Whole Abdominal Radiation Therapy for Chemo- Refractory Adult Granulosa Cell Tumor of the Ovary: A Case Report
Images

Abstract
Granulosa cell tumors (GCTs) emerging from sex-cord stromal cells of the ovary represent less than 5% of all ovarian cancers. This report discusses a case of primary chemotherapy refractory adult GCT of the ovary with widespread abdomino-pelvic metastasis. Because GCTs are radiosensitive, whole abdominal radiation therapy (WART) is a useful treatment when surgical and systemic options have been exhausted. WART delivered with modern radiation techniques results in excellent clinical and radiological response rates with acceptable toxicity and the possibility of long-term disease control.
Keywords: Granulosa cell tumors, whole abdominal radiation therapy, ovarian cancer, volumetric- modulated arc therapy
Case Summary
A 43-year-old White woman was referred for consideration of whole abdominal radiation therapy (WART) in July 2019. Her history dates to May 2018 when the patient developed intermenstrual bleeding and intermittent abdominal pain. Initial imaging revealed a left ovarian cyst. She underwent a left salpingo-oophorectomy (LSO) with uterine dilation and curettage (D&C). At the time of surgery, left ovarian cyst rupture was noted. Pathology was consistent with adult type granulosa cell tumor (GCT). On immunohistochemical stains, the tumor cells were SF-1 positive, calretinin positive, and negative for epithelial membrane antigen (EMA) and synaptophysin, confirming the diagnosis. The patient then underwent a total laparoscopic hysterectomy, right salpingo-oophorectomy, bilateral pelvic and para-aortic lymphadenectomy, omentectomy, peritoneal biopsies, and cystoscopy in June 2018. Her final surgical staging was FIGO stage IIA. She then underwent 6 cycles of adjuvant carboplatin and paclitaxel chemotherapy, and her inhibin A and B levels were normal at completion of adjuvant therapy in November 2018.
