Substituting stereotactic body radiation therapy boost for brachytherapy in Mayo protocol for peri-hilar cholangiocarcinoma
Images

CASE SUMMARY
This is a 56-year-old man with history of primary sclerosing cholangitis (PSC) who was found to have an elevated CA 19-9 of 126 units/mL. Endoscopic retrograde cholangiopancreatography (ERCP) revealed atypical cells suspicious for adenocarcinoma, and fluorescent in situ hybridization (FISH) demonstrated a gain of 1q21 and deletion of 9p21. MRI revealed a dominant stricture of the right hepatic duct without a discrete mass, and a few mildly prominent peri-portal lymph nodes. A clinical diagnosis of peri-hilar cholangiocarcinoma was made based on the combination of a dominant stricture and FISH findings.1 The peri-portal lymph node was sampled by endoscopic ultrasound revealing benign lymphocytes, and was presumed to be reactive secondary to PSC. His lesion was determined to be unresectable given bilateral intrahepatic duct involvement. Following multidisciplinary discussion, neoadjuvant chemoradiation followed by orthotopic liver transplant was recommended. The patient received external-beam radiation therapy (EBRT) (45 Gy in 30 fractions delivered twice daily [BID]) with concurrent capecitabine. He was subsequently scheduled for intrabiliary high-dose rate (HDR) brachytherapy; however, due to a long-standing PSC-related stricture and subsequent severe atrophy of the right liver, the right intra-hepatic duct could not be traversed by percutaneous transhepatic cholangiography. Thus, a stereotactic body radiation therapy (SBRT) boost (30 Gy in 3 fractions) was utilized to approximate the planned brachytherapy dose (Figure 1). One week following completion of the SBRT boost he underwent exploratory laparotomy to verify the absence of extra-hepatic disease, and was subsequently listed for transplant. He resumed capecitabine and ultimately underwent planned living donor total liver transplant 3 months after completing chemoradiation. Pathology revealed moderately differentiated adenocarcinoma involving the common hepatic duct and extending proximally to the confluence of the left and right hepatic ducts with invasion into adjacent liver parenchyma. The volume of residual disease was unable to be assessed as the tumor could not be differentiated grossly from extensive scarring. Surgical margins were negative for malignancy (though positive for high-grade dysplasia), and 0 of 3 lymph nodes were involved.
IMAGING FINDINGS
Magnetic resonance cholangiopancreatography (MRCP) demonstrated a stricture of the right hepatic duct without a discrete mass.
DIAGNOSIS
Peri-hilar cholangiocarcinoma (CCA)
DISCUSSION
Cholangiocarcinoma is a rare neoplasm arising from the epithelial cells of the bile ducts. Approximately 95% of cholangiocarcinomas involve the extra-hepatic bile ducts and, of those, 60% to 70% arise from the peri-hilar region. Patients with primary sclerosing cholangitis have a lifetime risk of CCA of 10% to 15%.2
The primary means of achieving a cure is a negative-margin resection, although this can be achieved in < 30% of patients due to bilateral liver parenchymal involvement, vascular invasion, or poor hepatic functional reserve. Thus, the search for better treatment alternatives has led to transplantation, which provides wide surgical margins and addresses underlying liver dysfunction.
Early experiences with transplantation alone were disappointing with 5-year overall survival < 30%.3 To improve outcomes, the University of Nebraska pioneered neoadjuvant chemoradiation followed by liver transplantation.4 This approach was later adopted by the Mayo Clinic utilizing neoadjuvant chemoradiation followed by transplant in carefully selected patients with early stage cholangiocarcinoma that is either unresectable or in the setting of primary sclerosing cholangitis. Outcomes were encouraging with 5-year survival rates of 71%,5 with a subsequent series verifying reproducibility of these outcomes in a multi-institutional setting.6 Given these excellent outcomes, some have even advocated for this approach in resectable patients, although this would be a resource-intensive approach overall.7
On this neoadjuvant protocol, patients are treated initially with EBRT (45 Gy at 1.5 Gy/fraction BID with concurrent 5-fluorouracil-based chemotherapy to an initial volume including the primary tumor plus margin and first echelon lymph nodes. At 2 to 3 weeks after completion of EBRT, an intraluminal brachytherapy boost (20-30 Gy initially utilizing low-dose-rate radium sources prescribed to 1 cm over the course of 1-2 insertions) was delivered to encompass the tumor and a 1.5- to 2-cm margin above and below the radiographic extent of disease. Currently, most institutions substitute high-dose rate brachytherapy with a variety of fractionation schedules to approximate the original low-dose rate prescription. Our practice is to utilize 5 Gy per fraction over 3 fractions prescribed to 1 cm depth given BID. After brachytherapy, patients undergo exploratory laparotomy to confirm lack of extra-hepatic disease and, after recovery, resume oral capecitabine until transplantation.
