Spontaneous Pregnancy Following Pelvic Irradiation for Anal Cancer: A Case Report
Images
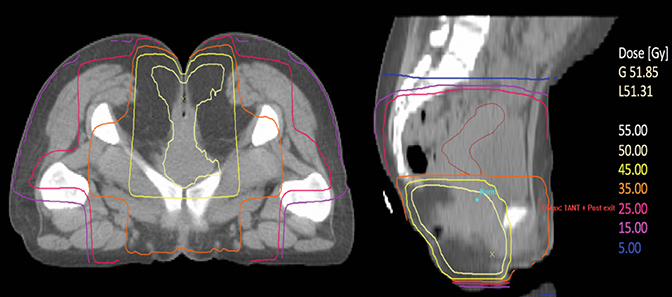


CASE SUMMARY
A 26-year-old woman, para 1 and on triple therapy for HIV, was treated with pelvic radiation therapy to 50.4 Gy (30.6 Gy/17 fractions [phase 1], 19.8 Gy/11 fractions [phase 2]). The left ovary mean dose was 1 Gy, right ovary 31.5 Gy, and uterus 34.5 Gy (Figure 1, 2) with concurrent 5-FU and mitomycin C for grade 1, cT1N0M0 squamous cell cancer of the anus. Her left ovary was transposed into the abdomen prior to treatment to maintain premenopausal status. Her right ovary received more than double the effective sterilizing dose of radiation. Her treatment response was excellent with no evidence of disease on post-treatment imaging. Somewhat unexpectedly, she continued to menstruate with a regular cycle post-treatment. Hysteroscopy to investigate this bleeding demonstrated a small uterine cavity of normal appearance. Endometrial curettings were normal histologically. Colposcopy was also normal. Five years later she spontaneously conceived and vaginally delivered a small-for-dates, but otherwise healthy baby, requiring a significant episiotomy during delivery.
IMAGING FINDINGS
Transverse and sagittal plane images of planning computed tomography (CT) demonstrate radiation dose delivered to pelvic structures (Figure 1A). Images of radiation therapy planning CT with both ovaries outlined show that the left ovary has been transposed into the abdomen prior to commencing radiation therapy (Figure 1B). Dose-volume histogram for ovaries and uterus is shown in Figure 2.
DIAGNOSIS
Grade 1, cT1N0M0 squamous cell cancer of the anus
DISCUSSION
Anal cancer is associated with the human papillomavirus (HPV) and is recognized as a common non-AIDS-defining cancer in HIV-positive individuals.1 Chemoradiotherapy using 5-FU and mitomycin has been the standard treatment for anal cancer since the Anal Cancer Trial (ACT1) trial.2
Unfortunately, the dose of radiation therapy used to treat anal cancer is generally high enough to sterilize the ovaries. Furthermore, current trials are using higher doses of pelvic irradiation (to larger volumes) in certain anal cancers (ACT5 of PLATO);3 thus, the probability of conception post-treatment will reduce even further. The Faddy-Gosden model, which is a mathematical model of natural oocyte decline, has estimated that a dose of < 2 Gy will destroy half of the immature oocytes following pelvic irradiation.4 The effective sterilizing dose (ESD) is the dose of fractionated radiation therapy that causes premature ovarian failure immediately after treatment in 97.5% of patients. Naturally, the ESD decreases with increasing age at the time of treatment, due to physiological factors of female reproduction. The ESD at birth is 20.3 Gy, decreasing to 16.5 Gy at 20 years and 14.3 Gy at 30 years.5
Previous studies regarding changes seen on dynamic contrast-enhanced MRI in premenopausal women undergoing pelvic RT have demonstrated that the blood vessels of the irradiated uteri contained marked circumferential intimal thickening and decreased luminal diameter. Furthermore, changes in cervical length, loss of uterine junctional zone anatomy and myometrial atrophy and fibrosis were seen following RT treatment.6 Although this patient was not offered the use of a vaginal dilator, it is something that should be considered as part of treatment in all women receiving pelvic radiation therapy.7 Studies are underway to improve compliance with dilator use.8
Three factors in this case are interesting due to the rarity of their occurrence. First, the probability of spontaneous conception was extremely low considering this patient received more than twice the effective sterilizing dose to her right ovary (ESD at 26 years old is approximately 15 Gy) during treatment for her anal cancer.5 Furthermore, although a previous hysteroscopy in this patient had described a small uterine cavity, she successfully carried the fetus to 36 weeks of gestation. It is unclear as to how the uterus was able to maintain a fetus for this length of time, but we can only presume that the dose it received (mean 34.5 Gy) was, in this woman, below the threshold beyond which significant vascular compromise occurred. Finally, her vaginal delivery of a 3.5-pound baby is remarkable considering that radiation therapy often renders the vaginal canal fibrosed and stenosed. We presume that the transposed ovary helped maintain this patient’s premenopausal state but it was the ovary left within the pelvis that must have released the egg that was fertilized.
