Radiation therapy in a pediatric patient with Gorham Stout syndrome
Images

CASE SUMMARY
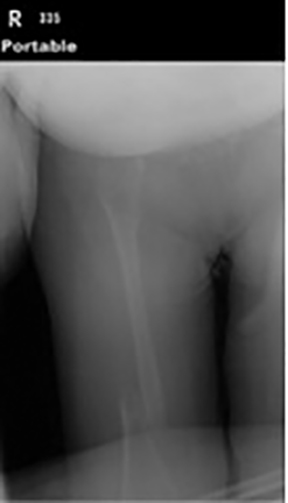
A 14 year-old female presented to the University of Arizona Cancer Center, Tucson, 8 years ago with a pathologic fracture in the right distal femur secondary to a diagnosis of Gorham Stout syndrome (GSS) established since infancy (Figure 1). She was successfully treated with radiation therapy (RT) and had a complete pain response.
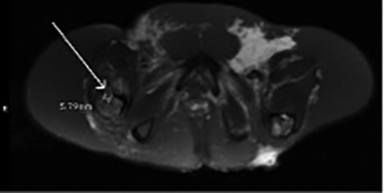
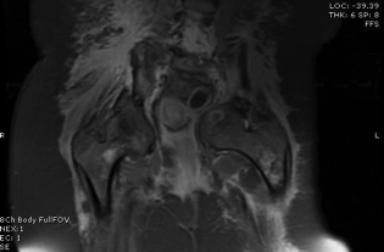
She returned to the clinic several months ago with severe pain in the right hip, requiring hospitalization. A magnetic resonance image (MRI) of the pelvis revealed cortical destruction of the anterior aspect of the proximal right femur at the intertrochanteric region secondary to lymphatic malformation consistent with GSS. She was treated with 3-dimensional conformal radiation therapy (3D-CRT) using 15 MV photons to the right femoral head to 3,000 cGy in 200 cGy fractions. The previous radiation fields were reviewed to avoid overlapping treatment fields.
The patient had excellent pain response to treatment and continues to be pain free 6 months later without the need for pain medications. Furthermore, she remains pain free in the previously treated distal right femur 8 years after treatment.
IMAGING FINDINGS
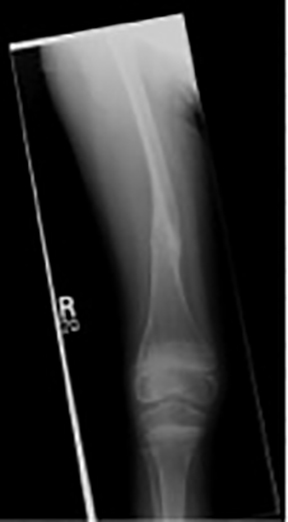
At age 6, the patient experienced a pathological fracture of the distal right femur related to her GSS (Figure 1). The lesion was treated successfully with 3,000 cGy in 300 cGy fractions with a complete pain response. A more recent radiograph revealed new osteolytic lesions in the right femoral head and neck.
An MRI of the pelvis demonstrated lymphatic malformation involving the anterior aspect of the right intertrochanteric region measuring approximately 6 mm (Figure 2). There was associated disruption of the cortex and extension of the lymphatic malformation into the adjacent musculature, as well as a small effusion in the right hip joint.
DIAGNOSIS
The patient’s presentation and radiographic evidence are consistent with a diagnosis of GSS. The differential diagnosis includes angiosarcoma, osteolytic metastases, juvenile Paget’s disease, Langerhans cell histiocytosis, and mastocytosis.
DISCUSSION
Prior to radiation therapy, the patient’s right hip lesion was managed with oral and IV pain medications, femoral nerve block, bisphosphonates, calcitonin, and immobilization with a pelvic girdle brace. Despite these efforts, the patient continued to complain of excruciating pain. She ultimately required conscious sedation during her CT simulation. The lesion was identified in the right femoral head and neck, and the images were reviewed and transferred to the Pinnacle planning system (Philips Healthcare, Andover, Massachusetts) for treatment planning.
Treatment with a standard anterior-posterior posterior-anterior (AP PA) field was initially planned. However, the patient’s pelvic girdle brace blocked a small portion of the treatment field and an additional right posterior oblique (RPO) field was required. As mentioned, the patient had a pathological fracture secondary to GSS located at the distal right femur 8 years ago. This was successfully treated to 3,000 cGy in 300 cGy fractions. Upon review, it was decided that the patient’s current lesion was situated reasonably outside of the old treatment field and could be safely treated.
