Radiation Therapy in Cancer of Accessory Breast Tissue: Questions for Treatment Guidelines, Case Series, and Literature Review
Images



Abstract
Background: The presence of accessory breast tissue (ABT) has been documented throughout medical literature and, like prepectoral breast tissue, is at risk of developing cancer. However, there is a clear lack of data and reported experience to guide clinicians in choosing the optimal treatment for ABT cancers. Specifically, there is limited information on the role of adjuvant radiation therapy (RT) and no clear consensus on guidelines.
Methods: We conducted a literature review of previously reported ABT cancers and their treatment. We also queried the database of breast cancer patients treated at our institution and identified 3 cases of ABT cancer for which adjuvant RT was offered.
Results: We present 3 cases of ABT cancer, treated at our institution, with 3 approaches to adjuvant RT. The first 2 were stage 1 ER/PR+ HER2-invasive ductal carcinomas in postmenopausal women. Of these, 1 received adjuvant endocrine therapy only without RT, while the other received hypofractionated RT, 26 Gy in 5 fractions. The third case was locally advanced ER/PR+ HER2+ invasive ductal carcinoma in a premenopausal female. She underwent adjuvant breast and nodal irradiation with a boost to the axillary tumor bed. In each of the cases, the rationale behind the recommendations, dose and treatment volume are discussed
Conclusions: In formulating treatment recommendations for patients with ABT cancer, clinicians face several questions without answers from the existing data. Small case series and literature reviews such as this one can be used to provide the framework for considering treatment options in these challenging cases.
Keywords: accessory breast tissue, ectopic breast tissue, supernumerary breast tissue, axillary breast tissue, adjuvant radiation therapy
The presence of accessory breast tissue (ABT) has been well documented throughout medical literature. Most accessory breast tissue develops along the mammary ride (milk line), and the current classification of ABT is based on the system first developed by Kajava in 1915. In most cases, individuals develop polythelia (extra nipples) without functioning ductal tissue. However, there have been reports of fully functioning ABT, which includes the production of milk.1 ABT, like regular breast tissue, is at risk of developing cancer, but literature regarding the incidence and treatment of cancer in ABT is limited. Specifically, there is little to no guidance on how to approach these cases from the standpoint of adjuvant radiation therapy (RT). Here we present 3 cases of resected carcinoma in the ABT treated at our institution, describe 3 radiotherapeutic approaches, summarize the current literature, and discuss the clinical considerations that may guide recommendations in this unusual scenario.