To our knowledge, this is the only reported case of a woman having a vaginal delivery following pelvic radiation therapy for anal cancer. The only other published case describes a woman who became pregnant but underwent elective caesarean section due to vaginal stenosis.9 Wald et al describe a spontaneous twin pregnancy following radiation therapy for rectal cancer; however, one child died due to premature labor with a complicated delivery.10 A case of a woman successfully conceiving and delivering vaginally following radiation therapy to the rectum for rectal MALT lymphoma has also been described.11 Although the ESD causes premature ovarian failure in 97.5% of patients, it is clear that in extremely rare cases, some ovarian function can remain and subsequent pregnancies can occur.
CONCLUSION
Transposing an ovary (or two) outside of the pelvis should be considered in all premenopausal women prior to anal cancer radiation therapy. The primary aim of this would be to maintain premenopausal status and minimize complications associated with premature ovarian failure. Despite the case highlighted here, it would be difficult to argue that one ovary should remain (attached to its fallopian tube) within the pelvis to maintain fertility. This case highlights the need to remind patients that, although rare, pregnancy is still possible after pelvic radiation therapy and contraception should be considered where necessary. Furthermore, both gestation and delivery carry high risk to the fetus due to the radiation effects on the pelvic anatomy and, if pregnancy occurs, close collaboration between obstetrics and oncology is essential and a vaginal delivery should be avoided.
REFERENCES
- Wang C-CJ, Sparano J, Palefsky JM. Human immunodeficiency virus/AIDS, human papillomavirus, and anal cancer. Surg Oncol Clin N Am. 2017;26:17-31.
- Party UACTW. Epidermoid anal cancer: results from the UKCCCR randomised trial of radiotherapy alone versus radiotherapy, 5-fluorouracil, and mitomycin. Lancet. 1996;348:1049-1054.
- ISRCTN registry. ISRCTN88455282 PLATO-Personalising anl cancer radiotherapy dose. Accessed August 14, 2020. https://doi.org/ 10.1186/ISRCTN88455282
- Wallace WHB, Thomson AB, Kelsey TW. The radiosensitivity of the human oocyte. Human Reprod. 2003;18:117-121.
- Wallace WHB, Thomson AB, Saran F, Kelsey TW. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int J Radiat Oncol Biol Phys. 2005;62:738-744.
- Milgrom SA, Vargas HA, Sala E, Kelvin JF, Hricak H, Goodman KA. Acute effects of pelvic irradiation on the adult uterus revealed by dynamic contrast-enhanced MRI. Br J Radiol. 2013;86:20130334.
- American Cancer Society. Treating Anal cancer. radiation therapy for anal cancer. Accessed September 23, 2020. https://www.cancer.org/cancer/anal-cancer/treating/radiation-therapy.html
- NIH U.S. National Library of Medicine ClinicalTrials.gov. Study of vaginal dilator use after pelvic radiotherapy. ClinicalTrials.gov Identifier: NCT00789893. Updated June 11, 2015. Accessed September 23, 2020. https://www.clinicaltrials.gov/ct2/show/NCT00789893
- Hurmuz P S-MD, Byrne P, Cooper R. Succesful spontaneous pregnancy after pelvic chemoradiotherapy for anal cancer. Clin Oncol. 2012;24:452-458.
- Wald K, Easterling T, Swisher EM. Spontaneous twin pregnancy after oophoropexy and pelvic radiation for rectal cancer. Obstet Gynecol. 2016;128:792-794.
- Hatayama Y, Masahiko A, Kawaguchi H, Hirose K et al. Safe and successful birth following pelvic radiotherapy for rectal muchosa-associated lymphoid tissue lymphoma: a case report. J Med Case Rep. 2017;11(26).
Citation
C M, C G. Spontaneous Pregnancy Following Pelvic Irradiation for Anal Cancer: A Case Report. Appl Radiat Oncol. 2020;(4):49-51.
December 24, 2020