In April 2019, rising inhibin levels prompted a computed tomography (CT) scan that revealed 1 pelvic, 2 hepatic surface and 2 peritoneal lesions. The hepatic lesion was biopsied and a relapse was confirmed. The multidisciplinary tumor board recommended systemic therapy with letrozole and bevacizumab for 3 months and then proceeding with maximal cytoreductive surgery (MCS). In October 2019, the patient underwent MCS. Her postoperative period was uneventful, with minimal residual disease (< 1 cm3) and normal inhibin A and B levels.
Letrozole and bevacizumab were re-initiated 8 weeks later. Unfortunately, small-volume disease progression was identified within 6 months of surgery and eventually the patient was switched to liposomal doxorubicin, followed by tamoxifen/megestrol, then paclitaxel, and finally etoposide/cisplatin. All of the aforementioned systemic agents were stopped prematurely due to disease progression while on therapy.
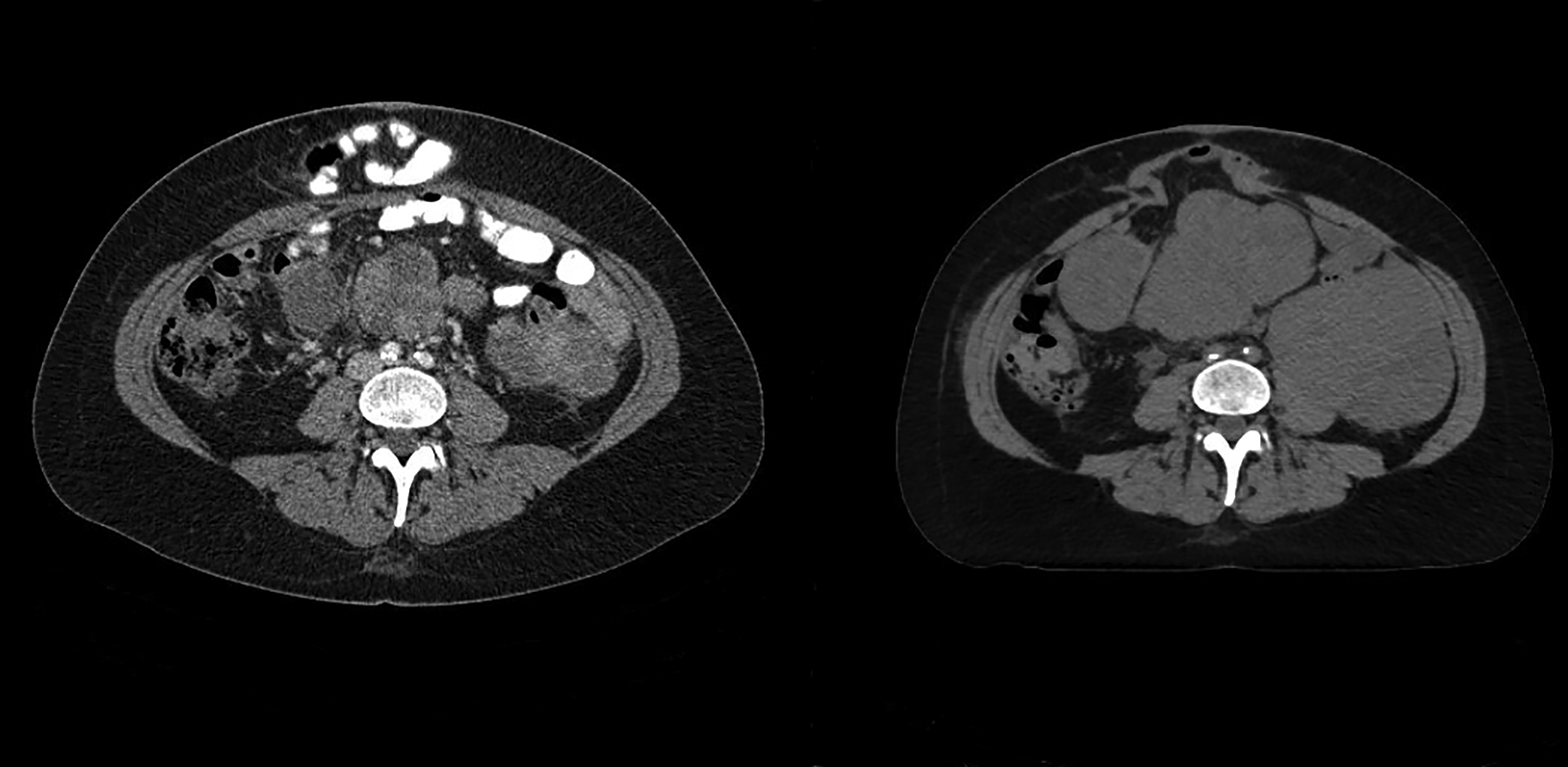
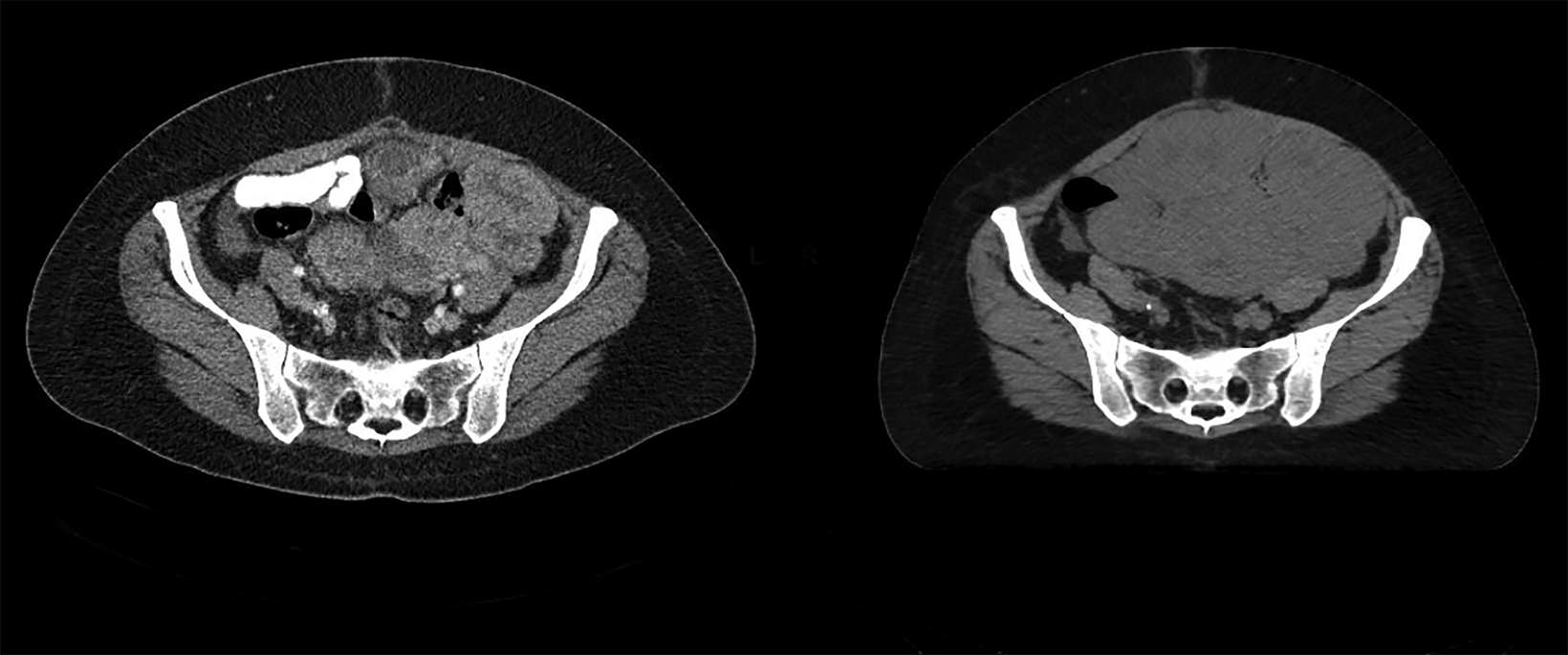
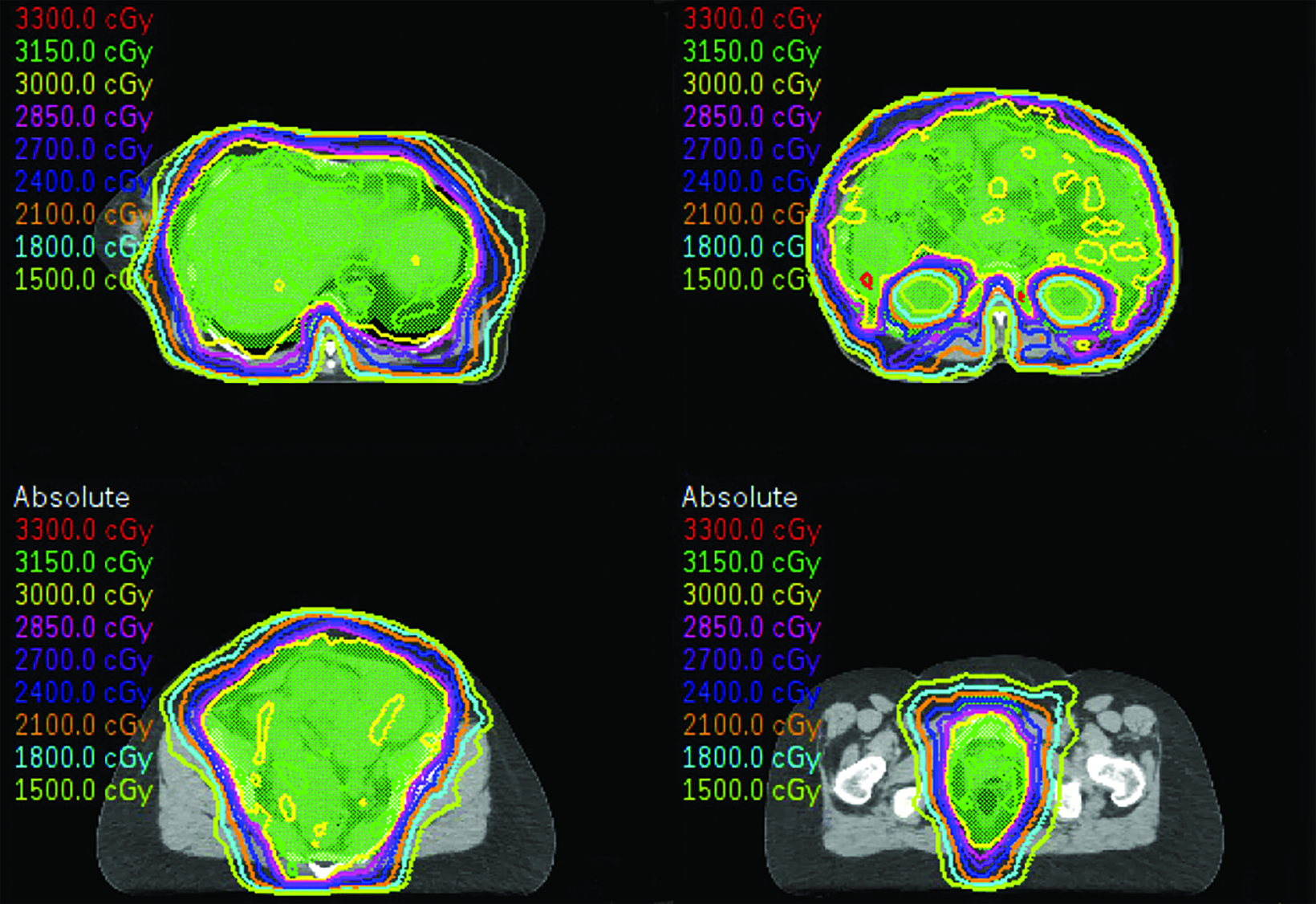
Although the patient was initially referred to us for radiation therapy in July 2019, it was our collective decision to exhaust all systemic options before embarking on whole abdominal radiation therapy (WART). In November 2021, the patient started WART utilizing multi-isocentric, image-guided, volumetric-modulated arc therapy (VMAT). At the time, her disease burden involved multiple hepatic surface and parenchymal metastasis, and