Case Presentations
Case 1
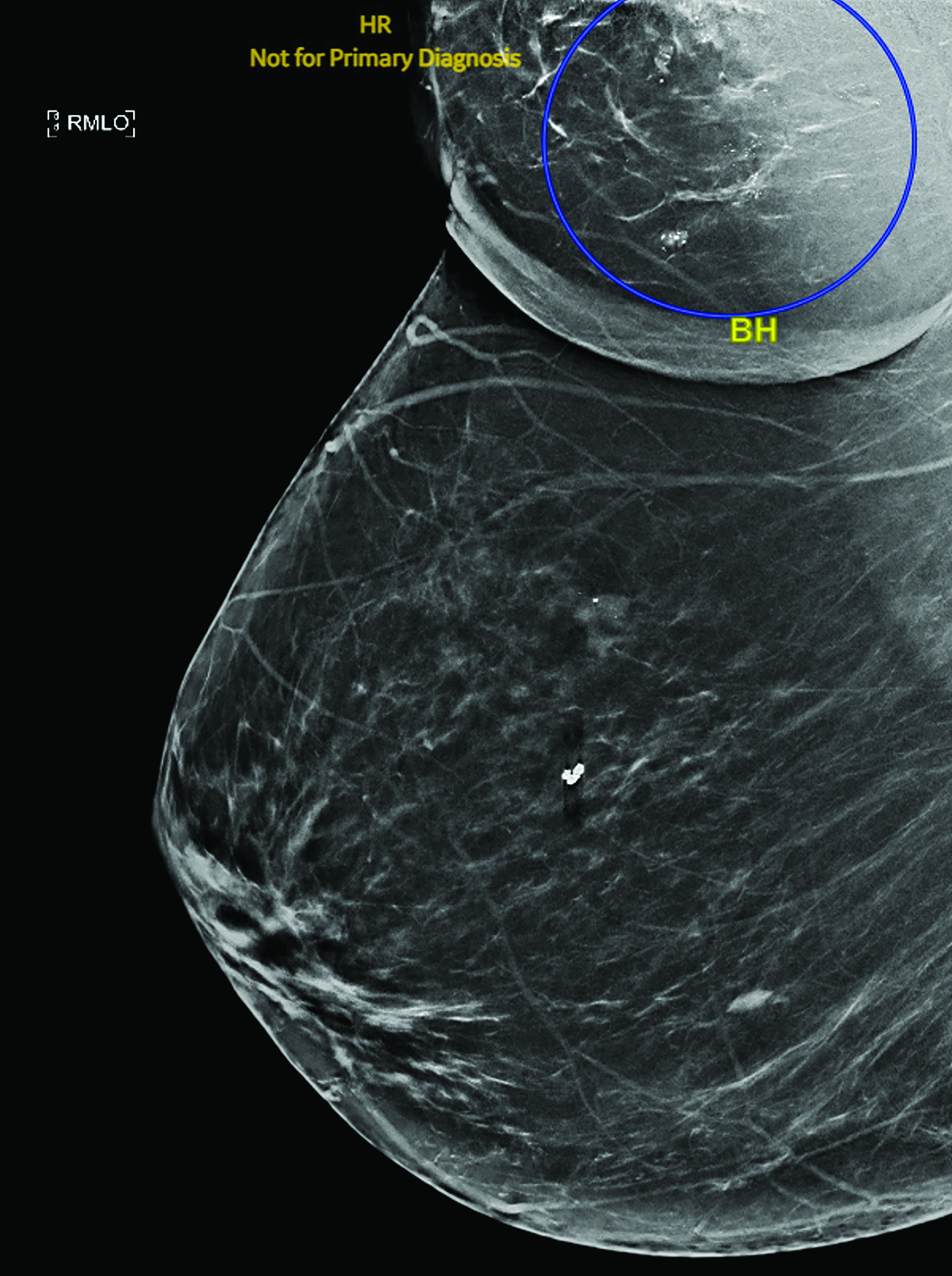
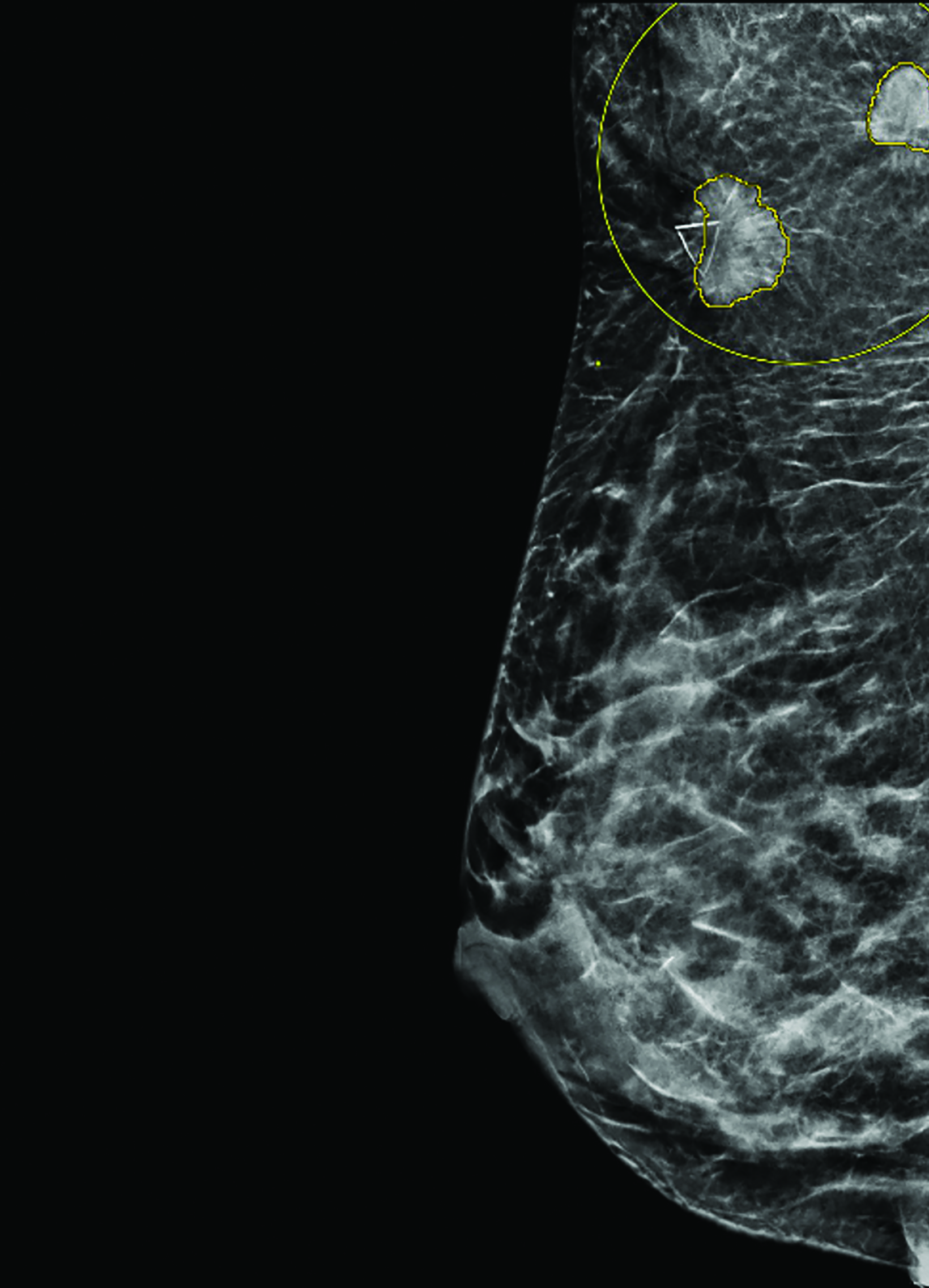
The first case involves a G1P1 61-year-old postmenopausal woman with a documented history of bilateral axillary ABT for several years. She had previously undergone a biopsy for suspicious calcifications in her left breast with benign findings. In March 2021, the patient underwent a routine bilateral screening mammogram. She was asymptomatic and had no acute concerns. Her mammogram demonstrated 9 foci of calcifications in the right axillary ABT (Figure 1).
Subsequent stereotactic biopsy demonstrated moderately differentiated invasive ductal cancer, estrogen receptor (ER) and progesterone receptor (PR) positive, HER2 negative, Ki-67 8%. She was clinically staged as T1N0. She was then evaluated by the breast surgical oncology team in April 2021 and elected to undergo wide local excision of the ABT with wire localization and sentinel lymph node biopsy. Surgical pathology demonstrated invasive ductal carcinoma (IDC) 20 mm at its largest width and ductal carcinoma in situ (DCIS) 5 cm at its largest width. The posterior and lateral surgical margins were identified by direct visualization of the deep tissue below the pectoralis and lateral to the latissimus muscles, respectively.
All final margins were negative for carcinoma (IDC and DCIS are more than 1 cm away from all margins). Tissue testing demonstrated similar molecular profile to that of the biopsy, and Oncotype Dx Recurrence score was 1. In addition, her sentinel lymph node biopsy was negative (0/1). She was therefore staged as pT1cN0. Postoperatively, she required multiple seroma aspirations and developed palpable cording in her axilla. She was evaluated by a medical oncologist and started on letrozole with plans to continue endocrine therapy for 5 years.
She then presented to the radiation oncology department for consideration of adjuvant radiation. The patient was presented with 2 treatment options: 1) RT to the axillary area only, since there was felt to be no anatomic communication between the ABT and regular breast tissue; and 2) no RT, since all ectopic mammary tissue had been removed, and this could, in a sense, be considered a full mastectomy of the supernumerary breast. After extensive discussion, she opted to forgo the RT. At the time of writing this paper she had completed an 18-month imaging follow-up and a 15-month clinic follow-up. She was tolerating letrozole well with minimal side effects, and her bilateral mammogram was negative for disease recurrence.
