Tiny Yet Mighty: Nanotechnology in Radiation Therapy
Images
Nanotechnology is the application of extremely small things—a nanometer is one billionth of a meter—used in science, engineering and technology. It involves the ability to view and control individual atoms and molecules and has been used in designing new therapeutics and diagnostics in medicine. As such, interest is mounting to harness nanotechnology’s potential to enhance radiation therapy and advance cancer care.
Background
In 2005, the National Cancer Institute (NCI) and the National Institutes of Health (NIH) established the NCI Alliance for Nanotechnology in Cancer program, convening scientists from physical sciences, chemistry and engineering to work at the nanoscale with biologists and clinicians. The program has been led from its inception by Piotr Grodzinski, PhD, initially within the Office of the Director at NCI and currently from Cancer Imaging Program and the Nanodelivery Systems and Devices Branch (NSDB), where Dr. Grodzinski is branch chief.
Although the alliance’s early research demonstrations favored diagnostics, the focus has gradually moved to therapeutic projects such as chemotherapy, immunotherapy, gene therapy, and radiation therapy.1
Dr. Grodzinski explains that during the early years of NCI’s nanotechnology programs, a key goal was to examine therapeutic and diagnostic strategies for nanotechnology and nanoparticles, building off work performed in academia and supported via funding from the National Science Foundation or the Defense Advanced Research Projects Agency (DARPA), an agency within the Department of Defense (DoD).
“We are now seeing a number of different therapeutics being approved by the FDA, many of them based on liposomal formulation, which is an earlier stage of nanoparticle design,” Dr. Grodzinski says.
While liposomal delivery is typically used for chemotherapy, nanotechnologies also focus on radiation therapy. “It’s not just radiotherapy enhancers, although that’s one bolus of them,” says Christopher M. Hartshorn, PhD, program director, NCI Nanodelivery Systems and Devices Branch, who works closely with Dr. Grodzinski. “There are combination platforms to deliver both chemo- and radiation therapy, specifically to provide local attenuation of the tumor microenvironment (TME) and make it more radiation sensitive. Then there are local radiation broadcasting effects that either enhance the x-rays themselves or generate photons locally to make it more similar to photodynamic therapy, albeit to deep tissue.”
One such company is Nanobiotix, maker of Hensify (NBTXR3), an aqueous suspension of functionalized crystalline hafnium oxide (HfO2) nanoparticles for intratumoral delivery, which is synchronized with the onset of the patient’s standard radiation therapy treatment. While Nanobiotix has received first market approval in Europe for the treatment of soft-tissue sarcomas, clinical trials are also underway in the US, Europe and Asia evaluating NBTXR3 in head and neck squamous cell carcinomas, lung and liver metastases, hepatocellular carcinoma and others. These trials assess applications including NBTXR3 as a single agent activated by radiation therapy, and as a combination agent with biologics, chemotherapy, and immune checkpoint inhibitors.
“Cancer treatment is very multi-modal in many ways so researchers are also looking at combination therapies combining different modalities: radiation, chemotherapy and/or immunotherapy together,” adds Dr. Grodzinski. “Radiation can be used to stimulate the effectiveness of immunotherapy. In one of the innovative works, Andrew Wang, MD [associate professor and radiation oncologist at the University of North Carolina at Chapel Hill] used nanoparticles to capture antigens released upon radiation therapy and then trafficked them through the body to stimulate T-cells.
According to Dr. Hartshorn, the NCI has also funded research on nanotechnology-based radiodynamic therapy to increase the efficacy of checkpoint inhibitor immunotherapies. The lead investigator, Wenbin Lin, PhD, the James Franck Professor of Chemistry and Radiation & Cellular Oncology at the University of Chicago, uses metal-organic framework nanoparticles, which combine metal nodes with organic binders injected into the tumor prior to radiation therapy. The external radiation interacts with nanoparticles and triggers reactive oxygen compounds toxic to the cancerous cells. Importantly, the design can incorporate an IDO inhibitor—a novel checkpoint inhibitor—as well as be delivered in combination with existing anti-PD-1/PD-L1 treatment strategies. In preclinical studies, these combinations displayed enhanced efficacy and stimulation of a more prolonged immune response than when used separately. Moreover, the response generated from the initial therapeutic perturbation enabled immune response at the tumor site as well as to nearby and, often, distant lesions.2
Dr. Lin’s company, RiMO Therapeutics, Inc., began a phase 1 clinical trial of RiMO-301 in patients with advanced tumors clinically accessible for injection.3 While the company’s strategy moving forward is likely for head and neck cancers, Dr. Hartshorn adds that preclinical work has focused on breast, colorectal and ovarian cancers.
