Reducing errors in radiation therapy through electronic safety checklists
Images


For decades, radiotherapy (RT) has been an effective treatment in saving and prolonging life for many cancer patients, but medical errors from radiation treatment can be fatal. For example, overdosing patients through RT has been reported to be lethal.1 While the error rate in patients treated with RT has been as low as 0.005%, one death is one too many.2
The World Health Organization (WHO) in combination with the International Atomic Energy Agency (IAEA) published a review in 2008 titled, “Radiotherapy Risk Profile.”3 In this document they describe that from 1976 to 2007, 3,125 reported patients were affected by RT incidents that led to adverse events. This literature noted that 1% (n=38) of the patients affected by RT incidents eventually died due to radiation toxicity.3 Per WHO’s review, the majority of errors were caused by a communication failure. After classifying where the errors occurred, they discovered that the majority of errors (38%; n=1,732) were related to transfer of information, while 18% (n=844) occurred during actual treatment delivery, and only 9% occurred during the treatment planning stage (n=420). The remaining 35% of the incidents were due to a combination of events during the planning process.
While reducing errors in radiation oncology should be a simple process, the reality is that it is a multistep process.4 Treatment of a single patient requires contributions from the nurse, physician, computed tomography (CT) simulation staff, dosimetrist, physicist and radiation therapist. Considering the many steps to delivering RT, a single error can be propagated throughout multiple steps of the process. Likewise, there are multiple opportunities to detect an error because of the multistep nature of the process.
While RT errors can be attributed to machine or software errors, the majority of errors are attributable to humans. The United States Nuclear Regulatory Commission (NRC) has recorded that of all reported RT incidents, about 60% or more are due to human error.5 These data suggest that most errors can be prevented if human errors can be prevented or caught early in the process.
An “incident” is defined by the IAEA safety standards as any unintended event that has consequences that are not negligible from the point of view of protection or safety, whereas a “near miss” is a potential significant event that did not occur owing to the facility conditions prevailing at the time.6 If “incidents” can be converted to “near misses” or good catches, then patients can be saved from harm.
Safety checklists have been implemented in different arenas to reduce human errors through duplication lists or safety timeouts. They have been implemented in the airline industry, NASA engineering, and operating rooms, and have proven successful in reducing human errors.2 For example, when used in surgery, they have been shown to reduce inpatient complications and deaths. In a study published in the New England Journal of Medicine by Haynes et al. titled, “Surgical Safety Checklist to Reduce Morbidity and Mortality in a Global Population,” checklists were enforced inn8 hospitals across 8 different cities. The study investigators demonstrated that checklist implementation reduced the rate of inpatient death after surgery from 1.5% to 0.8% along with the number of inpatient complications from 11% to 7%.7
The purpose of this project was to implement an electronic safety checklist program into the workflow of an academic radiation oncology department.
Technology
The implementation of our safety checklist program took about 6 months from origination of the idea to launching the software for department use. We started by forming a team that included a physicist, a therapist, a radiation oncologist, a radiation oncology resident, and a graduate physics student. This team then reviewed all of the errors that had been recorded in our electronic error-reporting system and classified them according to where the error originated. We then created a checklist for each area in our department by including the items that were most commonly missed according to our analysis of the reported errors. Checklists were made for CT simulation, physicians, dosimetrists, physicists and radiation therapists. We reviewed the checklists as a team and reduced the number of checklist items even further with the goal of creating short, powerful checklists for each area to maximize the impact of each checklist.
