The Role of Patient Reported Outcomes in Esophageal Cancer Patients Receiving Chemoradiation Therapy
Images
Abstract
Background and Objectives: To determine if primary esophageal cancer (EsoCa) characteristics were related to unique Edmonton Symptom Assessment Scale (ESAS) symptom reports.
Methods: Records of patients with EsoCa receiving chemoradiation therapy (CRT) were retrospectively screened against a single institutional ESAS database. The majority of patients received concurrent folinic acid, fluorouracil, and oxaliplatin (FOLFOX) and 5.5 weeks of radiation therapy (RT) to 50.4-56.0 Gy. During treatment, patients completed a weekly ESAS survey. Relationships between clinical variables and ESAS scores were analyzed using the Mann-Whitney U test and variables were correlated using Kendall’s tau-b tests.
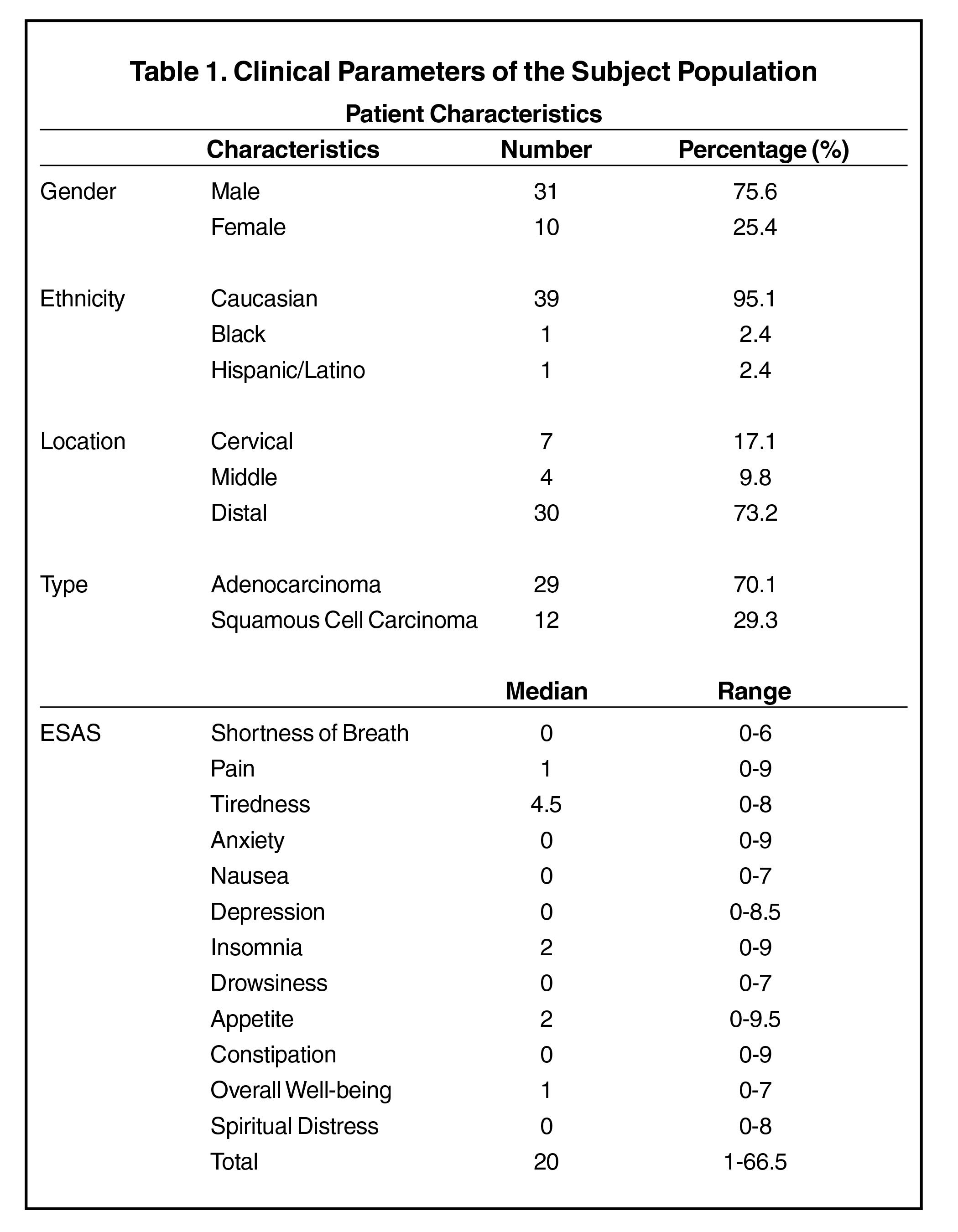
Results: A total of 87 patients with EsoCa receiving CRT completed ESAS between February 2017 and July 2019 with 41 completing ≥ 3 ESAS surveys (median = 5, mean = 5.6, range = 3-12). In this cohort, 75.3% were men (n = 31) and 95.1% were White/Caucasian (n = 39). Seven patients had cervical lesions (17.1%), four (9.8%) middle, and 30 (73.2%) distal. A total of 72.5% of patients had adenocarcinoma (n = 29). Tiredness had the highest median ESAS score (4.00, median total score 22.4). Patients with middle lesions were more likely to experience pain (4.25 vs 0.5, P = 0.038) and drowsiness (2.5 vs 0, P = 0.022). Distal and cervical lesions did not demonstrate statistically significant relationships.
