Radiation Therapy Techniques in the Management of Locally Advanced, High-Grade, Soft-Tissue Sarcoma
Images
Abstract
Objective: We sought to assess which prognostic factors are associated with local recurrence (LR) rates and wound complications of locally advanced, high-grade, soft-tissue sarcoma (STS).
Methods: Retrospective analysis was performed on patient data from 2005 to 2018, with high-grade STS of extremity or trunk, > 5 cm, histology-specific, with central pathology review. Wide-excision was performed in 100 patients along with radiation, whose radiation fields and dose plans were available for review, of which 31 also received ifosfamide-doxorubicin, with a minimum follow-up of 1 year. Multivariable analysis (MVA) of prognostic factors was calculated to see which variables were associated with LR, and nonhealing wound rates.
Results: Median follow-up was 5.8 years. Univariate analysis revealed that trunk location, distal and radial clinical-target-volume (CTV) margins of 1.5 cm had higher rates of LR vs ≤ 2 cm or presence of fascial boundary. MVA of these prognostic factors revealed that trunk location (P = 0.048), and radial CTV of 1.5 cm (P = 0.006) were independently associated with higher LR, as 10 of 15 LRs were at the edge of the radial margin. The bolus did not affect LR. The odds ratio for non-healing wounds at 3 months was higher for subcutaneous (T2a) disease, larger tumor size, proximal CTV ≤ 2 cm, wider field size, bolus technique, and lack of chemotherapy.
Conclusion: Longitudinal CTV margins of 3 cm seem adequate, but high-grade STS ≤ 5 cm may benefit from increased radial CTV margins of 2 to 2.5 cm in the absence of a fascial boundary, although larger CTV may increase nonhealing wound rates. Bolus techniques may increase wound complications in T2a-b STS and should not be routinely employed.
Keywords: soft-tissue sarcoma, radiation therapy, bolus, margins, complications
Introduction
Limb-salvage therapy using external-beam radiation therapy (EBRT) to reduce local recurrence (LR) allows surgeons to perform wide-excision of soft-tissue sarcoma (STS), allowing less radical surgery such as amputation. 1,2 In 2011, the Radiation Therapy
Oncology Group (RTOG) published guidelines for designing clinical target volume (CTV) margins in the treatment of extremity soft-tissue sarcoma (ESTS), which was verified by subsequent publications, where longitudinal proximal/distal CTV recommendations were 3 to 5 cm, but radial CTV was 1.5 cm. 3-6 One question was whether these guidelines can be applied to STS involving subcutaneous tissues, and the trunk, where space is more limited. Large treatment volumes can increase a patient’s risk of long-term complications. 7 However, this must be balanced against risks of LR when treatment volumes are too small, and thus our goal was to assess prognostic factors associated with higher LR rates and wound complications of locally advanced, high-grade, STS, and see which CTV margins may be optimal.
Methods
Retrospective analysis was performed on data from 100 adult patients from January 2005 to December 2018, who had primary high- grade STS of the extremity or trunk, > 5 cm, localized to the muscle or subcutaneous tissue, that were either synovial (16 patients), dedifferentiated liposarcoma (16), myxofibrosarcoma (13), round cell liposarcoma (7), undifferentiated pleomorphic sarcoma (39), or undifferentiated sarcoma not otherwise specified (9). We defined locally advanced as > 5 cm in size. Patients were T2 if the tumor was > 5 cm in maximal dimension, T2a for subcutaneous tumor, and T2b for muscle-invasive tumor. 8 Patients had to have their radiation fields and dose plans available for review, with a minimum of 1 year of follow-up. Bone sarcomas, gastrointestinal stromal tumors, pediatric sarcomas, and retroperitoneal sarcomas were excluded from this analysis. Central pathology review was performed by our soft-tissue pathologist (S.S.) and graded according to the Federation Nationale des Centres de Lutte Contra le Cancer (FNCLCC); 9 our study included only grade 3 or high-grade sarcomas. Radiation therapy was delivered using megavoltage photons, using either 6 MV or 15 MV, with the majority (78%) undergoing preoperative radiation to 4400-5000 cGy in 200 cGy fractions to the gross tumor with margin, followed by wide excision, which occurred about 6 weeks after radiation. Twenty-two percent underwent postoperative radiation to 4500-5040 cGy with a shrinking field boost to a total of 5940-6660 cGy in 180-200 cGy fractions. Additional boost was not performed on preoperative radiation patients who had a positive margin after wide excision. 