Accuray Receives FDA Clearance for VitalHold Breast Package on the Radixact System
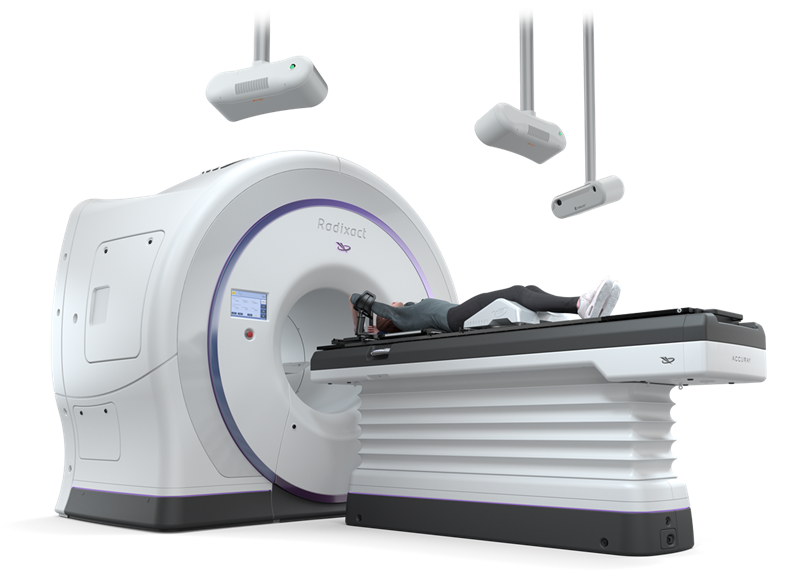
VitalHold facilitates deep inspiration breath hold (DIBH) treatments using the Radixact System’s fixed beam angle delivery mode (TomoDirect) and a fully integrated and automated surface-guided radiation therapy (SGRT) solution, featuring the Catalyst+ HD system from C-RAD. With the addition of this new functionality, the Radixact System offers a comprehensive tool set for the treatment of breast cancer.
DIBH treatments are often used to help minimize radiation dose to organs at risk (OARs) and reduce associated complications later in life. During treatment, a patient takes a deep breath which moves the heart away from the chest wall and the targeted tumor. The SGRT tracking system continuously monitors a patient’s breathing and position with cameras to confirm the patient is appropriately positioned to deliver radiation. SGRT is designed to enable medical care teams to effectively position the patient and confidently monitor the accuracy of that positioning throughout treatment.
“Our goal in introducing VitalHold for the Radixact System is to provide clinicians with additional choice and flexibility when treating the full spectrum of breast cancer cases they see daily in their practice. Now, Accuray can offer our customers the widest range of radiation treatment delivery techniques in one device, the Radixact System. This industry-leading approach demonstrates how our relentless focus on innovation has enabled Accuray to make an already great system even better, providing clinicians with tools that help ensure precise, truly personalized treatments are achievable for more patients,” said Suzanne Winter, President and CEO of Accuray.
“C-RAD is incredibly proud of our collaboration with Accuray. Together, we have created a seamless integration between our systems to ensure the highest level of accuracy and efficiency for the treatment of breast cancer,” said Cecilia de Leeuw, CEO and President of C-RAD AB.
“VitalHold is a powerful application of C-RAD’s proven SGRT technology — and one that’s sure to touch the lives of countless breast cancer patients.”
The CT linac based Radixact System offers multiple radiation delivery options to tailor each patient’s treatment based on their specific needs. The system’s extensive capabilities make it possible for clinicians to treat breast cases in as little as 2 minutes. With TomoHelical mode, the Radixact System continuously delivers radiation beams from 360 degrees around the patient, enabling the treatment of complex cases, such as cancer in the breast and surrounding lymph nodes. This approach also provides a valuable option for patients who are unable to hold their breath, which is key to undergoing DIBH treatments. TomoDirect mode facilitates delivery of radiation from specific fixed angles.
Due to its unique radiation delivery approaches, the Radixact System offers advantages in the treatment of breast cancer, including the ability to deliver precise radiation dose to any tumor bed, during every treatment, while minimizing dose to nearby healthy organs and tissues. With the Radixact System, radiation therapy may be an option for treating breast cancer at almost every stage and can be used after surgery, chemotherapy and other medications.