Conclusion: In this analysis of patient reported outcomes (PRO) in EsoCa, patients with middle esophageal lesions were more likely to experience pain and drowsiness.
Meaningful patient-centered care requires the measurement of patient concerns and implementation of tailored clinical solutions. To personalize therapy informed by the patient perspective, objective clinical data is ideally combined with collection and assessment of patient-reported outcomes (PROs). In addition to providing actionable symptom burden data for intervention, PROs have been shown to correlate with diagnosis,1,2 radiographic response to treatment,3 and early identification of disease progression.4
Our center has been collecting PRO data using the Edmonton Symptom Assessment Scale (ESAS) for several years in our radiation oncology and supportive care clinics. We have previously discussed the role of ESAS data in unselected RT patients5 and in specific cohorts with retroperitoneal sarcoma6 and multiple myeloma.7 Recently we have assessed the role of PRO in clinical scenarios such as anemia.8
The value of PROs to assess treatment-related toxicity and the effects of palliative chemotherapy and/or radiation therapy (RT) on the quality of life for patients with esophageal cancer has been confirmed.9 However, PROs are also more likely than clinical outcome measures to provide information pertinent to the functioning of patients with esophageal cancer.10 Patterns in the PROs of esophageal cancer populations may provide a basis to anticipate symptoms and provide proactive targeted treatment and increased support.11 We were interested in investigating the role of PROs, specifically ESAS, in EsoCa because of the disparate clinical behavior of lesions by location within the organ. For instance, neck masses, odynophagia, hoarseness or referred otalgia12 may be noted by patients with cervical lesions. Retrosternal pain may be due to mediastinal invasion of middle esophageal lesions. Advanced lesions of the distal esophagus often present solely with dysphagia and weight loss.13 Very few studies exist describing the relationship between clinical characteristics and ESAS scores in patients being treated with chemoradiation therapy (CRT) for esophageal cancer.
We analyzed our institutional ESAS data to better characterize associations of patient-reported symptoms with esophageal cancer location, since earlier identification and control of esophageal symptoms may reduce patient burden and help avoid unplanned hospitalizations or need for IV fluid interventions.
Materials and Methods
After institutional review board approval, we performed a single-institution retrospective analysis of records of patients with EsoCa receiving RT with concurrent chemotherapy. These were compared with the institutional ESAS database and pertinent data collated. Patients coded as having gastroesophageal junction lesions were excluded to reflect pure esophageal treatment since, typically, less of the esophageal mucosa is involved in the 50.4 Gy field during treatment of these lesions. Patients were assessed for gender, marital status, vital status, histology, and tumor location, which were then analyzed to determine relationships between these variables and ESAS scores. Remaining patient characteristics are available in Table 1.
The majority of patients received concurrent FOLFOX (folinic acid/fluroruracil/oxaliplatin) with 5.5 weeks of intensity-modulated radiation therapy (IMRT) to 50.4-56.0 Gy in 1.8-2.0 Gy/fraction.14-16 Patients on treatment were evaluated weekly by the staff radiation oncologist; on this visit they routinely completed an ESAS survey.