10 All patients underwent immobilization using a polystyrene-filled vacuum cradle. Gross tumor volume (GTV) contours included T1-weighted images with gadolinium enhancement of the tumor on MRI, and T2-weighted edema included in the CTV. CTV volumes were determined by the prescribing physician. CTV to planning target volume (PTV) expansion was 1 cm, as image guidance was not performed. A bolus was used in select cases at the discretion of the prescribing physician due to concern about scar/cutaneous recurrence. During the timeframe these patients were treated, there was no effort to treat the skin as an organ at risk (OAR). Three-dimensional conformal radiation therapy (3DCRT) was performed in 91 patients, and intensity-modulated radiation therapy (IMRT) was performed in 9 patients. The radiation planning images (digitally reconstructed radiographs) and isodose plans were scaled and compared to MRI evidence of LR, which was categorized as out of field if > 80% of the volume was outside of the irradiated volume, in field if > 80% of the volume was inside the irradiated volume, and marginal if recurrence was at the edge of the irradiated volume between the out-of-field and in-field parameters, listed above. Binary classifica- tion of CTV margins was performed. The first was a CTV of 1.5 cm. The second was a CTV margin ≥ 2 cm or if the CTV extended to a fascial boundary, as a fascial boundary is equivalent to a wide margin. For postoperative patients, CTV margins were recorded based on the original fields taken to 4500-5040 cGy, and not based on the boost margins.
The Charlson Comorbidity Index score is a method of categorizing comorbidities of patients based on the International Classification of Diseases, and was assessed for each patient, and tabulated in Table 1. 11 After therapy, patients without wound complications were generally followed every 6 months for 5 years, and yearly thereafter, with MRI of the local area and computed tomography (CT) of the chest.
Date of surgery was established as time zero, and Kaplan-Meier estimates were used to calculate local recurrence-free survival (LRFS). 12 Characteristics of patients were reported as percentages for categorical factors, and median with ranges was used for continuous factors. Fisher’s exact tests were used to test for categorical differences in treatment groups. Wilcoxon rank-sum tests were used to calculate differences on continuous factors. Multivariable analysis (MVA) of prognostic factors using the Cox proportional hazards models was used to estimate hazard ratios on LR. 13 Multivariate logistic regression was performed and odds ratios were calculated to detect the factors associated with the probability of wound complications or the presence of a wound vacuum device at 3 months after surgery. The forest plot was produced to display the results graphically. The Wald test was used to calculate P value for the odds ratio. 14 Statistical significance utilized a 2-sided P < 0.05. All analyses were conducted using SAS EG 7.13 (SAS Institute Inc.).
Results
Median follow-up was 5.8 years (range 1.0 to 15.0 years). Ifosfamide-doxorubicin was given in 31 patients, and 69 received radiation therapy alone as adjuvant therapy. Ifosfamide-doxorubicin was usually prescribed neoadjuvantly for 5 cycles followed by radiation, then followed by wide excision; whereas for postoperative radiation, ifosfamide-doxorubicin was administered after the completion of radiation. All patients underwent en bloc wide excision, but 6 patients had unintended excision, where the surgeon had violated the tissue planes. These 6 patients subsequently underwent en bloc wide re-excision.
Cohort characteristics using univariate comparisons revealed that trunk location (P = 0.001), longitudinal-distal CTV 1.5 cm (P = 0.019), and radial CTV 1.5 cm (P < 0.001) were associated with higher rates of LR, but longitudinal-proximal CTV 1.5 cm did not quite make statistical significance (P = 0.058) (Table 1). MVA of these factors revealed that trunk location (P = 0.048), and radial CTV 1.5 cm (P = 0.006) were independently associated with higher rates of LR (Table 2). No significant difference in LR was found with preoperative radiation, margin status, tumor size, unintended initial surgery, use of bolus, or chemotherapy (Table 1). Of the patients with LR, 10 out of 15 had a trunk location, and 10 of 15 were muscle-invasive (T2b). In relation to the field of radiation, 10 out of 15 were marginal at the radial edge of the field, 4 out of 15 were in-field recurrences (1 of which had a positive margin), and 1 in 15 recurred marginally at the longitudinal-proximal edge of the radiation field in which the CTV margin was 2 cm (Table 3). None of the LRs were completely outside the field of radiation. Most local recurrences (14 out of 15) correlated with the depth of their initial stage, and only 1 patient with T2a disease experienced a cutaneous recurrence, which had also recurred at the marginal edge of the radial margin, despite having a bolus applied. Most of the patients were treated with 3DCRT (91%), and only 9 (9%) patients were treated with IMRT.