Case 2
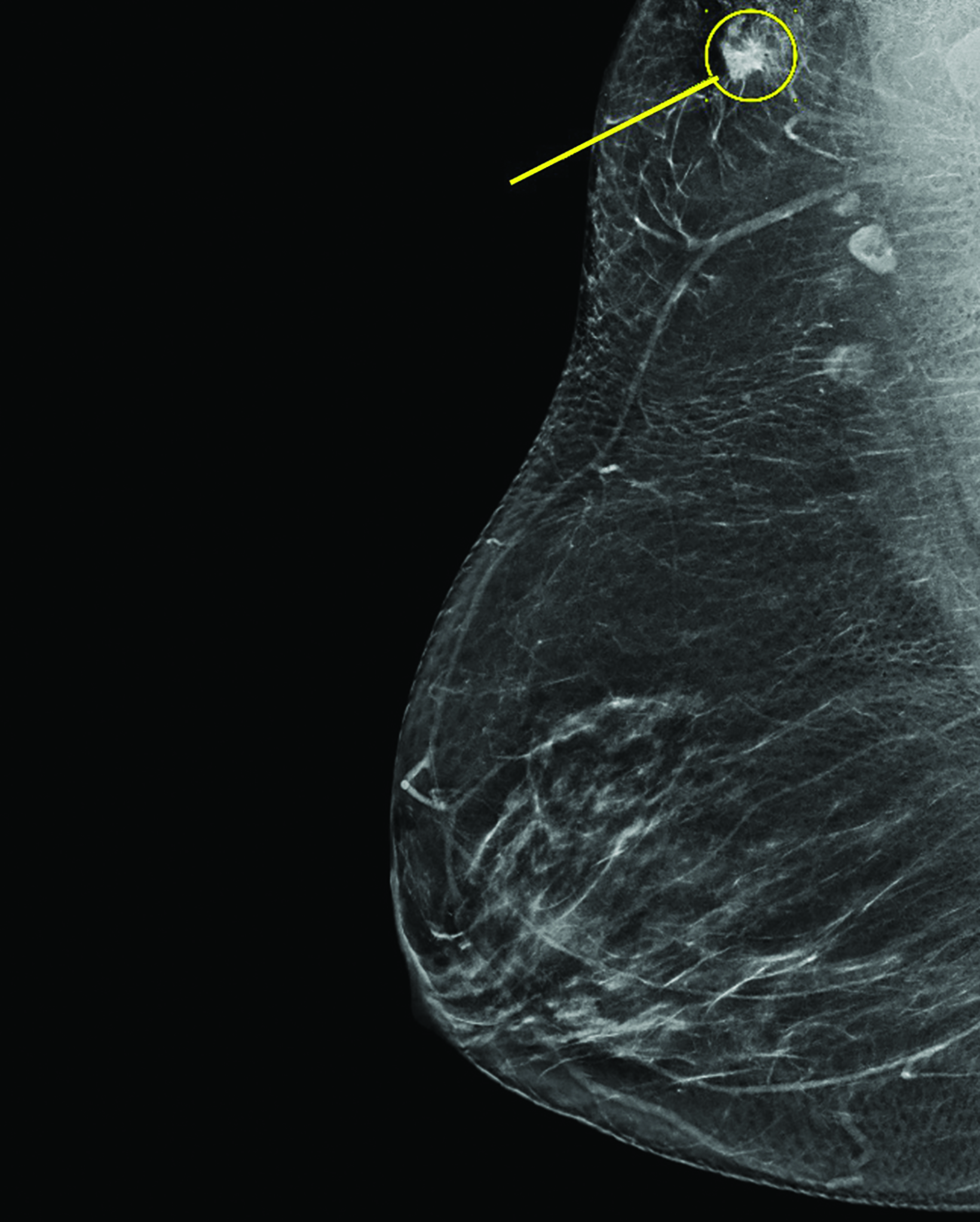
The second case involves a G3P2 76-year-old postmenopausal woman who had never been diagnosed with ABT. Over the years, she had undergone 2 biopsies for suspicious findings on screening mammograms, but both were benign. In November 2021, she underwent a routine bilateral mammogram where a 1.2 cm site of asymmetry was identified in the right axilla (Figure 2A). A biopsy of the site demonstrated IDC that was well differentiated, ER/PR positive, Her-2 negative, and Ki-67 5%.
She was then evaluated by the breast surgical oncology team and underwent local resection aided by SAVI SCOUT (Merit Medical) with sentinel lymph node biopsy. Surgical pathology demonstrated that the mass excised was an accessory breast with a 1.8-cm single site of IDC. On the surgical specimen, invasive carcinoma was 0.2 mm from the inked anterior margin, < 1 mm from the posterior margin, and 1.5 mm from the inferior margin. Additional superior, inferior, lateral, medial, anterior, and posterior margins measuring at least 1.0 × 1.0 × 0.5 cm, only contained benign breast tissue or benign fibroadipose tissue. All final margins were negative, and her sentinel lymph node biopsy was negative (0/1). Tissue testing demonstrated similar molecular profile to that of the biopsy sample, and Oncotype Dx Recurrence score was 16.
