Technology Trends: Proton Therapy Treatment for Breast Cancer
Images
When treating breast cancer with radiation therapy, several rare, yet serious, long-term side effects may occur. These include heart disease, radiation pneumonitis (inflammation of the lung), rib fractures, and the very rare side effect, brachial plexopathy, which is caused by radiation damage to the nerves in the upper chest (the brachial plexus).
With proton therapy, the energy distribution of protons into tissue can be more precisely directed and controlled than with conventional photon therapy, allowing it to potentially reduce long-term side effects in breast cancer patients. Protons are energized to specific velocities, and as they move through the body, they slow down and increase interaction with the electrons orbiting the atom of molecules, a fundamental component of all body tissue. Because proton beam therapy targets only the cancerous cells, there is less damage to surrounding healthy tissue.
In September 2014, initial results of a clinical trial using proton therapy for treating breast cancer reported excellent survival rates and cosmetic outcomes. The trial was conducted at the James M. Slater, MD, Proton Treatment & Research Center at Loma Linda University Medical Center (LLUMC) in Loma Linda, California, the first U.S. hospital-based proton therapy center. After opening in October 1990, LLUMC has treated more than 18,000 patients and over 20 types of cancer. According to principal investigator David Bush, MD, vice chairman of the Department of Radiation at LLUMC, the study included 100 women with early stage disease who had small, localized tumors that had not spread. The patients were monitored for an average of 5 years following treatment. Results showed that in-breast, recurrence-free survival rate was 97% with minimal side effects, and a good-to-excellent cosmetic rate in 90% of the cases.1
“We were able to show that by treating a smaller area of the breast, we could essentially eliminate exposure to the heart, chest wall, and lung,” Dr. Bush says. “There was no toxicity in those structures, tissues or organs, and the cosmetic rate was very good.”
Dr. Bush is embarking on another proton therapy breast cancer trial with 150 enrolled participants. The new study will also examine rate of recurrence; disease-free survival; and complication rates in the breast, chest wall and skin. While the new trial is similar to the first, it includes women with more adverse factors and slightly more advanced tumors. In general, Dr. Bush explains that ideal candidates for breast proton therapy are women with breast cancer who have small localized tumors < 3 cm, and whose disease has not spread, including to the lymph nodes.
An Evolution in Technology
Since its inception, proton therapy has used image guidance, notes Dr. Bush. Imaging, along with the use of implanted fiducial markers, are key ways LLUMC and many other sites manage uncertainty. Patient immobilization is also critical since proton therapy delivers a higher radiation dose than conventional photon therapy, and the breast is prone to movement from the patient’s respiratory cycle.
There is also the challenge of managing dose to the skin, Dr. Bush adds. “The targets we are treating are not deep in the body—they are often close to the skin line. So we need to reduce the dose to the skin to avoid burns by immobilizing the breast in an accurate, reproducible way.”
To do this, LLUMC developed a patented customized foam. The patient lies prone during treatment and the skin is in contact with this immobilization device, which eliminates motion due to breathing in an accurate and reproducible way, thus helping to spare the skin.
Scanning beam is another advance in proton therapy delivery under development at LLUMC. A scanning-beam technique electronically or magnetically steers a narrow beam (sometimes referred to as a pencil beam) as it “paints” the treatment volume in layers, voxel by voxel. “We believe that scanning-beam technology will likely be well-suited to treat patients who have large, more complicated volumes that need to be treated,” says Dr. Bush. This includes patients requiring whole-breast radiation or lymph node treatment.
Proton Therapy at Ackerman Center
As a physician-owned private proton therapy center, Ackerman Cancer Center in Jacksonville, Florida, began treating patients with the Mevion S250 Proton Therapy System in April 2015. “I believe proton therapy is an important part of the future of radiation oncology,” says Scot Ackerman, MD, medical director of the center. Approximately 10% to 15% of his patients have breast cancer, half of whom are receiving proton therapy treatment.
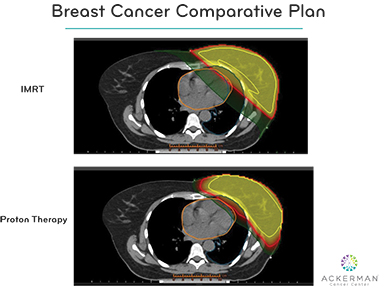
With every breast cancer patient, Dr. Ackerman and colleagues create a proton therapy and a conventional photon therapy plan to compare which course of treatment will be most efficient and effective (Figure 1).
“We generate comparative dosimetry plans with IMRT, proton beam and conformal radiation therapy to look at the dose delivered to the brachial plexus, lungs and heart,” Dr. Ackerman explains. “We also look at homogeneity in the patients we want to treat. Frequently, proton therapy is the superior plan, with lower radiation dose to the heart and significantly lower dose to the lungs.”
Women seek out Ackerman Cancer Center because they believe that proton therapy may offer them better care than traditional radiation for treating breast cancer, he adds. “Every 100 centigray of dose to healthy organs such as the heart can increase the risk for long-term complications, including coronary artery disease and congestive heart failure,” Dr. Ackerman says. With increased life expectancy, even if these complications manifest over 20 years, a marginal reduction in mean dose to healthy organs and tissues can be important considerations for women seeking treatment.
Proton therapy is generally considered for breast cancer patients receiving radiation therapy in the adjuvant setting. That said, every patient has a unique anatomy, and some physiological compositions make proton therapy more advantageous, such as in a woman whose heart is close to the chest wall, says Dr. Ackerman. This is because the protons “stop” before reaching the critical organ.
Dr. Ackerman uses daily image guidance as well as surgical clips placed at the time of lumpectomy to help manage uncertainty. “We use robust planning with each beam covering the entire treatment volume,” he explains. “We perform simulation 2 times, with the patient on the table, off, and then on again with a qualitative and a quantitative check to be sure the position is reproducible.”
The women are also directed to do shallow breathing, and the second simulation is performed to visualize movement of the chest wall and lungs.
Currently, the proton therapy system at Ackerman Cancer Center performs passive scanning, and the center has contracted for a second Mevion system featuring pencil-beam technology delivered at hyperspeed for spot scanning. “This system will allow us to treat more challenging cases, such as ones with lymph node involvement,” Dr. Ackerman says. It may also reduce treatment times.
Dr. Ackerman’s advice for clinicians interested in providing proton therapy is to invest sufficient time, energy and effort to train technical staff on the nuances of proton therapy. In addition to training off-site, Dr. Ackerman’s radiation therapists, dosimetrists, and medical physicists generated, reviewed and practiced comparative plans for 6 months before treating their first proton patient.
“It is also important to have well-trained support services, from oncology-certified nurses to social workers and nutritionists,” Dr. Ackerman adds. “As oncologists, we should be committed to treating the whole patient. This includes a focus on the patient’s wishes for effective treatment and positive long-term outcomes. The strength of [our] technology unlocks that opportunity for us in treating breast cancer with proton therapy.”
Reference
- Bush DA, Do S, Lum S, et al. Partial breast radiation therapy with proton beam: 5-year results with cosmetic outcomes. Int J Radiat Oncol Biol Phys. 2014;90(3):501-505.
Citation
MB M. Technology Trends: Proton Therapy Treatment for Breast Cancer. Appl Radiat Oncol. 2015;(4):31-32.
December 28, 2015