At Memorial Sloan Kettering Cancer Center, Director of Intraoperative Imaging Michelle Bradbury, MD, PhD, who is also professor of radiology at the Weill Cornell Medical College and co-leader of one of the Centers of Cancer Nanotechnology Excellence funded by the alliance program, and her team are using a type of nanotechnology called C-dots that are one-thousandth the size of a red blood cell. Much of the work in Dr. Bradbury’s lab, which is focused on cancer imaging uses a new generation of multimodal (PET-optical) C-dots-bearing peptides, co-developed with Ulrich B. Wiesner, PhD, Spencer T. Olin Professor of Engineering at Cornell University. Drs. Bradbury and Wiesner, along with colleagues, also re-purposed C-dots for use as drug-delivery vehicles to treat cancer. They published the results of a pre-clinical study demonstrating that these silica nanoparticles functionalized with melanoma-targeted peptides induce cell death in starved cancer cells and cancer-bearing mice.4 Furthermore, they have begun to incorporate alternative labeling approaches to this platform. The goal is to be able to perform β-particle / α-particle radiation therapy using131I and211At radionuclides, respectively.
For nearly a decade, Otto Zhou, PhD, led a group at the University of North Carolina at Chapel Hill in developing a carbon-nanotube-based microbeam radiation therapy (MRT) unit that works muck like a synchrotron, a circular particle accelerator, except it delivers radiation over 20 to 30 minutes compared to the fraction of a second in a synchrotron. Using the MRT, Dr. Zhou and co-authors demonstrated how the device applied a higher radiation dose while sparing normal brain tissue in a preclinical study.5
Unfortunately, despite promising preclinical studies of nanotechnologies in cancer treatment, moving to human clinical trials and regulatory approval is challenging. As Drs. Grodzinski and Hartshorn note, many of the innovative technologies are still in pre-clinical studies and are slowly transitioning beyond that stage. “A lot of these technologies are being developed by small companies,” Dr. Grodzinski says. “Clinical trials are expensive and these smaller companies can likely go to phase 1; however, they often establish partnerships with larger companies to take those trials further.”
It’s also the particular cancer application area of these novel nanotechnologies that can lead to success in clinical trials and ultimately regulatory clearance, Dr. Grodzinski adds. Using fairly well-established chemotherapeutic agents with nanotechnology carries less risk for a successful translation than using an inorganic, heavy particle for localized radiation as this requires more thorough characterization and, ultimately, more regulatory hurdles for safety.
Liposomal delivery platforms remain in that lower-risk threshold, says Dr. Hartshorn, because there is 40-plus years of in vivo data to support delivery parameters and well-controlled manufacturing as well as accepted safety parameters for their drug “cargo.”
“The broader question is whether these technologies will be accepted or not accepted over time by radiation oncologists,” Dr. Hartshorn adds. “In many cases with otherwise immunocompromised individuals, cachectic patients, or pediatrics, therapeutic toxicity doesn’t allow them to receive standard-of-care dosing of radiotherapy after a certain point or at all. Yet in most cases, nanoparticle strategies enable lower doses to these individuals, although similar efficacy of the higher dose over time, with decreased toxicity. Also, some of the radiation therapy combination platforms are showing modest curative effects in conjunction with immunotherapy. Collectively, these are strengths of these systems when used for radiotherapeutic applications and should, over time, help to establish them in clinical practice.”
In a recent paper, Dr. Grodzinski, Dr. Hartshorn and co-authors write that overcoming tumor heterogeneity hurdles relies on an understanding of nano-bio interactions, particle transport to tumor cells and targeting of TME, or premetastatic disease, to enhance treatment response. A similar approach should be utilized in preclinical research, using in vivo imaging to track carrier and drug delivery within heterogeneous tumor tissue and surrounding microenvironment. Further, nanotechnology/nanomedicine research should stay focused in areas where conventional approaches had little to no success and where the advantages of nanotechnologies can be effectively used. Rational design methodologies are also essential to bring technology from the bench to the bedside.6
Gold Nanoparticles
At Nanoprobes, founder James F. Hainfeld, PhD, has been researching the use of nanoparticles in cancer therapy and other diseases since his days as a senior scientist at Brookhaven National Laboratory in the 1970s. He and Frederic R. Furuya, PhD, developed the Nanogold cluster, comprised of gold compounds with a core of gold atoms and organic groups bound to the surface of gold atoms. Dr. Hainfeld frequently collaborates with Henry M. Smilowitz, PhD, associate professor in the Department of Cell Biology at the University of Connecticut.
Heavy atom nanoparticles made of gold or iodine absorb x-rays. When the radiation is delivered and absorbed by the tissue, the gold/iodine nanoparticles eject electrons, creating free radicals and damaging the tumor DNA. The calculated dose enhancement can be 5 to 8 times the delivered radiation dose and pre-clinical studies have demonstrated up to 10 times the life extension compared to animals that received radiation only.