multiple large soft-tissue masses throughout the abdomen and pelvis (average size of 8 cm and the largest measuring 11 cm). She received a total dose of 30 Gy in 20 fractions, over 4 weeks. The organs-at-risk dose constraints were as follows: kidney mean dose < 18 Gy and small bowel max point dose < 110%. The patient tolerated treatment well with acute RTOG grade-1 gastrointestinal side effects.1 At last follow-up, 6 weeks post WART, she had no residual acute side effects and had a Karnofsky Performance Status (KPS) of 90. CT scan confirmed good partial response (all lesions including index lesion ≥ 50% decrease) by RECIST criteria (Figures 1 A-C).2 Since her first relapse, she has been on 5 systemic agents and her disease was primary refractory to all agents. WART has been the only therapy modality able to induce a response.
Imaging Findings
A CT scan prior to WART showed multiple large hepatic, abdominal and pelvic masses, which had significantly reduced in size 6 weeks after radiation therapy (Figures 1 A-C). WART was planned using VMAT techniques to spare the renal parenchyma (Figure 2).
Diagnosis
Primary chemotherapy refractory adult GCT of the ovary with widespread abdomino-pelvic metastasis.
Discussion
GCTs arising from sex-cord stromal cells of the ovary are rare and represent under 5% of all ovarian cancers.3 They occur most commonly in women in the reproductive age group. They are characterized by a long natural history and have a tendency to relapse late.3 Based on clinical presentation and histological characteristics, GCTs are classified into juvenile and adult GCTs. The majority are adult GCTs and 5% are juvenile.4 Nearly all GCTs are secretory, producing estradiol, leading to precocious