Relationships between clinical variables and ESAS scores were analyzed using the Mann Whitney U test, and correlations between variables were calculated by Kendall’s tau-b tests performed using SPSS (Statistical Package for the Social Sciences) software.
Results
A total of 87 patients with EsoCa were identified who completed ESAS between February 2017 and July 2019. Of these patients, 41 completed ≥ 3 ESAS surveys (median = 5, mean 5.6, range 3-12) while on treatment and form the cohort further analyzed.
As outlined in Table I, most patients were men (75.6%, n = 31) and White/Caucasian (n = 95.1%, n = 39). Seven (17.1%) patients had cervical esophageal lesions, four (9.8%) had middle lesions and 30 (73.2%) had distal lesions. Most patients had adenocarcinoma (70.1%, n = 29). The ESAS item with the highest median score was tiredness (4.00) with a median total score of 22.4. Patients in this study were noted to lose 3.5% of their body weight after treatment.
Patients with middle esophageal lesions were more likely to experience pain (4.25 v. 0.5, P = 0.038) and drowsiness (2.5 v. 0, P = 0.022), but no statistically significant relationships were seen for those with distal or cervical lesions. Women (4.75 vs. 0.50, P = 0.02) and unmarried individuals (4.5 v. 0.5, P = 0.021) were more likely to have a worse appetite while those who died were more likely to experience constipation (2.5 v. 0, P = 0.005). Male gender, married status, histology, and remaining alive after treatment did not demonstrate any associations that were statistically significant.
The strongest correlation between symptoms were found between depression and spiritual pain (tb 0.645, P < 0.001). Shortness of breath was correlated with the most symptoms, including pain (0.373, P = 0.005), tiredness (0.283, P = 0.027), anxiety (0.314, P = 0.022), depression (0.462, P = 0.001), drowsiness (0.424, P = 0.002), appetite (0.298, P = 0.023), overall well-being (0.299, P = 0.023), and spiritual pain (0.342, P = 0.018).
Discussion
There is a paucity of studies discussing clinical characteristics and their relationship with ESAS scores. One study discussed the likelihood of severe symptom burden based on clinical characteristics and elapsed time after diagnosis while establishing the prevalence of various symptoms assessed using ESAS as a whole, but did not correlate symptoms with one another or with clinical characteristics,11 while others have been solely focused on patients undergoing palliative care.17,18 Additional studies have targeted the use of different PRO surveys and their association with T-stage,19 to compare patient-reported quality of life between patients receiving CRT and surgery vs surgery alone,20 to compare quality of life between patients receiving palliative brachytherapy and external beam radiotherapy,21 to determine impact of treatment on quality of life,22-24 and prognosis and/or survival25-28 in patients with esophageal cancer. None of these studies have discussed the association of distinct clinical characteristics with ESAS scores and, therefore, symptoms in those patients receiving CRT for esophageal cancer.
Studies such as this one may inform a patient’s potential for a variety of symptoms and provide proactive, personalized treatment tailored to the individual. We note that self-reporting of ESAS pain and drowsiness was only significant in patients with middle esophageal cancer, indicating that patients with esophageal cancer in different disease locations may demonstrate variability in self-reported symptoms as a function of the site of lesions. This variability may also point to differing risk for impairments in quality of life and care needs. For instance, some patients undergoing treatment may need pain medications due to treatment side effects or from the cancer itself. These medications tend to cause drowsiness and a host of other adverse effects, so those taking pain medications regularly are likely to experience more tiredness in their everyday life during treatment.
Additionally, some patients may encounter nutritional deficiencies due to treatment effects such as nausea or dysphagia. Patients who experience nausea, especially if it is refractory to antiemetic medication, may not be able to eat as much in terms of volume and variety of foods. In such cases, they may not have enough intake of calories or nutrients to sustain the energy levels they are used to.29,30 Patients in this study experienced a median weight loss of 3.5% from their pre-treatment weight during treatment, demonstrating possible difficulty maintaining the proper level of nutrition. Additionally, if a tumor is obstructing a portion of the esophagus, they may have difficulty eating foods of a specific type or texture, which can lead to similar sequalae.