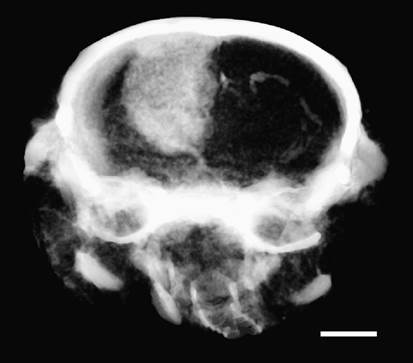
Drs. Hainfeld and Smilowitz were co-authors of a pre-clinical study using mice where these gold nanoparticles were shown to enhance radiation therapy (x-ray) uptake with a 19:1 tumor to normal brain ratio (Figure 1). With the gold nanoparticles, local radiation dose increased by approximately 300 percent and resulted in an average 53 percent tumor-free survival over 1 year compared to 9 percent using radiation alone.7
“We focus on brain tumors because cancer drugs are notoriously not effective due to the blood-brain barrier,” Dr. Hainfeld explains. “This is an unmet need in cancer.”
Although Temozolomide was a breakthrough drug for some brain tumors such as glioblastoma multiforme and anaplastic astrocytoma, there are few options for patients with brain metastases, he adds.
Dr. Hainfeld hopes to initiate human trials soon; in the meantime, he continues to work on refining the particles and delivery. He has learned that high atomic number nanoparticles seem to work best with radiation systems that use kilovolts rather than megavolts—or the older radiation therapy systems. Today’s modern linacs use megavoltage x-rays.
“If this nanoparticle method is further developed, then it could bring effective radiation therapy to lower income areas of the world where megavoltage radiation therapy systems are too expensive to acquire,” he adds. Dr. Hainfeld is working on segmenting the kilovoltage radiation to avoid the potential for skin burns, a disadvantage of this type of system if the radiation is delivered from only 1 direction.
Although many pharmaceutical companies are focused on small-molecule drugs, Dr. Hainfeld believes nanoparticles are a more versatile platform.
“If you inject or orally take a drug, it goes around the body through the blood and the accumulation in the tumor is not very high,” he explains. “Nanoparticles can be made to stay in the blood longer to accumulate in the tumor and not be rapidly excreted. With nanoparticles, we can incorporate multiple components for better targeting and functionality.”
First Commercial Radioenhancer
Nanobiotix has embarked on two clinical trials in the US to evaluate the safety and feasibility of Hensify (NBTXR3) in soft-tissue sarcomas. One clinical trial led by Adam Dicker, MD, Thomas Jefferson University Hospital, and Paul Nguyen, MD, Dana-Farber Cancer Institute, involves newly diagnosed patients with unfavorable intermediate risk or high-risk prostate adenocarcinoma.8 A second trial evaluating the safety, efficacy and tolerability of NBTXR3 activated by radiation therapy in combination with an anti-PD-1 therapy in 3 patient cohorts is ongoing. The first cohort includes patients with locoregional recurrence or relapsed metastatic head and neck squamous cell cancer with the target lesion in a previously irradiated field. In cohorts two and three, patients present with lung or liver metastases from any primary cancer eligible for anti-PD-1 therapy.9
The company also announced in early May that a phase 1 trial with NBTXR3 in pancreatic cancer is safe to proceed with The University of Texas MD Anderson Cancer Center per the US FDA.
According to Laurent Levy, PhD, CEO at Nanobiotix, the company has 15 clinical trials in its development plan, including 9 new phase 1/2 trials with MD Anderson treating 6 cancer indications and involving 340 patients.
Key considerations in the development of NBTXR3 were that it fit into the current oncology workflow, can work with any type of radiation therapy system, including proton therapy, and does not change the fundamental processes in radiation therapy.
“The initial question that triggered the work we are doing was, can we use physics to influence a cell from the inside?” says Dr. Levy. “Our product answers that question by improving the dose in the tumor without increasing the dose in surrounding tissue. It is making radiation more efficient, keeping the curative effect while decreasing the side effect (in surrounding healthy tissue).”
Dr. Levy explains the nanoparticle is 15 nanometers and is injected into the patient’s tumor the day before treatment using the same pathway used to biopsy the tumor. The particle is designed to absorb the x-rays/radiation—the higher the density of the material, such as hafnium, the higher the x-ray absorption. NBTXR3 remains in the patient’s body indefinitely as it is not degraded or excreted from the body and there is no toxicity to the patient. However, in the soft-tissue sarcoma trials, 8 percent of patients had acute immunological response at the time of injection, he adds.
“By adding this one injection of a nanoparticle with hafnium, we can change the outcome for the patient,” Dr. Levy says. “We see for most of the head and neck patients it is a sustainable, complete response, with a large proportion of them still alive at 24-month follow-up. Looking at the literature, the median overall survival for this type of patient is around 12 to 13 months.”