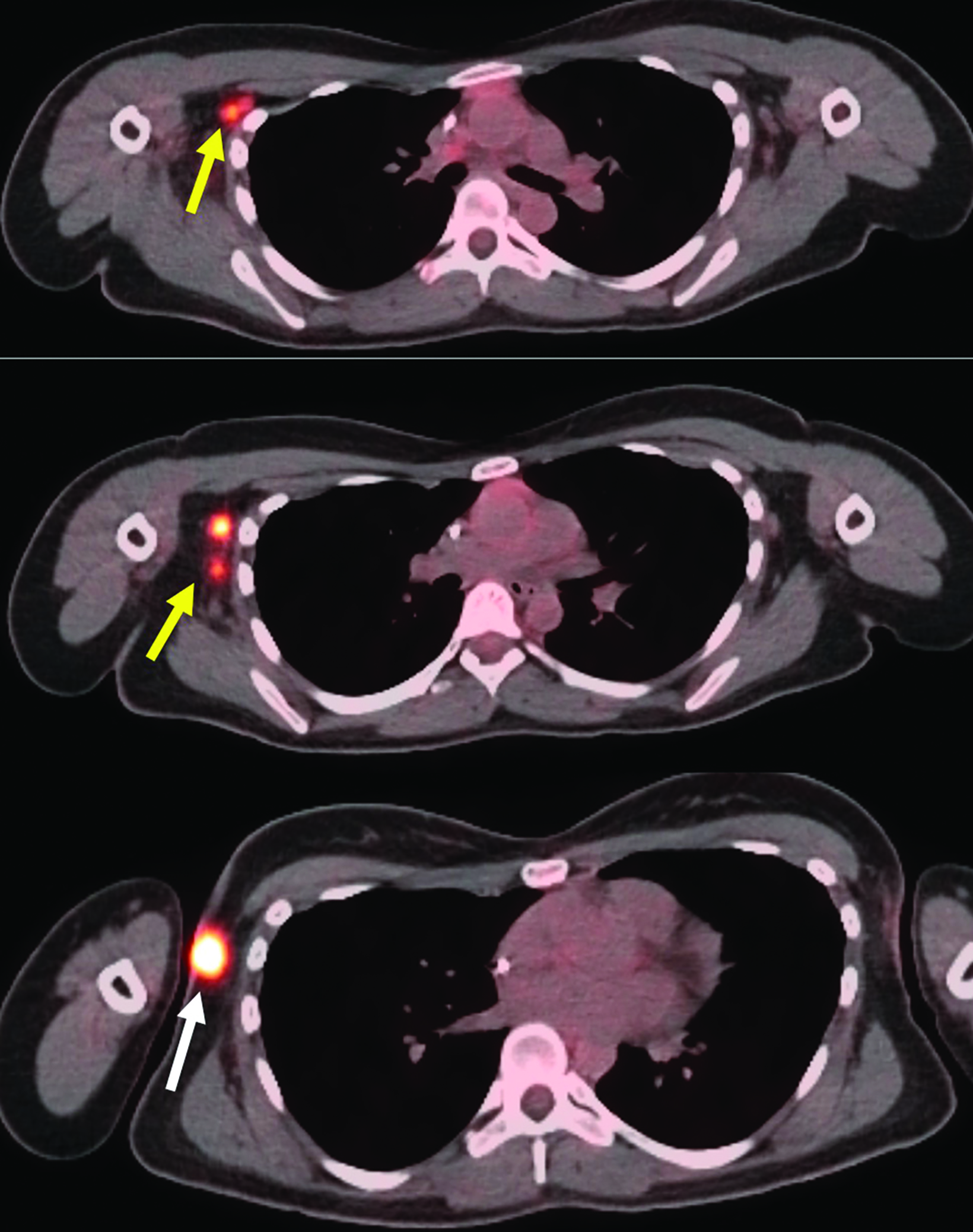
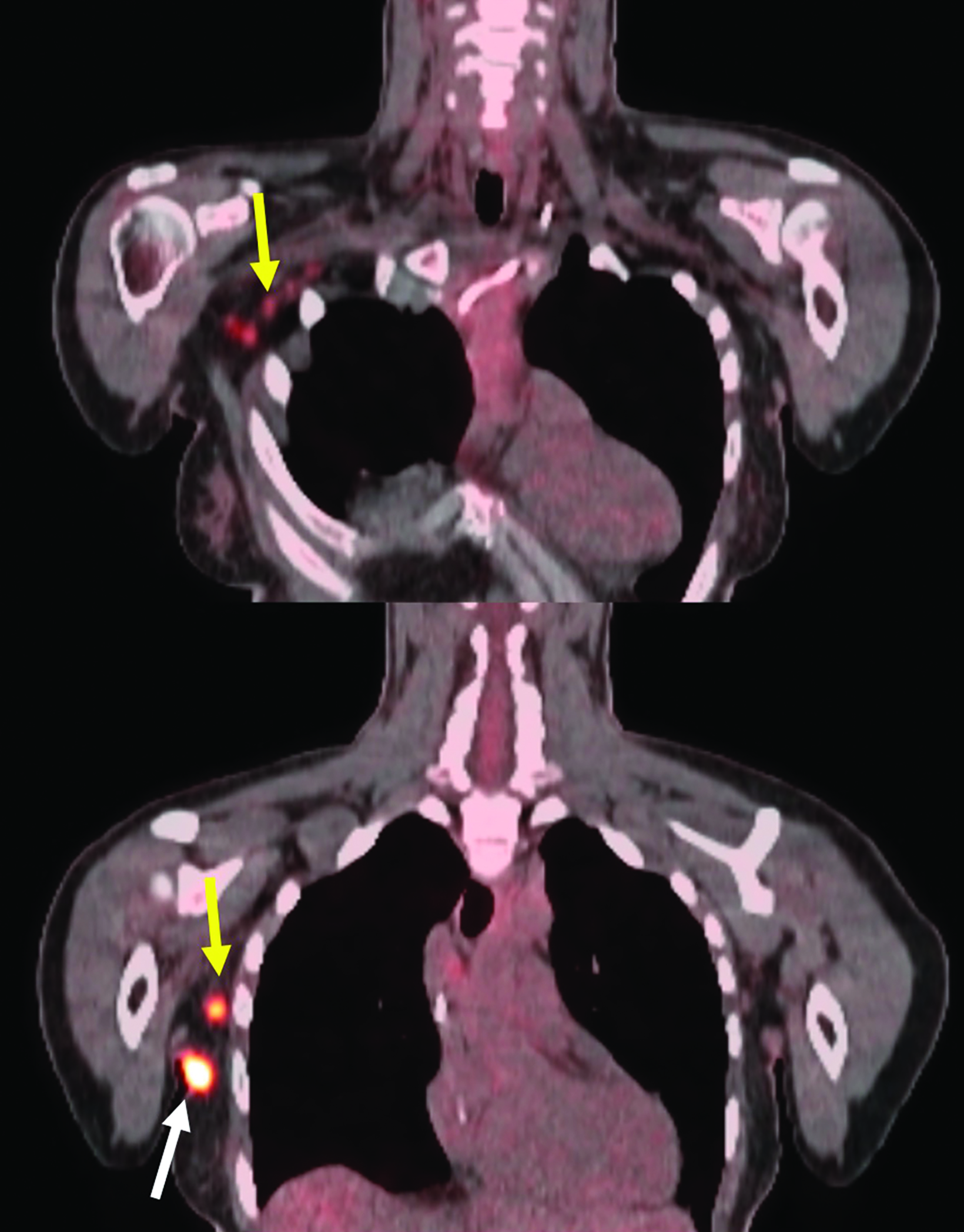
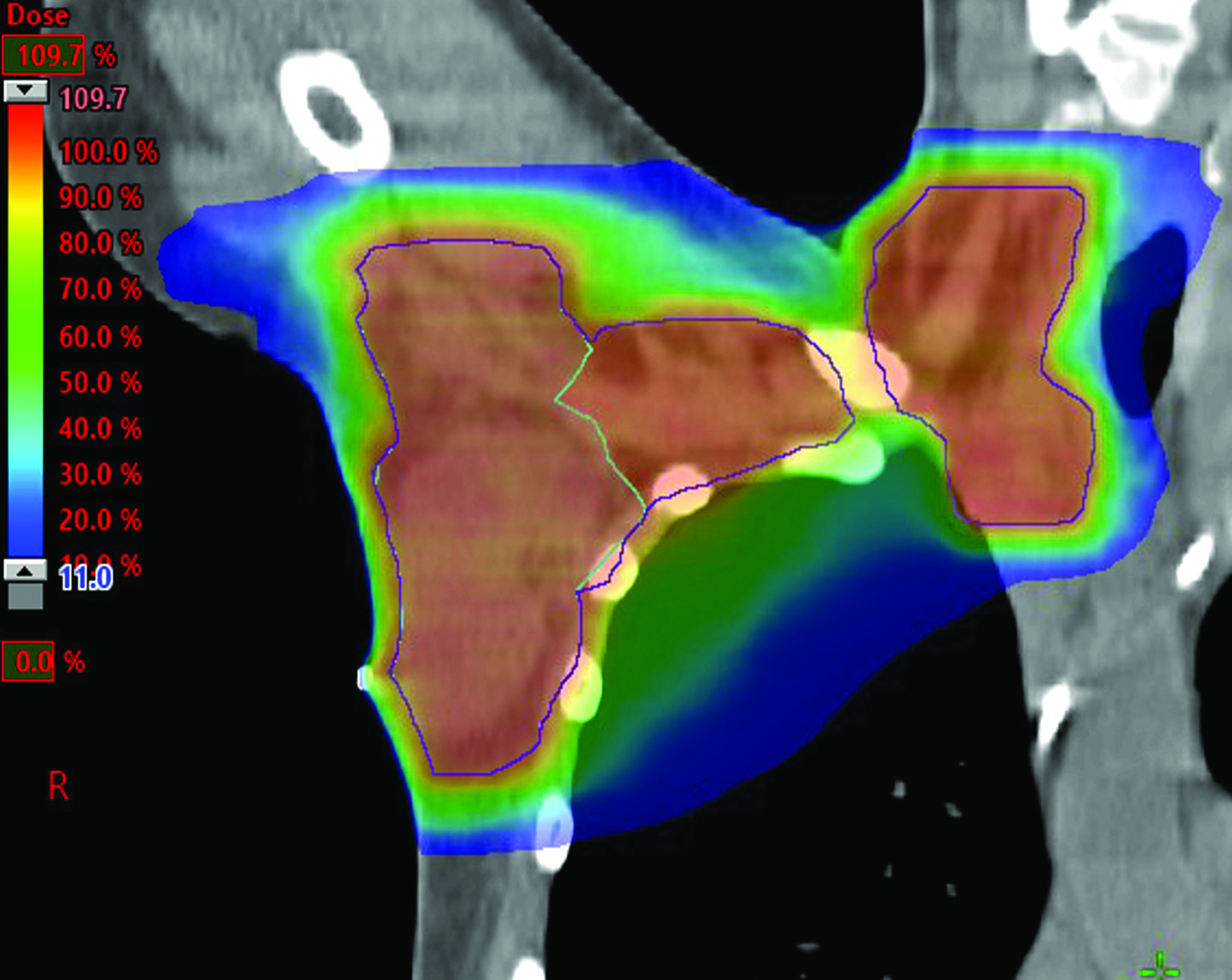
She was diagnosed with pT1cN0 IDC of a right axillary accessory breast. She met with a medical oncologist and was started on anastrozole. She then presented to the radiation oncology department for evaluation of adjuvant radiation. Similar to the previous case, this patient was presented with the options of RT to the axillary area only vs no RT since all the ectopic mammary tissue had been removed. After extensive discussion, this patient opted to complete 2600 cGy in 5 fractions to the axillary area (Figures 2B-C). At the time of writing, she had completed a 9-month imaging follow-up and a 10-month clinic follow-up. She was tolerating anastrozole well with minimal side effects, and her bilateral mammogram was negative for disease recurrence.
Case 3
The third case involves a G1P1 33-year-old premenopausal woman. The patient reported first feeling bilateral axillary fullness at the time of her pregnancy in 2019. She indicated she was able to express milk from tissue in her left axilla during nursing. The right-sided axillary fullness persisted after she stopped nursing and she had reported feeling a nodule in her right axilla. She underwent an ultrasound in August 2021, demonstrating a 1.2-cm hypoechoic nodule in what was initially believed to be the right axillary tail, and 2 likely reactive hypoechoic lymph nodes measuring 1.3 cm and 1.6 cm, respectively (BIRADS 3).
