. Lutathera, a β-emitting [177Lu]Lu-labeled somatostatin analog approved for GEP-NETs,63 targets somatostatin receptor-expressing cells. The combination therapy was well tolerated. Furthermore, 1 out of 7 patients achieved a partial response (PR), while 2 with pulmonary atypical carcinoid maintained stable disease (SD) for 6 months. Notably, the patient with PR exhibited the highest tumor uptake of ⁶⁸Ga-DOTATATE on PET/CT, underscoring the potential of this approach.60
A phase I study (NCT03996473) sought to evaluate the safety and efficacy of combining [223Ra]RaCl2 with pembrolizumab in metastatic non-SCLC. The trial included patients who were either treatment-naïve for advanced disease or had progressed after prior PD-1/PD-L1 checkpoint blockade. The primary objectives were assessing tumor shrinkage, duration, and treatment safety. However, the study was closed early due to insufficient accrual.
Renal Cancer
Clear cell renal cell carcinoma (ccRCC) is characterized by carbonic anhydrase IX expression resulting from von Hippel-Lindau loss, representing a compelling target for RPT-based therapies. The integration of RPT with immunotherapy in advanced ccRCC is gaining momentum, with 2 phase II clinical trials currently underway (NCT05239533; NCT05663710). These trials aim to evaluate the safety and efficacy of combining [177Lu]Lu-girentuximab with nivolumab as a novel treatment strategy for advanced ccRCC.61,62
Merkel Cell Carcinoma
Two case reports underscore the significant therapeutic potential of combining RPT with ICI in metastatic Merkel cell carcinoma (MCC). These cases involved patients who had progressed on first-line avelumab or second-line therapies combining ipilimumab, nivolumab, and EBRT.64,65 While up to half of patients with MCC either may not respond to or may develop resistance to ICIs,65 the frequent expression of somatostatin receptors in MCC makes it a suitable target for [177Lu]Lu-DOTATATE. In one case, a patient with extensive MCC metastases treated with [177Lu]Lu-DOTATATE and anti-PD-L1 therapy demonstrated a rapid response, achieving a near-complete response within 1 month.64 Another patient receiving [177Lu]Lu-DOTATOC, along with ipilimumab and nivolumab, achieved and sustained a PR for 5 months.65 Clinical trials (NCT05583708; NCT04261855) have been initiated to evaluate [177Lu]Lu-DOTATATE combined with nivolumab or pembrolizumab in patients with metastatic MCC.
Other ongoing clinical trials are exploring RPT with ICI, including radioiodine (131I) with durvalumab (NCT03215095) for thyroid cancer, 131I-MIBG with nivolumab and dinutuximab (anti-GD2 monoclonal antibody) for refractory neuroblastoma (NCT02914405), and177Lu-DOTA0-Tyr3-Octreotate with pembrolizumab (NCT03457948) for NETs with liver metastases.
Challenges and Future Perspectives
Combining RPT with immunotherapy is a promising therapeutic option for metastatic cancers. With its targeted radiation delivery and ability to modulate the TME, RPT can complement the systemic antitumor effects of immunotherapy. Preclinical studies highlight the potential of RPT and ICI combination,47-54 but robust clinical evidence remains limited. Nevertheless, few studies have shown promising results, including case reports with [177Lu]Lu-DOTATATE or [177Lu]Lu-DOTATOC plus ICI in MCC metastases,64,65 and the phase I PRINCE trial with [177Lu]Lu-PSMA-617 in combination with ICI in mCRPC.59 Beyond these studies, we are awaiting results from ongoing clinical trials (Table 2). Nevertheless, challenges persist, including increased toxicities57 with immune-related events and radiation-induced toxicities. The immunosuppressive TME, influenced by regulatory T cells and immune checkpoint expression, may further dampen treatment efficacy. Variability in patient responses, driven by tumor heterogeneity, highlights the need for predictive biomarkers for optimal patient selection. Economic and logistical barriers also hinder implementation.66-70 The production and administration of RPT require specialized infrastructure and expertise, while its high costs necessitate cost-benefit analyses for integration into clinical practice. Future research should optimize trial designs for sequencing, dosing, and timing of RPT-ICI combinations. Advances in imaging, dosimetry, and collaboration among specialists, along with efforts to reduce costs and improve access, are key to transforming metastatic cancer treatment. Moreover, most trials do not clearly differentiate whether observed toxicities stem from immune-related effects or radiation exposure. Gaining a deeper understanding of the predominant mechanism, whether immune-mediated or radiation-induced, is essential for optimizing toxicity management and improving the safety profile of these combinations.