There were 15 patients with LR, with an estimated 5-year LRFS of 83.0%. Sixty-nine percent of patients had a wide radial margin since the field edge was beyond the fascial boundary with 7.2% LR. Of these with a wide fascial margin, those with a positive margin had 20% LR, whereas those with a negative margin had 5.1% LR. Patients with a 1.5 cm radial CTV that was not beyond the fascial boundary experienced 45.4% LR, but those with radial CTV ≤ 2.5 cm did not experience LR, despite not having a fascial boundary (Table 4).
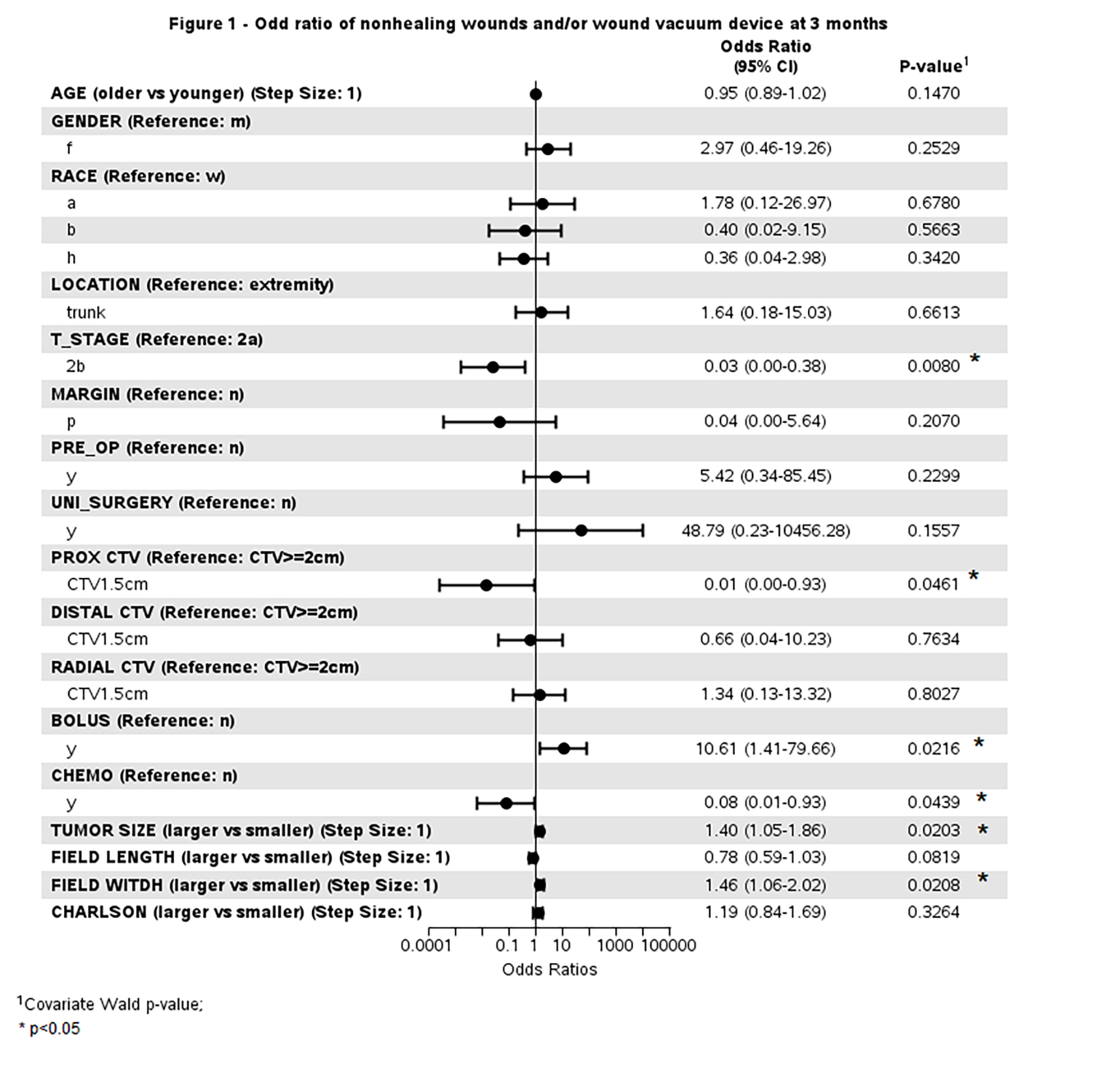
Requirement of a wound vacuum device and/or open wounds occurred in 27 (27%) patients, ranging from 3 days to 29.7 months, with the median duration of wound vacuum devices/open wounds being 4.7 months. The odds ratio for open wounds at 3 months or the presence of a wound vacuum device showed a higher risk for patients with larger tumor size (P = 0.02), larger field width (P = 0.02), and use of a bolus (P = 0.02). Patients with muscle-invasive disease had a lower odds ratio of wound complications compared with subcutaneous disease (P = 0.008). Also, patients with a smaller proximal CTV of 1.5 cm (P = 0.046) and those who underwent chemotherapy (P = 0.044) experienced a lower risk of wound complications (Figure 1).
