Radiation-induced Carotid and Vertebral Artery Stenosis in the Intensity-modulated Radiation Therapy Era: A Case Report
Images

Abstract
Carotid artery stenosis is a well-documented late complication of radiation therapy (RT) for head and neck cancers. Despite innovations in radiation delivery technique that spare uninvolved adjacent structures from undue radiation dose, vascular stenosis can be a life-threatening complication. We present a case of symptomatic radiation-induced internal carotid and vertebral artery stenosis and highlight the importance not only of early, but routine screening for arterial changes in post-radiation therapy head and neck cancer survivors in the intensity-modulated radiation therapy (IMRT) era.
Keywords: Carotid artery stenosis, head and neck cancer, vascular stenosis, vertebral artery stenosis, intensity-modulated radiation therapy, IMRT
Case Summary
A 45-year-old man with no significant past medical history presented with palpable left-sided neck adenopathy in 2010. He was a former smoker who, at the time, was still using chewing tobacco; however, he denied any cerebrovascular history. Examination was notable for a left-sided immobile submandibular mass, approximately 2 cm in diameter. Positron emission tomography – computed tomography (PET-CT) revealed multiple enlarged and F-18 fluorodeoxyglucose (FDG)-avid level II lymph nodes; fine needle aspiration of the neck mass detected malignant cells compatible with a head and neck squamous cell carcinoma (SCC) primary.
Subsequent biopsy of the left tonsil was positive for invasive, moderately-to-poorly differentiated SCC and in-situ hybridization for high-risk human papillomavirus (HPV) types, and was ultimately diagnostic of HPV-related pT1N2bM0 squamous cell carcinoma of the left tonsil.
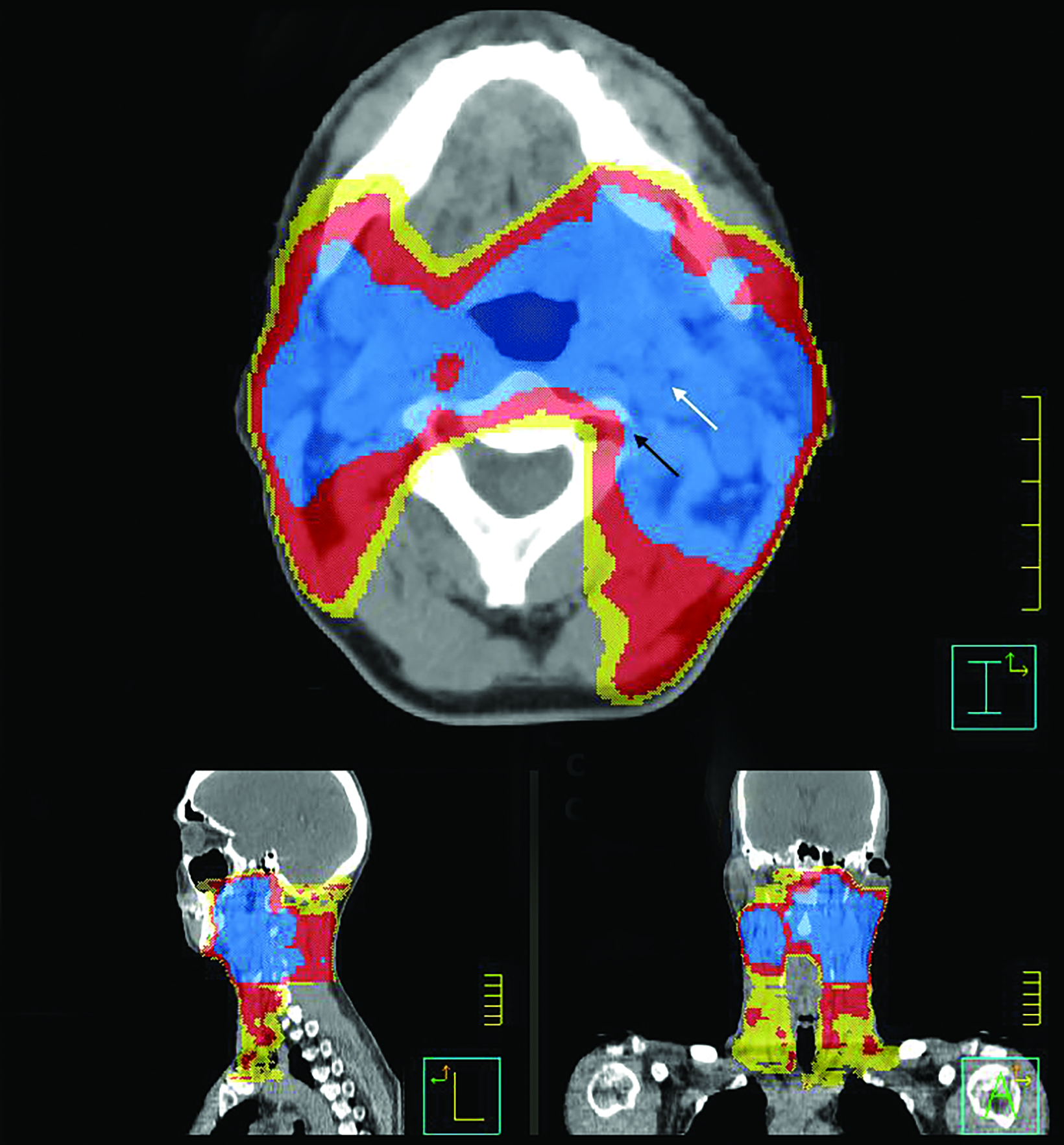
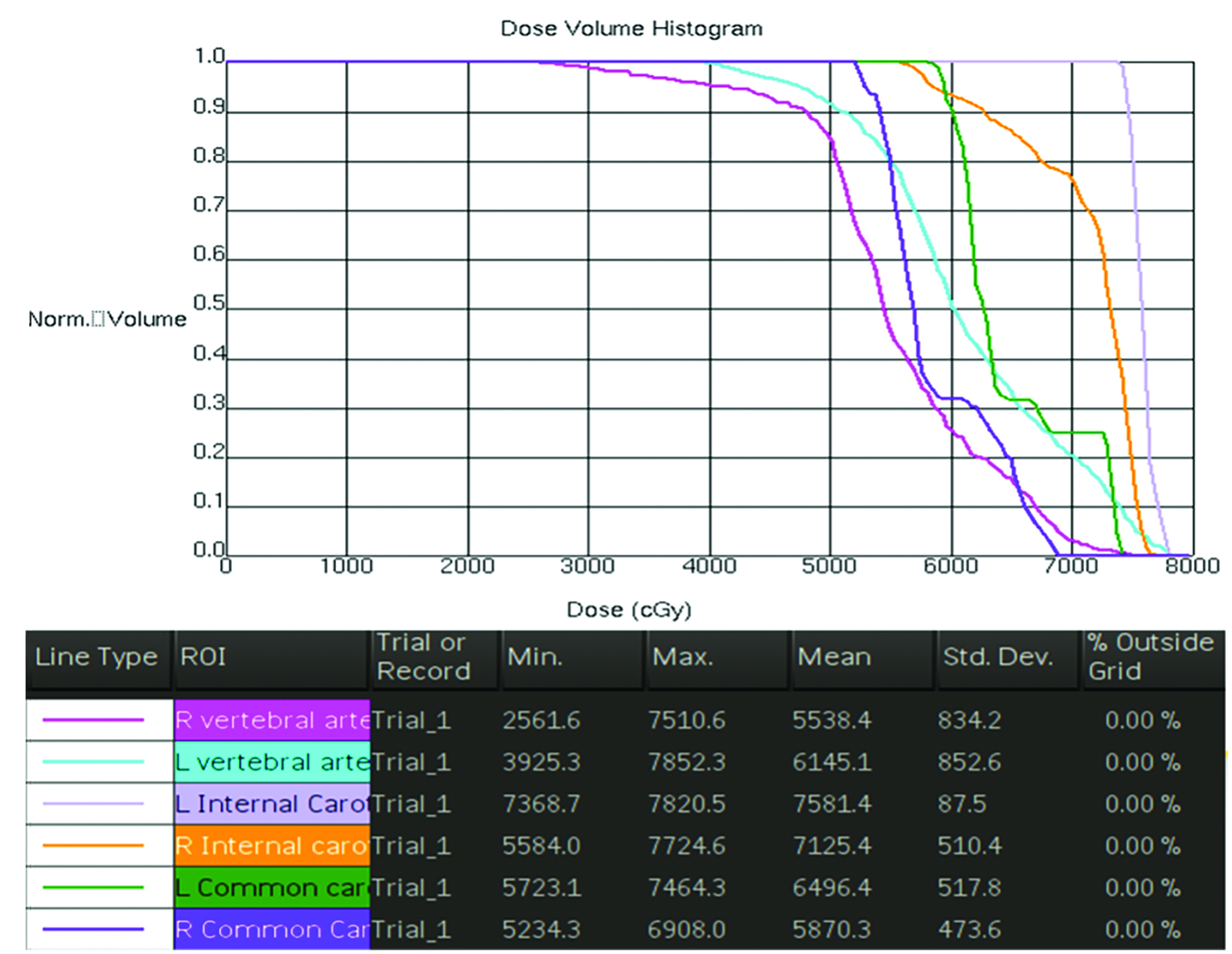