Conclusion
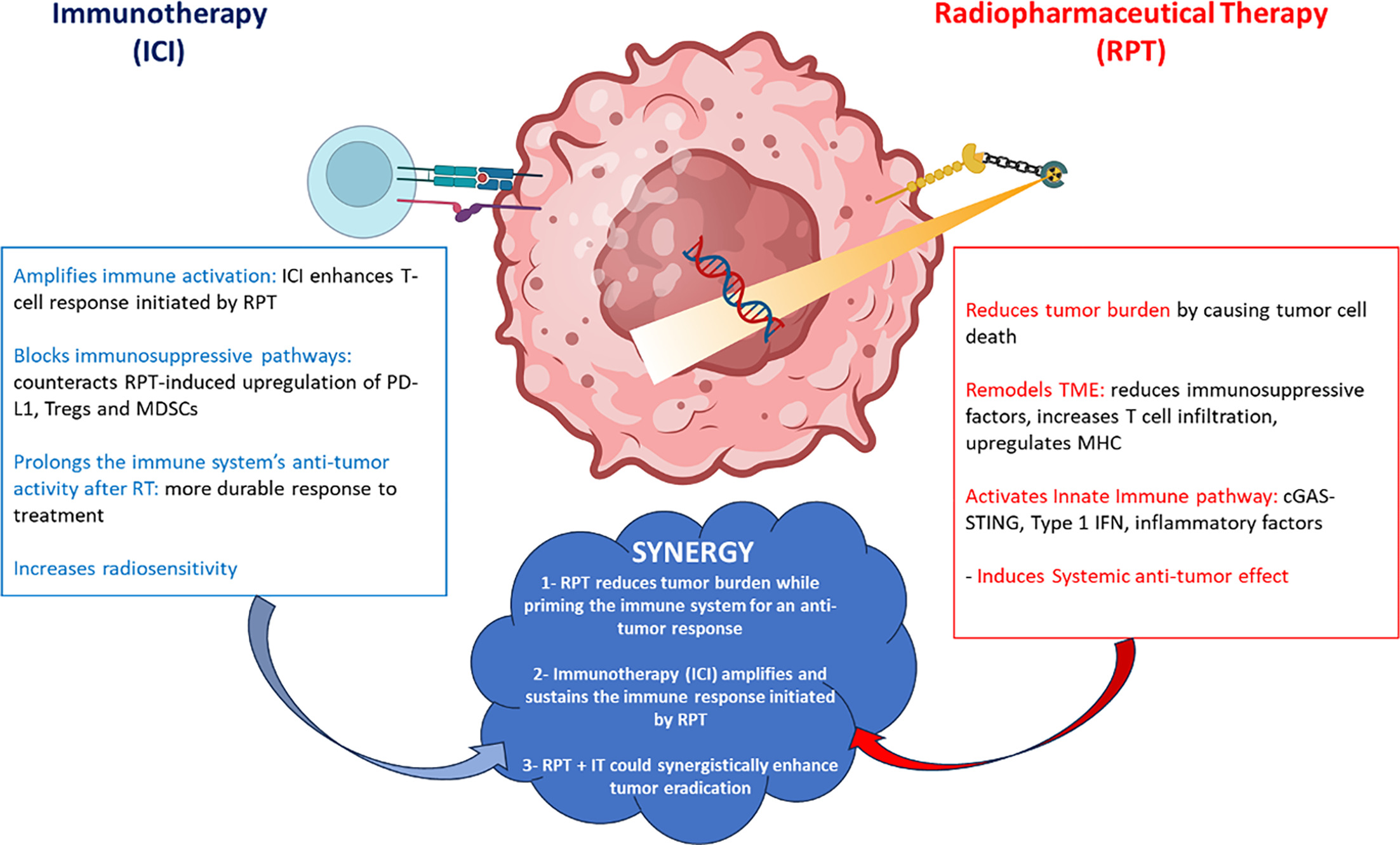
The combination of RPT and immunotherapy offers a transformative approach to metastatic cancer, overcoming current treatment limitations. As shown in Figure 3, RPT synergizes with immunotherapy, including ICIs, by reducing tumor burden, releasing neo-antigens, enhancing MHC-I expression, and modifying the TME, while immunotherapy amplifies and sustains these effects, countering immune evasion and optimizing tumor control, especially in “cold” tumors. Despite challenges such as toxicity and logistical barriers, advances in radiopharmaceutical design, immune modulation, and personalized biomarkers driven by interdisciplinary collaboration could redefine cancer care for advanced, treatment-resistant, and metastatic malignancies.