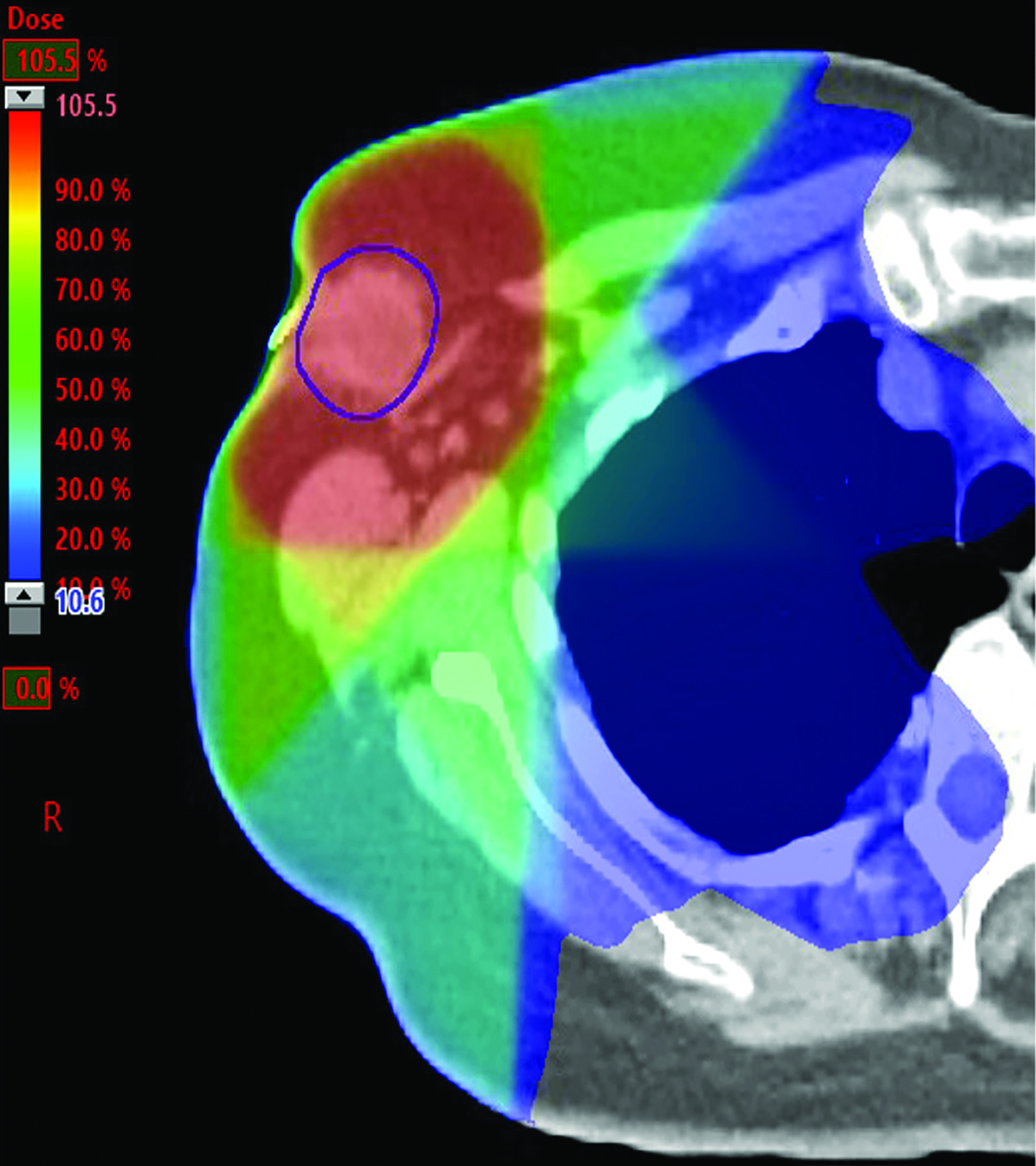
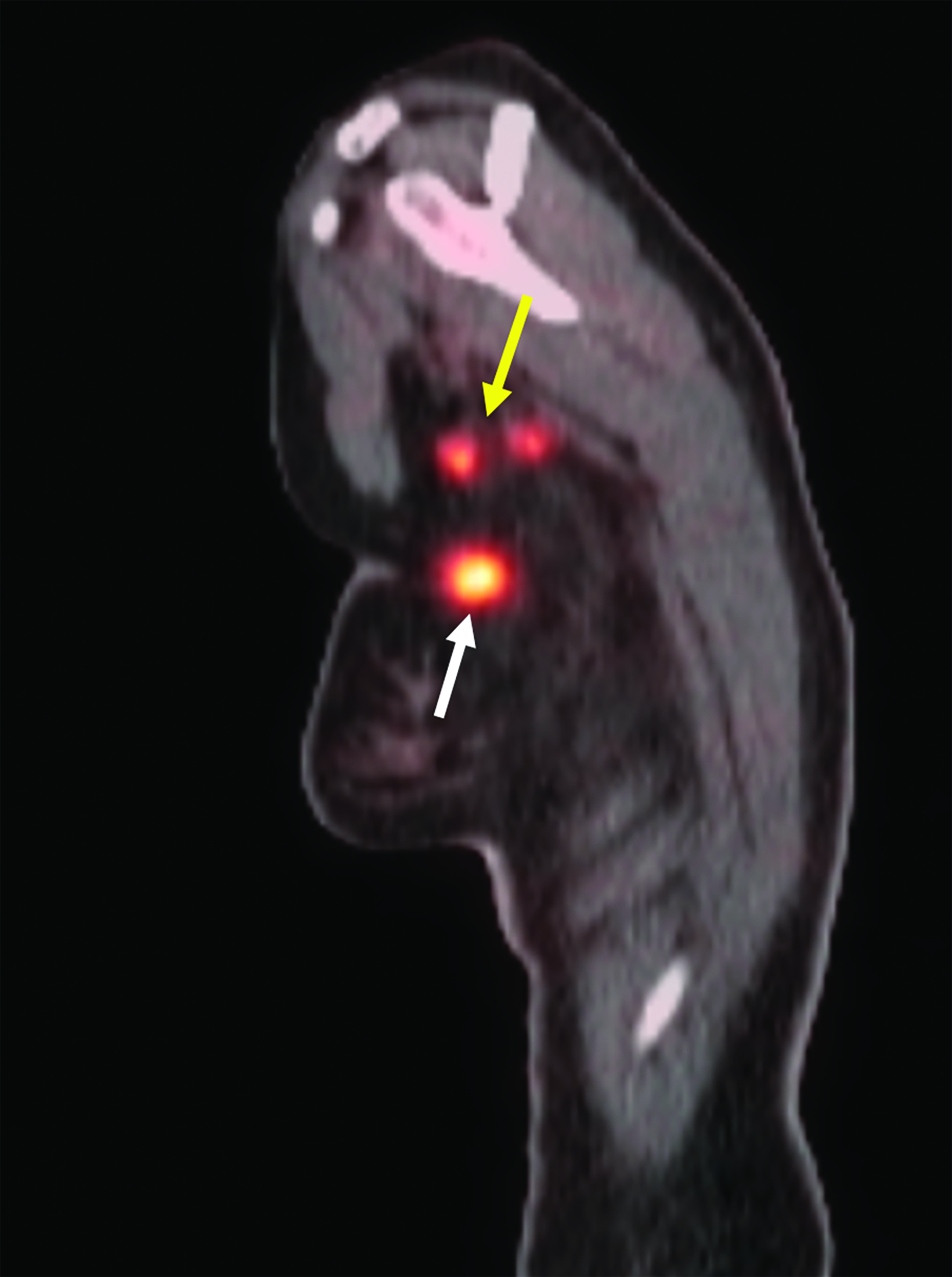
Repeat ultrasound and mammogram (Figure 4A) in November 2021 demonstrated a 1.7-cm irregular spiculated mass in the right axillary tail. A second irregular spiculated mass likely representing a 1.2-cm lymph node was noted in the right axilla. A biopsy of the right axillary tail mass demonstrated poorly differentiated IDC measuring 1.4 cm and was associated with calcifications, ER 82%, PR 35%, HER2+3 positive, and Ki-67 43%. PET demonstrated a hypermetabolic right axillary lesion corresponding to her known malignancy as well as multiple hypermetabolic right axillary lymph nodes consistent with metastases (Figure 3). She was therefore staged as cT1N2.
The patient completed 4 cycles of neoadjuvant doxorubicin/cyclophosphamide followed by 4 cycles of TCHP (Taxotere/Carboplatin/Herceptin/Perjeta). She was then evaluated by a breast surgeon in May 2022 and underwent local resection aided by SAVI SCOUT and sentinel lymph node biopsy. The surgical specimen measured 7.7 × 1.9 × 3.1 cm and pathology was negative for residual carcinoma. Sentinel lymph node biopsy did not show residual carcinoma (0/5), yet multiple lymph nodes exhibited treatment effect with evidence of fibrous scarring and focal calcifications.
Of note during the operation, the surgeon noted a distinct anatomical separation between the axillary tail of the breast and area containing the primary lesion. Thus, this was determined to be an ABT cancer rather than an axillary tail cancer, contiguous with the breast, as had been initially assumed. All final surgical margins (≥ 1 mm) were negative, and the patient was staged ypT0N0. She was re-evaluated by the medical oncologist after the surgery, and the recommendation was to start KADCYLA. She then presented to the radiation oncology department for discussion of adjuvant radiation. Given her young age and the locally advanced nature of her initial disease, the options presented to her were different from those discussed with the patients above. Specifically, the issues discussed were: 1) regional nodal irradiation to include the axillary and supraclavicular nodal stations but not internal mammary lymph nodes; 2) irradiation of the axillary tumor bed but not the entire right breast because, as in the other cases, there was felt to be an anatomic barrier between the ABT and regular breast tissue; 3) boosting the axillary tumor bed (which is normally given after a lumpectomy but not after a mastectomy).