Other factors contributing to a patient’s experience during treatment include various lifestyle changes. Smoking and alcohol use are two major risk factors for esophageal cancer. Unfortunately, while some patients may stop these activities during and even after treatment, others continue these behaviors throughout treatment.31 This can lead to a worsening of side effects during treatment, including increasing odynophagia, which can also lead to nutritional problems since this would likely be exacerbated while eating. These variables were not studied in our patient cohort so we cannot comment on their relevancy to our findings.
Another factor to consider is the level of support a patient may have. A patient with a robust support system may be able to better adjust to the changes observed when undergoing treatment.32 Friends or family who prepare meals for them, perform household work, help them make lifestyle changes, and provide emotional support may drastically lift some of the burden on these patients so that they can focus their energy on healing rather than continuing to expend energy on other tasks. Including such additional factors was beyond the scope of this project but future work is planned to incorporate variables relating to the degree of support.
As the reliability and predictability of PROs linked to specific diagnoses such as EsoCa are confirmed, PROs may become important tools for clinicians to help plan treatments and supportive care. While intriguing, this retrospective analysis should be interpreted cautiously. Nevertheless, further analysis with other large PRO libraries is indicated to validate these findings.
References
- Paparrizos J, White RW, Horvitz E. Screening for pancreatic adenocarcinoma using signals from web search logs: feasibility study and results. J Oncol Pract. 2016;12(8):737-744.
- White RW, Horvitz E. Evaluation of the feasibility of screening patients for early signs of lung carcinoma in web search logs. JAMA Oncol. 2017;3(3):398-401.
- Victorson D, Soni M, Cella D. Metaanalysis of the correlation between radiographic tumor response and patient-reported outcomes. Cancer. 2006;106(3):494-504.
- Denis F, Lethrosne C, Pourel N, et al. Randomized trial comparing a web-mediated follow-up with routine surveillance in lung cancer patients. J Natl Cancer Inst. 2017;109(9).
- Johnstone PAS, Lee J, Zhou JM, et al. A modified Edmonton Symptom Assessment Scale for symptom clusters in radiation oncology patients. Cancer Med. 2017;6(9):2034-2041.
- Palm RF, Jim HSL, Boulware D, Johnstone PAS, Naghavi AO. Using the revised Edmonton symptom assessment scale during neoadjuvant radiotherapy for retroperitoneal sarcoma. Clin Transl Radiat Oncol. 2020;22:22-28.
- Nanda R, Boulware D, Baz R, et al. Patient-reported outcomes regarding radiation therapy in patients with multiple myeloma. Acta Oncol. 2020:1-5.
- Johnstone PAS, Alla R, Yu HM, et al. Patient-reported outcomes: using ESAS to screen for anemia. Support Care Cancer. 2020;28(9):4141-4145.
- Amdal CD, Jacobsen AB, Guren MG, Bjordal K. Patient-reported outcomes evaluating palliative radiotherapy and chemotherapy in patients with oesophageal cancer: a systematic review. Acta Oncol. 2013;52(4):679-690.
- Cella D, Riley W, Stone A, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008. J Clin Epidemiol. 2010;63(11):1179-1194.
- Gupta V, Allen-Ayodabo C, Davis L, et al. Patient-reported symptoms for esophageal cancer patients undergoing curative intent treatment. Ann Thorac Surg. 2020;109(2):367-374.
- Marks JE, Kurnik B, Powers WE, Ogura JH. Carcinoma of the pyriform sinus. An analysis of treatment results and patterns of failure. Cancer. 1978;41(3):1008-1015.
- Schrump DS AN, Firastiere AA, Minsky BD. Cancer of the Esophagus. In: DeVita V, Hellman S, Rosenberg SA, ed. Cancer: Principles and Practice of Oncology 6th edition (July 2001). 6th ed. Lippincott Williams & Wilkins; 2001:1058-1059.
- Goodman KA, Hall N, Bekaii-Saab TS, et al. Survival outcomes from CALGB 80803 (Alliance): a randomized phase II trial of PET scan-directed combined modality therapy for esophageal cancer. J Clin Oncol. 2018;36(15_suppl):4012-4012.
- La TH, Minn AY, Su Z, et al. Multimodality treatment with intensity modulated radiation therapy for esophageal cancer. Dis Esophagus. 2010;23(4):300-308.