References
- Seyfried T, Huysentruyt L. On the origin of cancer metastasis. Crit Rev Oncog. 2013;18(1-2):43-73. doi:10.1615/critrevoncog.v18.i1-2.40.
- Spano D, Heck C, De Antonellis P, Christofori G, Zollo M. Molecular networks that regulate cancer metastasis. Semin Cancer Biol. 2012;22(3):234-249. doi:10.1016/j.semcancer.2012.03.006.
- Ganesh K, Massagué J. Targeting metastatic cancer. Nat Med. 2021;27(1):34-44. doi:10.1038/s41591-020-01195-4.
- Coleman R. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20 pt 2):6243s-6249s. doi:10.1158/1078-0432.CCR-06-0931.
- van Roij J, Brom L, Youssef-El Soud M, van de Poll-Franse L, Raijmakers N. Social consequences of advanced cancer in patients and their informal caregivers: a qualitative study. Support Care Cancer. 2019;27(4):1187-1195. doi:10.1007/s00520-018-4437-1.
- Costa L, Badia X, Chow E, Lipton A, Wardley A. Impact of skeletal complications on patients’ quality of life, mobility, and functional independence. Support Care Cancer. 2008;16(8):879-889. doi:10.1007/s00520-008-0418-0.
- Lambert A, Pattabiraman D, Weinberg R. Emerging biological principles of metastasis. Cell. 2017;168(4):670-691. doi:10.1016/j.cell.2016.11.037.
- Sgouros G, Bodei L, McDevitt M, Nedrow J. Radiopharmaceutical therapy in cancer: clinical advances and challenges. Nat Rev Drug Discov. 2020;19(9):589-608. doi:10.1038/s41573-020-0073-9.
- Salih S, Alkatheeri A, Alomaim W, Elliyanti A. Radiopharmaceutical treatments for cancer therapy, radionuclides characteristics, applications, and challenges. Molecules. 2022;27(16). doi:10.3390/molecules27165231.
- Murphy J, Hussey R, Nakahara W, Sturm E. Studies on X-ray effects: Vi. Effect of the cellular reaction induced by X-rays on cancer grafts. J Exp Med. 1921;33(3):299-313. doi:10.1084/jem.33.3.299.
- Storozynsky Q, Hitt M. The impact of radiation-induced DNA damage on cgas-STING-mediated immune responses to cancer. Int J Mol Sci. 2020;21(22). doi:10.3390/ijms21228877.
- Constanzo J, Faget J, Ursino C, Badie C, Pouget J. Radiation-induced immunity and toxicities: the versatility of the cgas-STING pathway. Front Immunol. 2021;12. doi:10.3389/fimmu.2021.680503.
- Dumortier J, Decullier E, Hilleret M. Adjuvant intraarterial lipiodol orI-lipiodol after curative treatment of hepatocellular carcinoma: a prospective randomized trial 131. J Nucl Med. 2014;55(6):877-883. doi:10.2967/jnumed.113.131367.
- Shea A, Idrissou M, Torres A. Immunological effects of radiopharmaceutical therapy. Front Nucl Med. 2024;4. doi:10.3389/fnume.2024.1331364.
- Brady D, O’Sullivan J, Prise K. What is the role of the bystander response in radionuclide therapies?. Front Oncol. 2013;3. doi:10.3389/fonc.2013.00215.
- Idrissou M, Pichard A, Tee B. Targeted radionuclide therapy using auger electron emitters: the quest for the right vector and the right radionuclide. Pharmaceutics. 2021;13(7). doi:10.3390/pharmaceutics13070980.
- Gudkov S, Shilyagina N, Vodeneev V, Zvyagin A. Targeted radionuclide therapy of human tumors. Int J Mol Sci. 2015;17(1). doi:10.3390/ijms17010033.
- Capello A, Krenning E, Bernard B. In-labelled somatostatin analogues in a rat tumour model: somatostatin receptor status and effects of peptide receptor radionuclide therapy 111. Eur J Nucl Med Mol Imaging. 2005;32(11):1288-1295. doi:10.1007/s00259-005-1877-x.
- Poeppel T, Handkiewicz-Junak D, Andreeff M. EANM guideline for radionuclide therapy with radium-223 of metastatic castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging. 2018;45(5):824-845. doi:10.1007/s00259-017-3900-4.