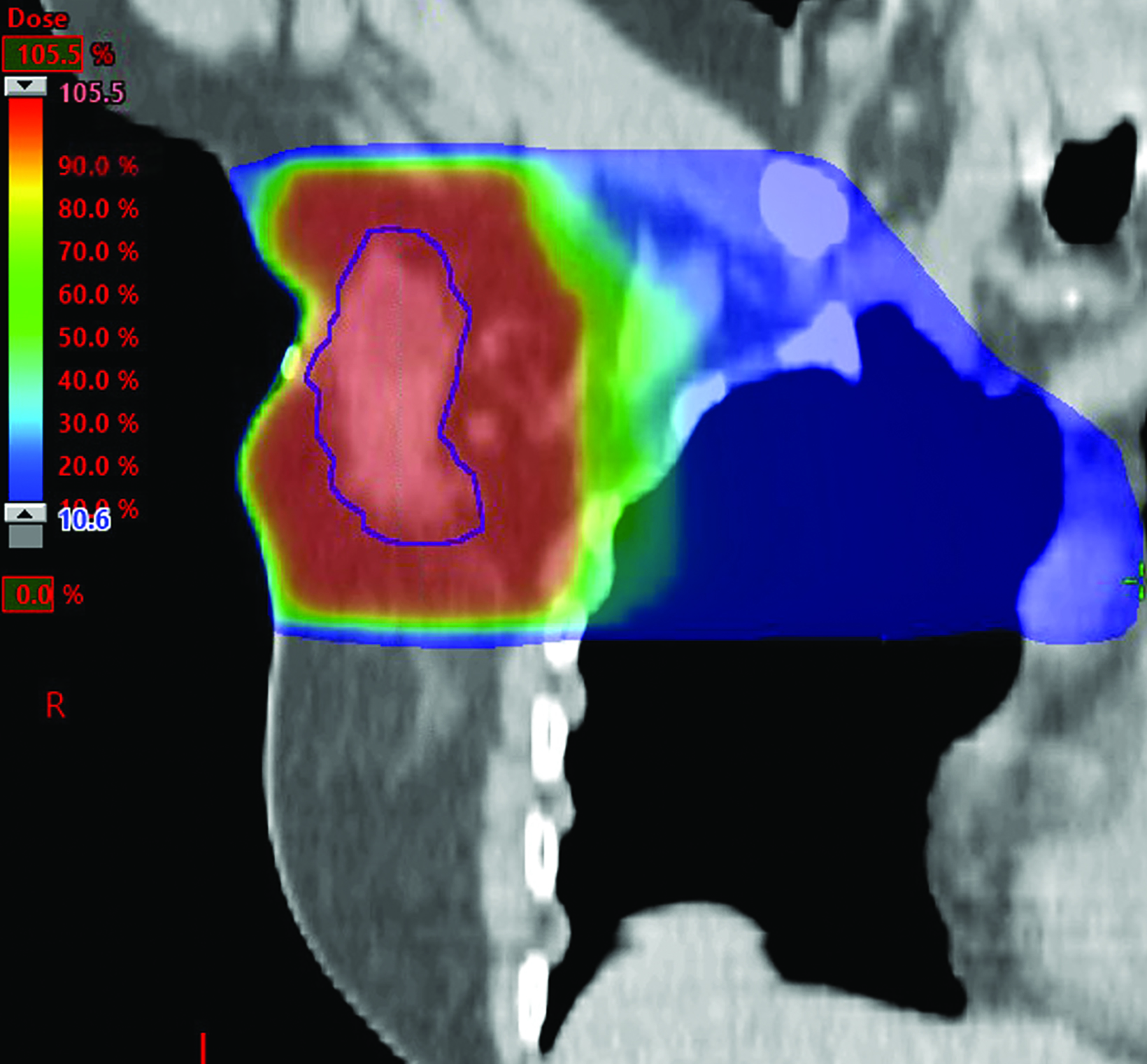
After extensive discussion, the patient opted to undergo a hypofractionated course of 4256 cGy in 16 fractions to the right axilla and supraclavicular fossa, followed by a boost of 1000 cGy in 4 fractions to the axillary tumor bed (Figures 4B-D).
At the time of writing, the patient has completed the intended RT course with minimal toxicity and completed 16 cycles of KAD- CYLA. She has also completed a 3-month imaging follow-up PET/CT, which showed no F-18 fluorodeoxyglucose (FDG)-avid lesions in the right axilla and supraclavicular fossa, and a 6-month clinic follow-up at which time her breast examination was negative for evidence of disease.
Discussion
ABT is an umbrella term encompassing both supernumerary and aberrant anatomy. Supernumerary breast tissue is found along the milk line — an embryologic landmark running bilaterally from the axilla to the inguinal region. By contrast, aberrant refers to breast tissue found close to the normal breast tissue and by definition lacks organized secretory systems.2 In either case, the defined mass is an isolated fragment of glandular tissue without any connection to the native or “pectoral” breast. ABT is present in roughly 2% to 5% of the general population and incidence varies with ethnicity, sex, geographic region, and method used to determine its presence.
The presence of ABT was first described in detail in 1915, and first was classified into 8 types according to the existence of glandular tissue, nipple, areola, skin, and patch of hair.1 This system is still widely used in the literature today. Class 1 (also termed polymastia) consists of a complete breast with a nipple, areola and glandular tissue. Class 2 refers to glandular tissue and a nipple but no areola. Class 3 consists of an areola and glandular tissue but no nipple. Class 4 is glandular tissue only. Class 5 (also called pseudomamma) tissue contains a nipple and areola. Class 6 is composed of a nipple only (also termed polythelia) and class 7 consists of an areola only. Lastly, class 8 refers to just a patch of hair. All of our cases are strictly fibroglandular tissue in the axilla, which represents class 4 according to this system.
Literature is divided on whether there is an increased risk of malignant transformation in ABT. Of note, cancer in ABT appears to arise at an earlier age than in developmentally normal pectoral tissue. One review of 82 cases found the mean age at diagnosis to be 53.3 years in patients with cancer of ABT, whereas the mean age for pectoral breast cancer according to the National Cancer Institute is 61 years.3 Moreover, patients with ABT cancer tend to present with more advanced disease.4 It is important to note that ABT is generally not included in routine breast cancer screening studies. This may, in part, explain why some patients present at a later stage than traditionally located breast cancer.
To date, the literature is limited to case reports of ABT cancer with no established treatment paradigms. A literature review was conducted utilizing key words and phrases such as accessory breast tissue and ectopic breast tissue in PubMed to identify reported cases and guidelines. Upon our review, most case reports present a combination of preoperative chemotherapy, surgical resection, postoperative chemotherapy, RT, and endocrine therapy. RT is still recommended for maximizing locoregional control. In a 2009 review of case reports by Madej B et al,5 most radiation prescriptions were 5000 to 6600 cGy in 200 cGy per fraction. More recently in 2013, Hallam et al6 proposed extrapolating from the UK START B trial where 4000 cGy was given over 15 fractions. Notably though, there was no consensus on treatment fields. Proposed plans ranged from treating the tumor bed alone to including the ipsilateral uninvolved pectoral breast, axilla, and supraclavicular fossa even in the absence of nodal metastases.