Discussion
CTV Margins
RTOG Guidelines for ESTS published in 2011 recommended using CTV of 3-5 cm longitudinally, but only 1.5 cm radially. 3-5 At that time, it was unclear if this could be extrapolated to trunk lesions, where space is more limited, or to subcutaneous tumors. In the current study, although longitudinal proximal/distal CTV of 1.5 cm was only associated with increased LR on univariate analysis, the reason may be that the standard of care based on RTOG guidelines was already a CTV of 3 cm. This resulted in only a small percentage of our patients undergoing a longitudinal CTV of 1.5 cm, as 11% of patients had a longitudinal CTV of 1.5 cm, making statistical significance more difficult on MVA. Thus, it seems that a longitudinal proximal/distal CTV of 3 cm should be adequate, although this can be reduced in the presence of a fascial boundary. However, for high-grade sarcomas > 5 cm, a radial CTV of 1.5 cm may not be sufficient without a fascial boundary. American Society for Radiation Oncology (ASTRO) guidelines published in 2021 concluded that radial CTV of 3-4 cm is now recommended for subcutaneous disease; so why not for muscle-invasive disease? 15 In our study, we found a significant rate of LR using a CTV of 1.5 cm for both subcutaneous and muscle-invasive disease, in the absence of a fascial boundary, with the majority being marginal field-edge recurrences. Without a fascial boundary, high-grade sarcomas can easily extend radially to adjacent musculature for T2b disease, and thus we would only recommend a radial CTV of 1.5 cm only in the presence of a fascial boundary. It may be that a radial CTV of 1.5 cm is adequate for low-intermediate grade sarcomas or smaller high-grade sarcomas ≤ 5 cm, but a one-size-fits-all approach may not be suitable, especially for high-grade sarcomas > 5 cm. There was a slightly higher LR for positive margins, but this was not significant, possibly due to the small number of patients with positive margins, and that radiation may have some role in making up for positive margins. 16,17 Despite findings of increased LR with a radial CTV of 1.5 cm in the current study, there are studies that support reducing field size when using radiation as part of limb-salvage therapy. A randomized study using brachytherapy has led to the possibility of using smaller CTV margins, although the magnitude of the benefit seemed smaller than the randomized study using EBRT, where larger margins were used. 2,18 RTOG 0630 performed a phase II trial on STS, utilizing 3 cm longitudinal proximal/distal CTV, and 1.5 cm radial CTV, in which 74 patients underwent preoperative radiation followed by surgery. There were 5 patients with LR, and all were in-field. However, only 48.1% had high-grade histology, and 11.4% had smaller T1 lesions, so only about 32 patients had sarcomas > 5 cm of high grade, with a shorter median follow-up of 3.6 years. 19
ASTRO published updated guidelines in 2021, which recommend a longitudinal proximal/distal CTV of 3 cm, but still maintain a radial CTV of 1.5 cm for muscle-invasive disease. However, for subcutaneous disease, ASTRO now recommends radial CTV margins of 3-4 cm. 15 Our data support these guidelines for subcutaneous disease, although our data suggest also expanding radial CTV to at least 2-2.5 cm for muscle-invasive disease, due to higher marginal LR rates at the radial margin when a 1.5 cm radial CTV is applied, in the absence of a fascial boundary. Most of our patients (69%) had a radial margin that was beyond a fascial boundary, so a 1.5 cm CTV was considered adequate in these patients, but in the absence of a fascial boundary, we found a higher LR with a 1.5 cm radial CTV margin.