- Hennrich U, Eder M. [Lu]LU-PSMA-617 (pluvictotm): the first FDA-approved radiotherapeutical for treatment of prostate cancer 177 TM. Pharmaceuticals. 2022;15(10):1292. doi:10.3390/ph15101292.
- Hennrich U, Kopka K. Lutathera: the first FDA- and EMA-approved radiopharmaceutical for peptide receptor radionuclide therapy ®. Pharmaceuticals. 2019;12(3). doi:10.3390/ph12030114.
- Wu Y, Song Y, Wang R, Wang T. Molecular mechanisms of tumor resistance to radiotherapy. Mol Cancer. 2023;22(1). doi:10.1186/s12943-023-01801-2.
- Gao Z, Zhao Q, Xu Y, Wang L. Improving the efficacy of combined radiotherapy and immunotherapy: focusing on the effects of radiosensitivity. Radiat Oncol. 2023;18(1). doi:10.1186/s13014-023-02278-5.
- Bellavia M, Patel R, Anderson C. Combined targeted radiopharmaceutical therapy and immune checkpoint blockade: from preclinical advances to the clinic. J Nucl Med. 2022;63(11):1636-1641. doi:10.2967/jnumed.122.264373.
- Zhang T, Lei H, Chen X. Carrier systems of radiopharmaceuticals and the application in cancer therapy. Cell Death Discov. 2024;10(1). doi:10.1038/s41420-023-01778-3.
- Zhao Z, Ukidve A, Kim J, Mitragotri S. Targeting strategies for tissue-specific drug delivery. Cell. 2020;181(1):151-167. doi:10.1016/j.cell.2020.02.001.
- Almagro J, Kodangattil S. Novel Approaches and Strategies for Biologics, Vaccines and Cancer Therapies. 2015:147-170. doi:10.1016/B978-0-12-416603-5.00007-9.
- Cruz E, Kayser V. Monoclonal antibody therapy of solid tumors: clinical limitations and novel strategies to enhance treatment efficacy. Biologics. 2019;13:33-51. doi:10.2147/BTT.S166310.
- Beckman R, Weiner L, Davis H. Antibody constructs in cancer therapy: protein engineering strategies to improve exposure in solid tumors. Cancer. 2007;109(2):170-179. doi:10.1002/cncr.22402.
- Grillo-López A. Zevalin: the first radioimmunotherapy approved for the treatment of lymphoma. Expert Rev Anticancer Ther. 2002;2(5):485-493. doi:10.1586/14737140.2.5.485.
- Friedberg J, Fisher R. Iodine-131 tositumomab (Bexxar®): radioimmunoconjugate therapy for indolent and transformed B-cell non-hodgkin’s lymphoma. Expert Rev Anticancer Ther. 2004;4(1):18-26. doi:10.1586/14737140.4.1.18.
- FDA approves lutetium Lu 177 dotatate for treatment of GEP-NETS. FDA. 2024.
- European Medicines Agency (EMA). 2018.
- Fallah J, Agrawal S, Gittleman H. FDA approval summary: lutetium lu 177 vipivotide tetraxetan for patients with metastatic castration-resistant prostate cancer. Clin Cancer Res. 2023;29(9):1651-1657. doi:10.1158/1078-0432.CCR-22-2875.
- Qaim S. Therapeutic radionuclides and nuclear data. Radiochim Acta. 2001;89(4-5):297-304. doi:10.1524/ract.2001.89.4-5.297.
- Howell R, Rao D, Hou D, Narra V, Sastry K. The question of relative biological effectiveness and quality factor for auger emitters incorporated into proliferating mammalian cells. Radiat Res. 1991;128(3):282-292. doi:10.2307/3578051.
- Rao D, Howell R, Narra V, Govelitz G, Sastry K. IN-vivo radiotoxicity of DNA-incorporated 125I compared with that of densely ionising alpha-particles. Lancet. 1989;334(8664):650-653. doi:10.1016/S0140-6736(89)90896-9.
- Marcu L, Bezak E, Allen B. Global comparison of targeted alpha vs targeted beta therapy for cancer: in vitro, in vivo and clinical trials. Crit Rev Oncol Hematol. 2018;123:7-20. doi:10.1016/j.critrevonc.2018.01.001.
- Pouget J, Navarro-Teulon I, Bardiès M. Clinical radioimmunotherapy—the role of radiobiology. Nat Rev Clin Oncol. 2011;8(12):720-734. doi:10.1038/nrclinonc.2011.160.