In evaluating the cases presented here, we were faced with several key questions to help guide treatment recommendations:
The first question is how to classify the extent of surgery. Does the fact that the ABT, from which the carcinoma arises, and is an independent structure from the pectoral breast, imply that the surgical resection is effectively a mastectomy? If so, one decision-making approach would be to extrapolate the indications for postmastectomy radiation therapy from the classic studies, ie, only offer it in the cases of nodal involvement, T3 or T4 disease, and positive margins. If, on the other hand, this is considered a lumpectomy rather than mastectomy, in a low-risk patient one may consider applying criteria from the Hughes et al trial (CALGB C9343)7 for patients older than 70 or the PRIME II trial8 to identify those in whom omitting postoperative RT is an option. If radiation is offered as part of breast conservation therapy in these cases, we must also consider the applicability of accelerated partial breast irradiation (APBI) regimens, such as 600c Gy × 5 fractions.9.10 In a similar vein, if we are considering it whole-breast irradiation for the supernumerary breast tissue, the appropriateness of ultrahypofractionated regimens, such as 2600 cGy in 5 fractions as per the UK FAST FORWARD trial, should be assessed, particularly with respect to conferring a higher risk of lymphedema. In the first 2 cases, the patient stage was pT1cN0, the profile was consistent with luminal A molecular subtype, and the Oncotype Score was low. Considering these factors, as well as patient age and performance status, we presented several options, including: 1) hypofractionated RT to the axillary area of 4000-4256 cGy/15-16Fx +/- boost 1000-1600 cGy/5-8Fx; 2) Ultrahypofractionated RT 2600 cGy/5Fx as per UK FAST FORWARD trial (we felt this to be somewhat safer than the 600 cGy × 5 fractions APBI regimen from the standpoint of causing lymphedema); 3) endocrine therapy alone. The case for no RT can be made if one considers surgical resection of ABT as a mastectomy. One can also consider omitting RT based on relatively low recurrence rates in select low-risk patients after BCT, as seen in the CALGB and PRIME II studies.
Another important question is the utility of Oncotype scores in guiding treatment recommendations for ABT cancers, in the fashion that is being explored in the ongoing MA-3911 and DEBRA12 trials. Of note, our first patient would have been a candidate for the DEBRA trial had her cancer been in the pectoral breast.
Finally, in a locally advanced case, such as the one presented above, what is the optimal target volume, which nodal areas need to be included, and is a tumor bed boost warranted? In our third case the patient was cT1cN2 with biopsy-proven nodal involvement, and abnormal PET-avid nodes identified in both level 3 of the axilla and the supraclavicular fossa. Despite having a complete response to neoadjuvant chemotherapy, she was deemed a high-risk patient and adjuvant RT was offered to both the tumor bed and regional lymphatics. As in the other cases, the pectoral breast was not included in the treatment field as there was no anatomic connection between it and the resected ABT. Similarly, internal mammary LNs were not included in the treatment field, because the risk of spread to that location from the ABT was considered exceedingly low. Despite concerns about increased risk of lymphedema and after extensive discussion with the patient, a boost was given to the tumor bed because of the initially high tumor grade and Ki-67.
Conclusion
In formulating treatment recommendations for these patients, we were faced with several questions without answers from existing data. Considering the rarity of such clinical scenarios, there is little chance that large-scale randomized clinical trials can be carried out to guide clinicians in the future. In the absence of these trials, our hope is that small case series and literature reviews, such as this one, can at least provide the framework for considering treatment options in these challenging cases. To our knowledge, this is the first report where several possible radiotherapeutic approaches to carcinoma of the ABT are delineated.
References
- Kajava Y. The proportions of supernumerary nipples in the Finnish population. Duodecim. 1915;31:143-170. Accessed June 11, 2022. https://www.scienceopen. com/document?vid=0e2368fe-6e58-424f-9685-15b27c323485
- Laor T, Collins MH, Emery KH, Donnelly LF, Bove KE, Ballard ET. MRI appearance ofbaccessory breast tissue: a diagnostic consideration for an axillary mass in a peripubertal or pubertal girl. Am J Roentgenol. 2004;183(6):1779-1781. doi:10.2214/ajr.183.6.01831779
- Francone E, Nathan MJ, Murelli F, Bruno MS, Traverso E, Friedman D. Ectopic breast cancer: case report and review of the literature. Aesthetic Plast Surg. 37(4):746-749. doi:10.1007/s00266-013-0125-1
- DeFilippis EM, Arleo EK. The ABCs of accessory breast tissue: basic information every radiologist should know. Am J Roentgenol. 2014;202(5):1157-1162. doi:10.2214/AJR.13.10930
- Madej B, Balak B, Winkler I, Burdan F. Cancer of the accessory breast – a case report. Adv Med Sci. 2009;54(2):308-310. doi:10.2478/v10039-009-0031-6
- Hallam S, Aggarwal A, Predolac D, Cunnick G, Ashford R. Primary ectopic breast carcinoma in a supernumerary breast arising in the anterior chest wall: a case report and review of the literature. J Surg Case Rep. 2013;2013(12):rjt107. doi:10.1093/jscr/rjt107
- Hughes KS, Schnaper LA, Bellon JR, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31(19):2382-2387. doi:10.1200/JCO.2012.45.2615
- Kunkler IH, Williams LJ, Jack WJ, Cameron DA, Dixon JM; PRIME II investigators. Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (PRIME II): a randomised controlled trial (published correction appears in Lancet Oncol. 2015;16(3):e105). Lancet Oncol. 2015;16(3):266-273. doi:10.1016/S1470-2045(14)71221-5
- Livi L, Meattini I, Marrazzo L, et al. Accelerated partial breast irradiation using intensity-modulated radiotherapy versus whole breast irradiation: 5-year survival analysis of a phase 3 randomised controlled trial. Eur J Cancer. 2015;51(4):451-463. doi:10.1016/j.ejca.2014.12.013
- Formenti SC, Gidea-Addeo D, Goldberg JD, et al. Phase I-II trial of prone accelerated intensity modulated radiation therapy to the breast to optimally spare normal tissue. J Clin Oncol. 2007;25(16):2236-2242. doi:10.1200/JCO.2006.09.1041
- Parulekar WR, Berrang T, Kong I, et al. Cctg Ma.39 tailor RT: a randomized trial of regional radiotherapy in biomarker low-risk node-positive breast cancer (NCT03488693). J Clin Oncol. 2019;37(15_suppl). doi:10.1200/jco.2019.37.15_suppl.tps602
- White JR, Anderson SJ, Harris EE, et al. NRG-BR007: a phase III trial evaluating de-escalation of breast radiation (DEBRA) following breast-conserving surgery (BCS) of stage 1, hormone receptor+, HER2-, rs ≤18 breast cancer. J Clin Oncol. 2022;40(16_suppl). doi:10.1200/jco.2022.40.16_suppl.tps613
Citation
S P, C N, S A, S R, A S. Radiation Therapy in Cancer of Accessory Breast Tissue: Questions for Treatment Guidelines, Case Series, and Literature Review. Appl Radiat Oncol. 2023;(1):31-37.
March 21, 2023