Wound Complications
The presence of an open wound at 3 months or the use of a wound vacuum device were significantly higher when using wider fields, and so an attempt to make radiation fields smaller is an important goal. In our study, utilizing a smaller longitudinal proximal CTV reduced wound complications (Figure 1). Interestingly, chemotherapy also reduced wound complication rates. Most of our patients underwent neoadjuvant sequential chemotherapy, in which preoperative radiation followed chemotherapy, despite it causing immune suppression. By the time radiation started, patient blood counts had time to recover, and the neoadjuvant chemotherapy caused shrinkage of these large sarcomas. It may be this cytoreduction that led to lower wound complications from chemotherapy, as we only included the most chemotherapy-sensitive STS histology in our study (Figure 1). 20
Lastly, we found that the use of a bolus was associated with higher wound complications. In our experience, only 1 patient with T2a disease had a cutaneous recurrence, which recurred at the radial edge in which the CTV was 1.5 cm, and the skin was part of the CTV where the bolus was applied. However, in most cases our soft-tissue surgeon will remove the overlying skin when sarcomas are close to the dermis. The majority of our LRs were not cutaneous, making it less likely for a bolus to impact LR rates. Thus, we concur with the most recent ASTRO guidelines from 2021, which do not recommend the use of a bolus in the treatment of STS with radiation. 15
Limitations and Final Thoughts
One limitation of this study is that the majority of our patients were treated with 3DCRT techniques, mostly using opposed fields. This leads to a more rapid falloff in radiation dose at the radial edge. One study published that IMRT had a lower rate of LR, and this may be due to the more gradual falloff of radial dose, which can still be effective in controlling subclinical disease, and thus it’s possible that a smaller radial CTV, such as 1.5 cm, could be achievable with IMRT, and that the current study utilized older techniques of treatment. 21,22 Thus, we anticipate the VORTEX trial results (randomized trial of volume of postoperative radiation therapy given to adult patients with ESTS), although our study predominantly used preoperative radiation. Another limitation of our study was the omission of image-guided radiation therapy (IGRT), in which studies have suggested a reduction in the CTV to PTV expansion from 1.0 cm to 0.5 cm and relatively lower complications with the use of IGRT. 19,23
Thirdly, none of the patients had a radial CTV of 2.0 cm, although we did have some with a longitudinal CTV of 2.0 cm (Tables 3,4). Due to the influence of the RTOG and ASTRO guidelines, it appears that there was a significant application of the use of a radial CTV of 1.5 cm. 3-5,15
We suggest when using 3DCRT that radial CTV for subcutaneous lesions can be extended to 2.0-2.5 cm, as this would be similar to 3 cm longitudinally due to lower scatter contribution at the proximal and distal edges of the radiation field. 24 For muscle-invasive disease, we also think that a 2.0-2.5 cm radial margin would be appropriate, although this must be balanced with the possibility of a slightly higher risk of wound complications. By increasing the CTV by a small amount, we can potentially reduce marginal recurrences, as there is evidence of higher LR rates leading to a decline in overall survival in STS. 25
Conclusion
Longitudinal proximal/distal CTV margins of 3 cm seem adequate, but high-grade STS ≤ 5 cm may benefit from increased radial CTV margins of 2-2.5 cm in the absence of a fascial boundary, although larger CTV may increase nonhealing wound rates. Bolus techniques may increase wound complications in T2a-b STS, and should not be routinely employed.
References
- Rosenberg SA, Tepper J, Glatstein E, et al. The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg. 1982;196:305-315.
- Yang JC, Chang AE, Baker AR, et al. Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol. 1998;16:197-203.
- Wang D, Bosch W, Roberge D, et al. RTOG sarcoma radiation oncologists reach consensus on gross tumor volume and clinical target volume on computed tomographic images for preoperative radiotherapy of primary soft tissue sarcoma of extremity in radiation therapy oncology group studies. Int J Radiat Oncol Biol Phys. 2011;81:e525-e528.
- Dickie CI, Griffin AM, Parent AL, et al. The relationship between local recurrence and radiotherapy treatment volume for soft tissue sarcomas treated with external beam radiotherapy and function preservation surgery. Int J Radiat Oncol Biol Phys. 2012;82:1528-1534.