puberty in juvenile patients, and menstrual irregularities and virilization in adults.5 GCTs are also known to increase the risk of endometrial and breast cancers due to a hyperestrogenic state.6,7 Under microscopy, the adult GCTs appear round to oblong with scant cytoplasm and classic “coffee-bean” grooved nuclei. The cells arrange themselves in rosettes around a central cavity and this pattern is called “Call-Exner bodies,” which is pathognomonic for GCTs.
Somatic mutations in FOXL2 are identified in 97% of cases.8 On immunohistochemistry, they are commonly positive for inhibin, calretinin, CD56 and CD99.9
Surgery remains the mainstay for early stage GCTs and late-stage tumors if maximal debulking is feasible.10,11 Stage I disease has an excellent prognosis and no adjuvant therapy is recommended.3 Residual disease after surgery is associated with poor prognosis and chemotherapy has not been found to increase disease-free interval.3,12 Multiple retrospective studies have shown a survival benefit with radiation therapy when used in the adjuvant setting as well as for recurrent disease.3,13,14 A retrospective study from MD Anderson identified 10 patients treated with WART to a total dose of 27 to 28 Gy, 4 of whom had a complete clinical response to WART. The patients lived for 5, 10, 13 and 21 years.13
With the advent of modern radiation techniques like VMAT, safe and precise delivery of WART is possible. Acceptable acute and late toxicity has been reported at doses of 30 Gy when WART was used for gynecological cancers.15,16 We plan to monitor our patient with periodic CT scans. A sequential boost was decided against due to the presence of complicating ventral hernias from abdominal tumor burden.