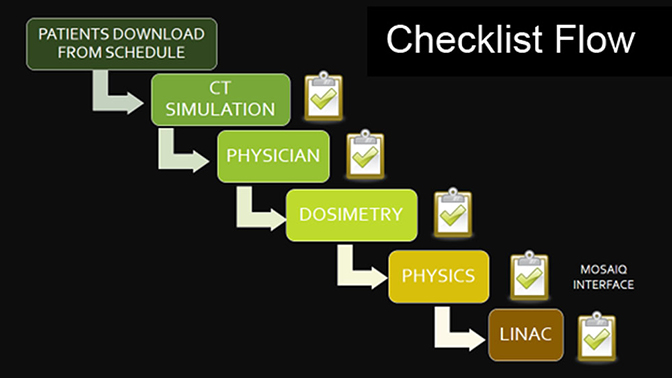
To determine how to best integrate the checklists into our workflow, we diagramed the workflow from CT simulation to the start of radiation therapy (Figure 1). The ideal checklist program would automatically generate a list of patients scheduled for CT simulation that day. The first checklist to be completed would be the CT simulation checklist. Once completed, the patient’s plan would then advance into the queue of the subsequent checklist area, from the physician to dosimetry, physics, and then the therapists at the treatment machine. At each of these steps, the checklist would be completed before the patient’s plan could progress to the next step.
To best integrate our plan into practice, we developed software written in VB.NET using a serial workflow based on a checklist philosophy used in vertically integrated manufacturing. The software identified and tracked the completion of tasks appropriate to each patient’s treatment, including generation of documentation and multiple/parallel monitoring points. This software was integrated into MOSAIQ (Elekta, Stockholm, Sweden), a common electronic medical system used in radiation oncology, to auto-deposit the generated documents that indicate the listing status of required tasks for each staff member. Microsoft Outlook API (Microsoft Corporation, Redmond, Washington) was used for communication among staff and to coordinate issue resolution through email or text messaging.
The main view of the checklist software screen is shown in Figure 2 demonstrating what the safety checklist software program looks like when it is first opened. The patient list auto-populates from the Mosaiq CT simulation schedule each day, so there is no need to manually enter a patient’s name into the system. This patient list is the work queue for the CT simulation technician. Each step of the process from CT simulation to radiation therapy start has a work queue generated by the completion of the checklist at the prior step. For example, once the CT simulation group has completed its patient checklist for Patient 1, this patient will automatically show up on the physician’s work queue in the checklist program, notifying the physician that Patient 1 is ready for contouring. Once the physician has completed contouring and written a radiotherapy prescription, he or she can then select Patient 1 from his or her list and complete the checklist on Patient 1.
Once the physician completes the checklist, Patient 1 appears on the dosimetry work queue, notifying dosimetry that Patient 1 is ready for treatment planning. When the radiotherapy plan for Patient 1 has been completed and reviewed by the attending physician, the dosimetrist completes his or her checklist, and Patient 1 appears on the Physics work queue. The physicist then knows that the plan for Patient 1 is ready to be checked. After the plan is checked, the physicist completes the checklist and Patient 1 then appears on the radiation therapy work queue. This work queue notifies the therapists on each machine that the plan for Patient 1 is ready to be checked.
The checklist program software creates a date and time stamp when each checklist is completed, allowing us to track how long the patient’s record has spent in each area of the treatment process. The software includes time analysis functionality to analyze the completion times. Our hope is to eventually use this data to help speed up our treatment planning process.
Clinical application
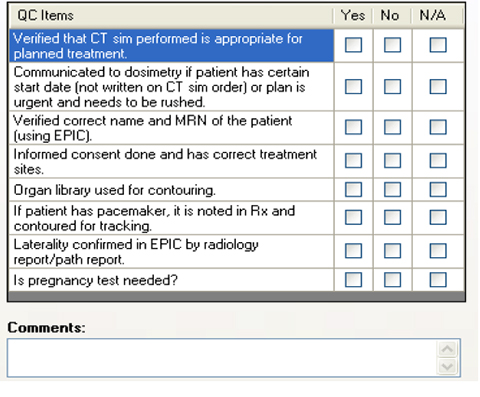
The clinical utility of the electronic safety checklist program became evident early on. Within the first weeks of going live, we caught several potentially serious errors. These were near misses that were not reported in the error-reporting system because they were caught by the checklist program at the very beginning of the planning process. For example, a physician working on the safety checklist for a female patient of childbearing age noticed she had not taken a pregnancy test. The treating physician ordered a pregnancy test, which revealed that the patient was pregnant even though the patient denied that as a possibility on initial consultation. Reminding the physician to check the pregnancy status of a woman of childbearing age prevented a serious error. In another example, a radiation prescription was written a few weeks into the implementation of the checklist program. The physician typed the prescription to specify the treatment site as the left neck. While completing the checklist, the physician noticed that the right neck had been contoured as it was the pathologic side of disease. This obligatory double-check that took less than 2 minutes of the physician’s time potentially averted a serious error. To review other important items on our safety checklists, see the physician and therapist checklists in Figure 3.