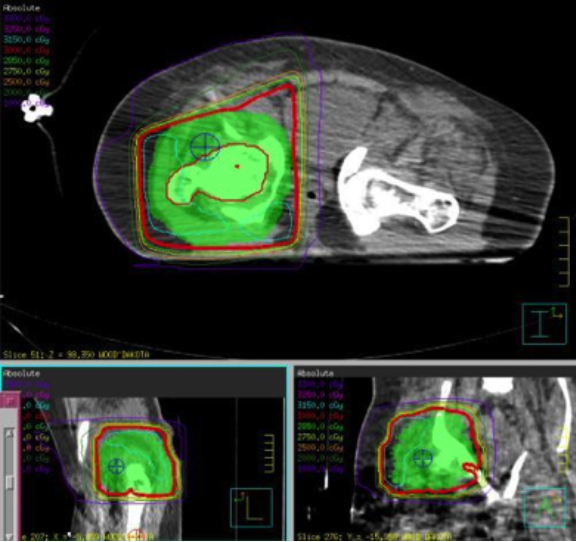
The patient was treated with 3D-CRT using 15 MV photons to the right femoral head to 3,000 cGy in 200 cGy fractions. The mean dose to the femur and femoral head were 3,074.3 and 3,110.9 cGy, respectively (Figure 3).
It is worth noting that the patient also required conscious sedation for each treatment visit. She could not tolerate being transferred from her hospital bed to the treatment table without anesthesia due to excruciating pain in her right hip. This did not cause any complications with her treatments, and she reported doing well at each on-treatment visit. Her pain began to improve significantly as the treatment dose approached the prescribed dose. She had a complete pain response after 2 weeks of treatment.
The patient completed her radiation therapy to the prescribed dose without interruptions or complications. She denied any adverse radiation effects, including skin erythema and desquamation. Six months after treatment, she reported improved mobility in her right leg, and continues to be pain free, while remaining off of all pain medications. Furthermore, the patient’s lesion in the right distal femur remains asymptomatic 8 years following radiation treatment.
GSS is a rare skeletal disorder characterized by unregulated interosseous lymphovascular proliferation leading to progressive bone resorption and/or destruction. It was initially described by Gorham et al1 in 1954, but its cause remains idiopathic with no evidence of infection, malignancy, or other etiology being described in the literature. Other names used to describe this disease include massive osteolysis, vanishing bone disease and phantom bone disease.2
In a recent case series by Hu et al.3, the mean age at diagnosis was 28 years. However, the proper diagnosis is often delayed months to years due to the rarity of this condition, and a high index of suspicion is required to arrive at an early diagnosis. Any part of the skeletal system can be involved and the most common sites are the shoulders, skull and pelvic girdle. A handful of cases have been described in the humerus.4 Most patients present with a progressive dull aching pain and the disease is rarely fatal. Other signs and symptoms include weakness and pathologic fracture.
Treatment options include surgical intervention, bisphosphonates, alpha 2B interferon and radiation therapy. Recently, Nir et al.5 reported that propranolol may be a therapeutic option for GSS. Hu et al. reported the results of a literature review consisting of 67 patients of GSS treated with different modalities.3 Surgery alone was the most popular treatment modality, with 27 patients. Only 6 patients were treated with radiation therapy alone, 4 of whom experienced symptom control with a mean follow-up of 14 months. The efficacy of one modality over another was not established in this study due to the limited number of cases.
Heyd et al.6 reported a retrospective series of 10 patients treated with radiation therapy for the management of GSS and found that 3,000 to 4,500 cGy conferred local control in 8 patients with a median follow-up of 42 months. Radiation was well-tolerated with no patient developing grade 2 or higher toxicity. Four patients developed grade 1 toxicity, including erythema and dysuria.
The mechanism by which radiation therapy controls disease progression and provides pain relief is unknown. We hypothesize that it causes lymphovascular involution similar to the effect of radiation therapy in lymphangiomatous malformations.
CONCLUSION
We report a pediatric patient with a painful intertrochanteric lesion secondary to GSS who responded well to radiation therapy. She had complete pain remission after treatment, and continues to be pain free 6 months after treatment without the need for pain medications. She was previously treated with radiation therapy to the distal femur for a separate lesion, which has remained pain free 8 years after treatment.
The diagnosis of GSS requires a high clinical suspicion, and it should be considered in the differential diagnosis in all children and adolescents presenting with extensive osteolysis. There are limited reports of GSS in the literature with no consensus on standard of care. In our experience, radiation therapy appears to be a well-tolerated and viable therapeutic option for symptomatic patients.
REFERENCES
- Gorham LW, Wright AW, Shultz HH, Maxon FC. Disappearing bones: a rare form of massive osteolysis: report of two cases, one with autopsy findings. Am J Med. 1954;17:674-682.
- Patel DV. Gorham’s disease or massive osteolysis. Clin Med Res. 2005;3(2):65–74.
- Hu P, Yuan XG, Hu XY. Gorham-Stout syndrome in mainland China: a case series of 67 patients and review of the literature. J Zhejiang Univ Sci B. 2013;14(8):729-35. doi:10.1631/jzus.B1200308.
- Yalniz E, Alicioglu B, Benlier E. Gorham-Stout disease of the humerus. JBR–BTR. 2008;91:14-17.
- Nir V, Guralnik L, Livnat G. Propranolol as a treatment option in Gorham–Stout syndrome: a case report. Pediatr Pulmonol. 2013. doi: 10.1002/ppul.22869.
- Heyd R, Micke O, Surhold C. Radiation therapy for Gorham-Stout syndrome: results of a national patterns-of-care study and literature review. Int J Radiat Oncolo Biol Phys. 2011;81(3):e179-85.