- Dekempeneer Y, Keyaerts M, Krasniqi A. Targeted alpha therapy using short-lived alpha-particles and the promise of nanobodies as targeting vehicle. Expert Opin Biol Ther. 2016;16(8):1035-1047. doi:10.1080/14712598.2016.1185412.
- Czerwińska M, Bilewicz A, Kruszewski M, Wegierek-Ciuk A, Lankoff A. Targeted radionuclide therapy of prostate cancer-from basic research to clinical perspectives. Molecules. 2020;25(7). doi:10.3390/molecules25071743.
- Poty S, Francesconi L, McDevitt M, Morris M, Lewis J. α-emitters for radiotherapy: from basic radiochemistry to clinical studies-part 1. J Nucl Med. 2018;59(6):878-884. doi:10.2967/jnumed.116.186338.
- Sgouros G, Roeske J, McDevitt M. MIRD pamphlet no. 22 (abridged): radiobiology and dosimetry of alpha-particle emitters for targeted radionuclide therapy. J Nucl Med. 2010;51(2):311-328. doi:10.2967/jnumed.108.058651.
- Stokke C, Kvassheim M, Blakkisrud J. Radionuclides for targeted therapy: physical properties. Molecules. 2022;27(17). doi:10.3390/molecules27175429.
- Othman M, Mitry N, Lewington V, Blower P, Terry S. Re-assessing gallium-67 as a therapeutic radionuclide. Nucl Med Biol. 2017;46:12-18. doi:10.1016/j.nucmedbio.2016.10.008.
- Pirovano G, Jannetti S, Carter L. Targeted brain tumor radiotherapy using an auger emitter. Clin Cancer Res. 2020;26(12):2871-2881. doi:10.1158/1078-0432.CCR-19-2440.
- Gaipl U, Multhoff G, Scheithauer H. Kill and spread the word: stimulation of antitumor immune responses in the context of radiotherapy. Immunotherapy. 2014;6(5):597-610. doi:10.2217/imt.14.38.
- Filatenkov A, Baker J, Mueller A. Ablative tumor radiation can change the tumor immune cell microenvironment to induce durable complete remissions. Clin Cancer Res. 2015;21(16):3727-3739. doi:10.1158/1078-0432.CCR-14-2824.
- Bianchi M. DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol. 2007;81(1):1-5. doi:10.1189/jlb.0306164.
- Takeshima T, Chamoto K, Wakita D. Local radiation therapy inhibits tumor growth through the generation of tumor-specific CTL: its potentiation by combination with th1 cell therapy. Cancer Res. 2010;70(7):2697-2706. doi:10.1158/0008-5472.CAN-09-2982.
- Potluri H, Ferreira C, Grudzinski J. Antitumor efficacy of 90Y-NM600 targeted radionuclide therapy and PD-1 blockade is limited by regulatory T cells in murine prostate tumors. J Immunother Cancer. 2022;10(8). doi:10.1136/jitc-2022-005060.
- Hernandez R, Walker K, Grudzinski J. 90Y-NM600 targeted radionuclide therapy induces immunologic memory in syngeneic models of T-cell non-hodgkin’s lymphoma. Commun Biol. 2019;2(1). doi:10.1038/s42003-019-0327-4.
- Perrin J, Capitao M, Allard M. Targeted alpha particle therapy remodels the tumor microenvironment and improves efficacy of immunotherapy. Int J Radiat Oncol. 2022;112(3):790-801. doi:10.1016/j.ijrobp.2021.10.013.
- Lejeune P, Cruciani V, Berg-Larsen A. Immunostimulatory effects of targeted thorium-227 conjugates as single agent and in combination with anti-PD-L1 therapy. J Immunother Cancer. 2021;9(10). doi:10.1136/jitc-2021-002387.
- Zukotynski K, Jadvar H, Capala J, Fahey F. Targeted radionuclide therapy: practical applications and future prospects: supplementary issue: biomarkers and their essential role in the development of personalised therapies. Biomark Cancer. 2016;8s2. doi:10.4137/BIC.S31804.
- Chakraborty M, Gelbard A, Carrasquillo J. Use of radiolabeled monoclonal antibody to enhance vaccine-mediated antitumor effects. Cancer Immunol Immunother. 2008;57(8):1173-1183. doi:10.1007/s00262-008-0449-x.
- Fong L, Morris M, Sartor O. A phase Ib study of atezolizumab with radium-223 dichloride in men with metastatic castration-resistant prostate cancer. Clin Cancer Res. 2021;27(17):4746-4756. doi:10.1158/1078-0432.CCR-21-0063.