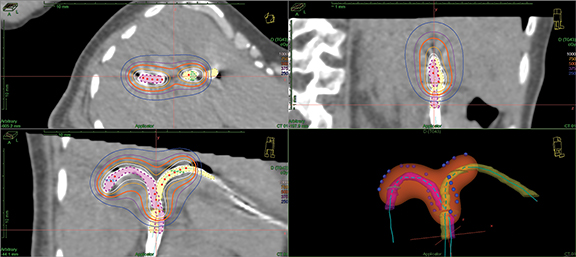
Intraluminal brachytherapy is an important component in the treatment of cholangiocarcinoma. The advantage of intraluminal brachytherapy over EBRT is the ability to deliver high doses to the tumor, while maximally sparing normal tissue due to rapid dose falloff. Brachytherapy has a very different dose distribution than EBRT with extremely high doses at the surface, and rapid fall-off based on the inverse square of the distance from the source. In selecting our SBRT prescription dose of 10 Gy per fraction, we sought to approximate the brachytherapy surface dose, which is an estimate of the dose to a small biliary tumor after downsizing from EBRT. Admittedly this is not precise and varies depending on catheter orientation and the thickness of the tubing used. As noted in Figure 2, the 10 Gy line lies for the most part just beyond the surface of the HDR tubing. Given the significant heterogeneity of brachytherapy (small portions of the bile duct surface appear to receive significantly > 10 Gy per fraction) we did not emphasize uniformity of SBRT dosing in our treatment plan. While SBRT can achieve relatively rapid dose fall-off, it is certainly not as rapid as that achieved by brachytherapy and, as such, the volume of the 10 Gy dose using SBRT is significantly higher in our plan than it would have been with brachytherapy. We presumed that this would be safe given that the toxicity of a serial structure such as the bile duct should be driven more by point doses and hot spots leading to risk of stricture rather than volume. Some parameters for central biliary dose and toxicity have been reported and, of note, cholangiocarcinoma patients are at highest risk given direct tumor involvement of the bile ducts;8 however, these are not uniformly consistent across reports. Acute toxicity to the ducts appeared modest in the published neoadjuvant series5,6 noted above, albeit with prophylactic stenting for most patients and subsequent transplant to prevent further late toxicity. Furthermore, a single retrospective series reported primary SBRT for the full course of treatment in 12 patients with peri-hilar cholangiocarcinoma (50-60 Gy in 3-5 fractions) prior to transplant, with 6 of those patients reaching transplant with an acceptable 1-year survival rate of 83% at the time of publication.9
SBRT has potential advantages including greater generalizability, minimal invasiveness, and greater simplicity compared to percutaneous transhepatic cholangiography (PTHC) placement and intraluminal brachytherapy. However, given the limited body of data using SBRT prior to transplant for peri-hilar cholangiocarcinoma, our preference remains to use conventional EBRT and a brachytherapy boost as described in the Mayo and multi-institutional series when feasible.
CONCLUSION
Peri-hilar cholangiocarcinoma portends a poor prognosis. Traditionally, resection has offered the highest chance of cure, although unfortunately most patients are unresectable due to frequent involvement of both bile ducts due to tumor location. Neoadjuvant chemoradiation with brachytherapy boost followed by liver transplantation has led to promising survival outcomes in early published experiences, perhaps even surpassing surgery alone. For patients in whom brachytherapy boost is not feasible, SBRT may provide a safe and effective alternative.
REFERENCES
- Dehaan R, Kipp B, Smyrk T, Abraham S, Roberts L, Halling K. An assessment of chromosomal alterations detected by fluorescence in situ hybridization and p16 expression in sporadic and primary sclerosing cholangitis-associated cholangiocarcinomas. Hum Pathol. 2007;38(3):491-499. doi:10.1016/j.humpath.2006.09.004
- Rizvi S, Gores GJ. Pathogenesis, diagnosis, and management of cholangiocarcinoma. Gastroenterology. 2013;145(6):1215-1229. doi:10.1053/j.gastro.2013.10.013
- Becker NS, Rodriguez JA, Barshes NR, O’Mahony CA, Goss JA, Aloia TA. outcomes analysis for 280 patients with cholangiocarcinoma treated with liver transplantation over an 18-year period. J Gastrointest Surg. 2008;12(1):117-122. doi:10.1007/s11605-007-0335-4
- Sudan D, DeRoover A, Chinnakotla S, et al. Radiochemotherapy and transplantation allow long-term survival for nonresectable hilar cholangiocarcinoma. Am J Transplant. 2002;2(8):774-779. http://www.ncbi.nlm.nih.gov/pubmed/12243499. Accessed August 5, 2019.
- Rosen CB, Heimbach JK, Gores GJ. Surgery for cholangiocarcinoma: the role of liver transplantation. HPB (Oxford). 2008;10(3):186-189. doi:10.1080/13651820801992542
- Darwish Murad S, Kim WR, Harnois DM, et al. Efficacy of neoadjuvant chemoradiation, followed by liver transplantation, for perihilar cholangiocarcinoma at 12 US centers. Gastroenterology. 2012;143(1):88-98.e3; quiz e14. doi:10.1053/j.gastro.2012.04.008
- Ethun CG, Lopez-Aguiar AG, Anderson DJ, et al. Transplantation versus resection for hilar cholangiocarcinoma. Ann Surg. 2018;267(5):797-805. doi:10.1097/SLA.0000000000002574
- Toesca DAS, Osmundson EC, Eyben R von, et al. Central liver toxicity after SBRT: an expanded analysis and predictive nomogram. Radiother Oncol. 2017;122(1):130-136. doi:10.1016/j.radonc.2016.10.024
- Welling TH, Feng M, Wan S, et al. Neoadjuvant stereotactic body radiation therapy, capecitabine, and liver transplantation for unresectable hilar cholangiocarcinoma. Liver Transpl. 2014;20(1):81-88. doi:10.1002/lt.23757
Citation
J B, S S, K B, K S. Substituting stereotactic body radiation therapy boost for brachytherapy in Mayo protocol for peri-hilar cholangiocarcinoma. Appl Radiat Oncol. 2019;(3):43-45.
September 4, 2019