- Haas RL, DeLaney TF, O’Sullivan B, et al. Radiotherapy for management of extremity soft tissue sarcomas: why, when, and where? Int J Radiat Oncol Biol Phys.2012;84:572-580.
- ICRU Report 50. Prescribing, recording, and reporting of photon beam theapy. Bethesda, MD: International Commission on Radiation Units and Measurement; 1993.
- Davis AM, O’Sullivan B, Turcotte R, et al. Late radiation morbidity following randomization to preoperative vs postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol. 2005;75:48-53.
- Edge SB, Byrd DR, Compton CC, et al. AJCC Cancer Staging Handbook, 7th ed. Springer; 2010.
- Coindre JM, Bertrand G, Chateau MD, et.al. Recommandations pour la prise en charge anatomo-pathologique des sarcomes des tissus mous de l’adulte. Ann Pathol. 1998;18:505-511.
- Yami AA, Griffin AM, Ferguson PC, et al. Positive surgical margins in soft tissue sarcoma treated with preoperative radiation: Is a postoperative boost necessary? Int J Radiat Oncol Biol Phys. 2010;77:1191-1197.
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-383.
- Kaplan EL, Meier P. Non-parametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457-500.
- Cox DR. Regression models and life tables. JR Stat Soc B. 1972;34:187-189.
- Tests for the Odds Ratio in Logistic Regression with One Normal X (Wald Test). NCSS.com. https://ncss-wpengine.netdna-ssl.com/wpcontent/themes/ncss/ pdf/Procedures/PASS/Tests_for_the_Odds_Ratio_in_Logistic_Regression_with_Two_Binary_XsWald_Test.pdf
- Salerno KE, Alektiar KM, Baldini EH, et al. Radiation therapy for treatment of soft tissue sarcoma in adults: executive summary of an ASTRO clinical prac- tice guideline. Pract Radiat Oncol. 2021;11:339-351.
- Gronchi A, Casali PG, Mariani L, et al. Status of surgical margins and prognosis in adult soft tissue sarcomas of the extremities: a series of patients treated at a single institution. J Clin Oncol. 2005;23:96-104.
- Jebsen NL, Trovik CS, Bauer HC, et al. Radiotherapy to improve local control regardless of surgical margin and malignancy grade in extremity and trunk wall soft tissue sarcoma: a scandinavian sarcoma group study. Int J Radiat Oncol Biol Phys. 2008;71:1196-1203.
- Pister PW, Harrison LB, Leung DH, et al. Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol. 1996;14:859-868.
- Wang D, Zhang Q, Eisenberg JM, et al. Significant reduction of late toxicities in patients with extremity sarcomas treated with image-guided radiation therapy to a reduced target volume: results of radiation therapy oncology group RTOG-0630 trial. J Clin Oncol. 2015;33:2231-2238.
- Goy BW, Syed S, Padmanabhan A, et al. The role of Ifosfamide-Doxorubicin chemotherapy in histology-specific, high grade, locally advanced soft tissue sarcoma, a 14-year experience. Radiother and Oncol. 2021;165:174-178.
- Folkert MR, Singer S, Brennan MF, et al. Comparison of local recurrence with conventional and intensity-modulated radiation therapy for primary soft-tissue sarcomas of the extremity. J Clin Oncol. 2014;32:3236-3241.
- Withers HR, Peters LJ, Taylor JM. Dose-response relationship for radiation therapy of subclinical disease. Int J Radiat Oncol Biol Phys. 1995;31:353-359.
- O’Sullivan BO, Griffin AM, Dickie CI, et al. Phase 2 study of preoperative image-guided intensity-modulated radiation therapy to reduce wound and combined modality morbidities in lower extremity soft tissue sarcoma. Cancer. 2013;119:1878-1884.
- Merredith WJ. Radium Dosage: The Manchester System. Livingstone, Ltd;1967.
- Lewis JJ, Leung D, Heslin M, et al. Association of local recurrence with subsequent survival in extremity soft tissue sarcoma. J Clin Oncol. 1997;15:646-652.
Citation
BW G, CS H, JF Y, SP S. Radiation Therapy Techniques in the Management of Locally Advanced, High-Grade, Soft-Tissue Sarcoma. Appl Radiat Oncol. 2022;(3):27-34.
October 14, 2022