A Transformational Combination Radiation Therapy
TAE Life Sciences is pursuing a combination biologically targeted radiation therapy through the development of new targeted boron-10 drugs and an accelerator-based neutron system. The company is developing this system, designed to fit in a hospital environment, which overcomes a key limitation: that neutrons for boron neutron capture therapy (BNCT) were historically only available from the core of a nuclear reactor.
BNCT is a combination therapy, comprised of a target drug and radiation, says Bruce Bauer, PhD, CEO of TAE Life Sciences. He explains that the target drug carries boron-10, which has a large cross section to capture neutrons. The drug is delivered intravenously and biologically targets the cancer cell. While it has a short retention within the cancer cell, the drug must achieve a sufficient concentration in the cell, requiring radiation therapy delivery approximately two hours after infusion. The boron-10 then captures the neutrons from the accelerator and begins the process of cytotoxicity.
“When that capture reaction occurs, there’s a secondary reaction that takes place that generates a lithium ion and alpha particle,” he says. “Most of the damage is done by the alpha particle with a very high linear energy transfer. So, the cytotoxic radiation, which is killing the cell, is generated inside the cell by this capture reaction from the combination of boron and low-energy neutrons.” Dr. Bauer adds that the cell death reaction is three times that of a photon and treatments can be delivered in one or two treatment sessions.
Currently, nine BNCT initiatives are underway in Japan, and most recently, BNCT has been approved by the Japanese regulatory bodies and insurers for head and neck cancer treatments. TLS recently announced a dedicated drug development program to improve boron delivery, retention and homogeneous distribution in the cancer cells and their first neutron-beam system is planned to be delivered to a hospital site in China later this year with a goal to start clinical trials in 2021.
References
- Hartshorn CM, Russell LM, Grodzinski P. National Cancer Institute Alliance for nanotechnology in cancer—catalyzing research and translation toward novel cancer diagnostics and therapeutics. WIREs Nanomed Nanobiotechnol. 2019;11(6):e1570. doi:10.1002/wnan.1570
- Lu K, He C, Guo N, et al. Low-dose x-ray radiotherapy–radiodynamic therapy via nanoscale metal-organic frameworks enhances checkpoint blockade immunotherapy. Nat Biomed Eng. 2018;2:600-610. doi:10.1038/s41551-018-0203-4
- Phase I study of RiMO-301 with radiation in advanced tumors. ClinicalTrials.gov Identifier: NCT03444714. Accessed June 3, 2020. https://clinicaltrials.gov/ct2/show/NCT03444714
- Kim SE, Zhang L, Ma K, et al. Ultrasmall nanoparticles induce ferroptosis in nutrient-deprived cancer cells and suppress tumour growth. Nat Nanotechnol. 2016;11(11):977‐985. doi:10.1038/nnano.2016.164
- Bazyar S, Inscoe CR, Benefield T, et al. Neurocognitive sparing of desktop microbeam irradiation. Radiation Oncology (London, England). 2017 Aug;12(1):127. doi: 10.1186/s13014-017-0864-2
- Hartshorn CM, Russell LM, Grodzinski P. National Cancer Institute Alliance for Nanotechnology in Cancer—Catalyzing research and translation toward novel cancer diagnostics and therapeutics. WIREs Nanomed Nanobiotechnol. 2019;11(6):e1570. doi:10.1002/wnan.1570
- Hainfeld JF, Smilowitz HM, O’Connor MJ, Dilmanian FA, Slatkin DN. Gold nanoparticle imaging and radiotherapy of brain tumors in mice. Nanomedicine (Lond). 2013;8(10):1601‐1609. doi:10.2217/nnm.12.165
- NBTXR3 Nanoparticles and EBRT or EBRT with brachytherapy in the treatment of prostate adenocarcinoma. ClinicalTrials.gov Identifier: NCT02805894. Accessed June 3, 2020. https://clinicaltrials.gov/ct2/show/NCT02805894?term=NBTXR3&cntry=US&draw=2&rank=1
- NBTXR3 activated by radiotherapy for patients with advanced cancers treated with an anti-PD-1 therapy. ClinicalTrials.gov Identifier: NCT03589339. Accessed June 3, 2020. https://clinicaltrials.gov/ct2/show/NCT03589339?term=NBTXR3&cntry=US&draw=2&rank=2
The author acknowledges Dr. Piotr Grodzinski and Dr. Christopher M. Hartshorn from the Cancer Imaging Program and the Nanodelivery Systems and Devices Branch (NSDB) of the NCI for assistance in the development of this article.
Citation
MB M. Tiny Yet Mighty: Nanotechnology in Radiation Therapy. Appl Radiat Oncol. 2020;(2):23-26.
June 22, 2020