- Choudhury A, Kwak L, Cheung A. Randomized phase II study evaluating the addition of pembrolizumab to radium-223 in metastatic castration-resistant prostate cancer. Cancer Immunol Res. 2024;12(6):704-718. doi:10.1158/2326-6066.CIR-22-0306.
- Sandhu S, Joshua A, Emmett L. PRINCE: phase I trial oflu-PSMA-617 in combination with pembrolizumab in patients with Metastatic Castration-Resistant Prostate Cancer (mcrpc) 177. JCO. 2022;40(16_suppl):5017-5017. doi:10.1200/JCO.2022.40.16_suppl.5017.
- Kim C, Liu S, Subramaniam D. Phase I study of the 177Lu-DOTA0-Tyr3-Octreotate (lutathera) in combination with nivolumab in patients with neuroendocrine tumors of the lung. J Immunother Cancer. 2020;8(2). doi:10.1136/jitc-2020-000980.
- Feldman D, Motzer R, Knezevic A. STARLITE 2: phase 2 study of nivolumab pluslutetium-labeled anti-carbonic anhydrase IX (CAIX) monoclonal antibody girentuximab (lu-girentuximab) in patients (pts) with advanced clear cell renal cell carcinoma (ccrcc) 177 177. JCO. 2022;40(16_suppl). doi:10.1200/JCO.2022.40.16_suppl.TPS4603.
- Jonasch E, Hasanov E, Flynt L. Phase 1b/2 study of combinationlu girentuximab plus cabozantinib and nivolumab in treatment naive patients with advanced clear cell RCC 177. JCO. 2024;42(16_suppl). doi:10.1200/JCO.2024.42.16_suppl.TPS4610.
- Strosberg J, Wolin E, Chasen B. NETTER-1 phase III: progression-free survival, radiographic response, and preliminary overall survival results in patients with midgut neuroendocrine tumors treated with 177-LU-dotatate. JCO. 2016;34(4_suppl):194-194. doi:10.1200/jco.2016.34.4_suppl.194.
- Kasi P, Sharma A, Jain M. Expanding the indication for novel theranostic 177Lu-dotatate peptide receptor radionuclide therapy: proof-of-concept of PRRT in merkel cell cancer. Case Rep Oncol. 2019;12(1):98-103. doi:10.1159/000496335.
- Ferdinandus J, Fendler W, Lueckerath K. Response to combined peptide receptor radionuclide therapy and checkpoint immunotherapy with ipilimumab plus nivolumab in metastatic merkel cell carcinoma. J Nucl Med. 2022;63(3):396-398. doi:10.2967/jnumed.121.262344.
- Decroisette C, Monnet I, Berard H. Epidemiology and treatment costs of bone metastases from lung cancer: A french prospective, observational, multicenter study (GFPC 0601). J Thorac Oncol. 2011;6(3):576-582. doi:10.1097/JTO.0b013e318206a1e3.
- Félix J, Andreozzi V, Soares M. Hospital resource utilization and treatment cost of skeletal-related events in patients with metastatic breast or prostate cancer: estimation for the portuguese national health system. Value Health. 2011;14(4):499-505. doi:10.1016/j.jval.2010.11.014.
- Hoefeler H, Duran I, Hechmati G. Health resource utilization associated with skeletal-related events in patients with bone metastases: results from a multinational retrospective - prospective observational study - a cohort from 4 european countries. J Bone Oncol. 2014;3(2):40-48. doi:10.1016/j.jbo.2014.04.001.
- Lüftner D, Lorusso V, Duran I. Health resource utilization associated with skeletal-related events in patients with advanced breast cancer: results from a prospective, multinational observational study. Springerplus. 2014;3(1). doi:10.1186/2193-1801-3-328.
- Pockett R, Castellano D, McEwan P. The hospital burden of disease associated with bone metastases and skeletal-related events in patients with breast cancer, lung cancer, or prostate cancer in spain. Eur J Cancer Care. 2010;19(6):755-760. doi:10.1111/j.1365-2354.2009.01135.x.
Disclosures
RH: consulting fees from MonoPar Inc and Archeus technologies Inc.; Chief Technology Officer for Archeus Technologies Inc and received stocks from Archeus. All other authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere.
Citation
. Clinical Evidence of Combining Radiopharmaceutical Therapy With Immune Checkpoint Inhibitors. Applied Radiation Oncology. 2025;14(1). doi:10.37549/ARO-D-24-00039.