- Venkat PS, Shridhar R, Naghavi AO, et al. Dose escalated neoadjuvant chemoradiotherapy with dose-painting intensity-modulated radiation therapy and improved pathologic complete response in locally advanced esophageal cancer. Dis Esophagus. 2017;30(7):1-9.
- Merchant SJ, Brogly SB, Booth CM, et al. Palliative care and symptom burden in the last year of life: a population-based study of patients with gastrointestinal cancer. Ann Surg Oncol. 2019;26(8):2336-2345.
- Merchant SJ, Kong W, Brundage M, Booth CM. Symptom evolution in patients with esophageal and gastric cancer receiving palliative chemotherapy: a population-based study. Ann Surg Oncol. 2021;28(1):79-87.
- Kidane B, Ali A, Sulman J, Wong R, Knox JJ, Darling GE. Health-related quality of life measure distinguishes between low and high clinical T stages in esophageal cancer. Ann Transl Med. 2018;6(13):270.
- Noordman BJ, Verdam MGE, Lagarde SM, et al. Effect of neoadjuvant chemoradiotherapy on health-related quality of life in esophageal or junctional cancer: results from the randomized CROSS Trial. J Clin Oncol. 2018;36(3):268-275.
- van Rossum PSN, Jeene PM, Rozema T, et al. Patient-reported outcomes after external beam radiotherapy versus brachytherapy for palliation of dysphagia in esophageal cancer: a matched comparison of two prospective trials. Radiother Oncol. 2021;155:73-79.
- Safieddine N, Xu W, Quadri SM, et al. Health-related quality of life in esophageal cancer: effect of neoadjuvant chemoradiotherapy followed by surgical intervention. J Thorac Cardiovasc Surg. 2009;137(1):36-42.
- Trudel JG, Sulman J, Atenafu EG, Kidane B, Darling GE. Longitudinal evaluation of trial outcome index scores in patients with esophageal cancer. Ann Thorac Surg. 2016;102(1):269-275.
- Blazeby JM, Farndon JR, Donovan J, Alderson D. A prospective longitudinal study examining the quality of life of patients with esophageal carcinoma. Cancer. 2000;88(8):1781-1787.
- Kidane B, Sulman J, Xu W, et al. Baseline measure of health-related quality of life (functional assessment of cancer therapy-esophagus) is associated with overall survival in patients with esophageal cancer. J Thorac Cardiovasc Surg. 2016;151(6):1571-1580.
- Djärv T, Lagergren P. Six-month postoperative quality of life predicts long-term survival after oesophageal cancer surgery. Eur J Cancer. 2011;47(4):530-535.
- Djärv T, Metcalfe C, Avery KN, Lagergren P, Blazeby JM. Prognostic value of changes in health-related quality of life scores during curative treatment for esophagogastric cancer. J Clin Oncol. 2010;28(10):1666-1670.
- Quinten C, Martinelli F, Coens C, et al. A global analysis of multitrial data investigating quality of life and symptoms as prognostic factors for survival in different tumor sites. Cancer. 2014;120(2): 302-311.
- Davidson W, Teleni L, Muller J, et al. Malnutrition and chemotherapy-induced nausea and vomiting: implications for practice. Oncol Nurs Forum. 2012;39(4):E340-345.
- Jordan T, Mastnak DM, Palamar N, Kozjek NR. Nutritional therapy for patients with esophageal cancer. Nutr Cancer. 2018;70(1):23-29.
- Steevens J, Schouten LJ, Goldbohm RA, van den Brandt PA. Alcohol consumption, cigarette smoking and risk of subtypes of oesophageal and gastric cancer: a prospective cohort study. Gut. 2010;59(1):39-48.
- Zhang Y, Cui C, Wang Y, Wang L. Effects of stigma, hope and social support on quality of life among Chinese patients diagnosed with oral cancer: a cross-sectional study. Health Qual Life Outcomes. 2020;18(1):112.
Citation
J M, AJ S, JM F, HM Y, RH N, D P, SE H, HSL J, PAS J. The Role of Patient Reported Outcomes in Esophageal Cancer Patients Receiving Chemoradiation Therapy. Appl Radiat Oncol. 2021;(2):22-25.
July 27, 2021