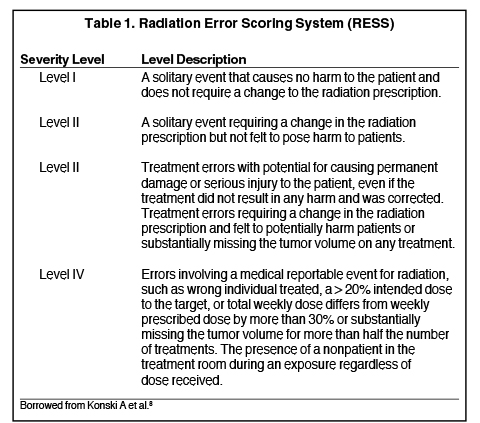
The number of errors caught before reaching the patient (which we call near misses or “good catches”) is growing in our department. The severity of errors was graded according to the Radiation Error Scoring System shown in Table 1.8 In this system, grade 1 and 2 errors are classified as near misses (or events that cause no harm to the patient as defined in the RESS), and grade 3 and 4 errors are those reaching the patient. This is by no means an ideal grading system, but we found that it is better suited for radiation oncology than other error grading systems. We noticed that the number of reported errors increased over time, including after the implementation of the safety checklists; we anticipate that the number of errors actually reaching the patient (grade 3 and 4 errors) is decreasing. Our early experience demonstrates that the number of good catches increased after the safety checklist program was implemented, and the number of serious treatment errors or “incidents,” as defined by the IAEA, decreased.
Conclusion
Safety and quality are extremely important to treating cancer patients not only in our radiation oncology department but throughout the nation. It took over 6 months to implement a new electronic safety checklist program. This checklist system has been successfully implemented in our department, identifying and improving clinical and communication issues. Following implementation, we found that the system helped reduce regulatory and treatment documentation compliance events, identify communication problems, and empower staff to submit “good catch” issues to a team working to improve workflow, improve treatment quality, and improve safety. The program also enabled us, through time analysis, to easily identify and improve treatment-related bottlenecks.
Not only did the electronic checklist system benefit the overall clinical workflow in regard to treatment planning, it also resulted in an increase in reported errors (good catches). There was a trend toward reducing the severity of errors (more reported “near misses,” fewer errors reaching the patient), although more time is needed to determine if the safety checklists actually reduce the number of errors reaching patients.
References
- Bogdanich W. Radiation offers new cures, and ways to do harm. New York Times. January 23, 2010.
- Albuquerque KV, Miller AA, Roeske JC. Implementation of electronic checklists in an oncology medical record: initial clinical experience. J Oncol Pract. 2011;7(4):222-6.
- World Health Organization (WHO). Radiotherapy Risk Profile. 2008. Geneva.
- Sternick ES. Development of a comprehensive radiation oncology quality and safety program. Front Oncol. 2014;4:30.
- Duffey RB, Saull JW. Know the Risk- Learning from Errors and Accidents: Safety and Risk in Today’s Technology. Oxford, United Kingdom: Butterworth-Heinemann 2002.
- International Atomic Energy Agency. IAEA safety glossary: terminology used in nuclear safety and radiation protection. Vienna, Austria: International Atomic Energy Agency; 2007.
- Haynes AB, Weiser TG, Berry WR, et al. A surgical safety checklist to reduce morbidity and mortality in a global population. N Engl J Med. 2009;360(5): 491-499.
- Konski A, Movsas B, Konopka M, Ma C, Price R, Pollack A. Developing a radiation error scoring system to monitor quality control events in a radiation oncology department. J Am Coll Radiol. 2009;6(1):45-50.