Conclusion
GCTs should be aggressively considered for surgery as it remains the standard of care. When appropriate, adjuvant chemotherapy should be considered, especially for large tumors or if a tumor ruptures at surgery. For advanced disease, maximal debulking surgery should be contemplated, followed by systemic agents. WART delivered with modern radiation techniques results in excellent clinical and radiological response rates with acceptable toxicity and the possibility of long-term disease control. In the future, prospective studies to further investigate the timing of WART, adjuvant vs late, may help guide therapeutic options for patients with advanced GCTs.
References
- Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995;31(5):1341-1346.
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228-247.
- Pectasides D, Pectasides E, Psyrri A. Granulosa cell tumor of the ovary. Cancer Treat Rev. 2008;34(1):1-12.
- Young RH. Sex cord-stromal tumors of the ovary and testis: their similarities and differences with consideration of selected problems. Mod Pathol. 2005;18 Suppl2:S81-98.
- Vassal G, Flamant F, Caillaud JM, Demeocq F, Nihoul-Fekete C, Lemerle J. Juvenile granulosa cell tumor of the ovary in children: a clinical study of 15 cases. J Clin Oncol. 1988;6(6):990-995.
- Hammer A, Lauszus FF, Petersen AC. Ovarian granulosa cell tumor and increased risk of breast cancer. Acta Obstet Gynecol Scand. 2013;92(12):1422-1425.
- Nasioudis D, Wilson E, Mastroyannis SA, et al. Increased risk of breast and uterine cancer among women with ovarian granulosa cell tumors. Anticancer Res. 2019;39(9):4971-4975.
- Shah SP, Kobel M, Senz J, et al. Mutation of FOXL2 in granulosa-cell tumors of the ovary. N Engl J Med. 2009;360(26):2719-2729.
- Nofech-Mozes S, Ismiil N, Dube V, et al. Immunohistochemical characterization of primary and recurrent adult granulosa cell tumors. Int J Gynecol Pathol. 2012;31(1):80-90.
- Belz J, Wakker R, Hoepffner J, Caselitz J. Metastasis surgery in granulosa cell tumor--determining indications and tumor after care. Langenbecks Arch Chir. 1992;377(1):25-27.
- Lauszus FF, Petersen AC, Neumann G, et al. Less extensive surgery compared to extensive surgery: survival seems similar in young women with adult ovarian granulosa cell tumor. Eur J Obstet Gynecol Reprod Biol. 2014;177:61-66.
- Meisel JL, Hyman DM, Jotwani A, et al. The role of systemic chemotherapy in the management of granulosa cell tumors. Gynecol Oncol. 2015;136(3):505-511.
- Wolf JK, Mullen J, Eifel PJ, Burke TW, Levenback C, Gershenson DM. Radiation treatment of advanced or recurrent granulosa cell tumor of the ovary. Gynecol Oncol. 1999;73(1):35-41.
- Hauspy J, Beiner ME, Harley I, et al. Role of adjuvant radiotherapy in granulosa cell tumors of the ovary. Int J Radiat Oncol Biol Phys. 2011;79(3):770-774.
- Randall ME, Filiaci VL, Muss H, et al. Randomized phase III trial of whole-abdominal irradiation versus doxorubicin and cisplatin chemotherapy in advanced endometrial carcinoma: a Gynecologic Oncology Group Study. J Clin Oncol. 2006;24(1):36-44.
- Arians N, Kieser M, Benner L, et al. Adjuvant intensity modulated whole-abdominal radiation therapy for high-risk patients with ovarian cancer FIGO stage III: final results of a prospective phase 2 study. Radiat Oncol. 2019;14(1):179.
Citation
S C, S A, A M, M A. Whole Abdominal Radiation Therapy for Chemo- Refractory Adult Granulosa Cell Tumor of the Ovary: A Case Report. Appl Radiat Oncol. 2022;(1):43-47.
March 25, 2022