The patient was treated with definitive concurrent chemoradiation with weekly cisplatin (70 Gy to the primary site and involved nodes, 60 Gy to the intermediate-risk bilateral neck, and 54 Gy to the low-risk bilateral neck) (Figure 1). Notably, the patient received mean doses of 75.8 Gy and 71.3 Gy to the left and right carotid arteries; as well as 61.4Gy and 55.4Gy to the left and right vertebral artery, respectively (Figure 2). Following radiation therapy, the patient’s recovery was uncomplicated and during follow-up visits, there was no evidence of cancer recurrence.
Nearly 2 years following conclusion of chemoradiation, the patient presented for follow-up with complaints of intermittent symptoms of left-sided tongue paralysis and facial numbness in the mandibular division (V3). At the time, carotid ultrasound was ordered and there was no signs of stenosis or significant occlusion by report. Further evaluation with MRI of the brain demonstrated small, nonspecific foci of white matter change consistent with small vessel ischemic change but no evidence of acute infarct or an intra-cranial mass. No immediate further workup was performed.
In 2016, 6 years after the conclusion of chemoradiation, the patient presented to the emergency department with intermittent right-hand numbness and weakness for 3 days and was noted to be hypertensive, blood pressure (BP) 196/108 mmHg. On exam, raising his right arm to shoulder height reproduced his symptoms, with symptom resolution with other positions. Neurologic exam was otherwise unremarkable, with normal grip strength bilaterally and intact cranial nerves. Immediate MRI of the cervical spine was unremarkable, with no visible arterial occlusion; similarly, MRI of the brain showed no evidence of acute intracranial hemorrhage or infarct. Given this, the patient was discharged to cardiology for outpatient follow-up. Subsequent carotid ultrasound demonstrated a mild to moderate plaque (<50% stenosis) of the right internal carotid artery (ICA) and total occlusion of the left ICA. Workup also demonstrated elevated low-density lipoprotein (LDL).
Imaging Findings
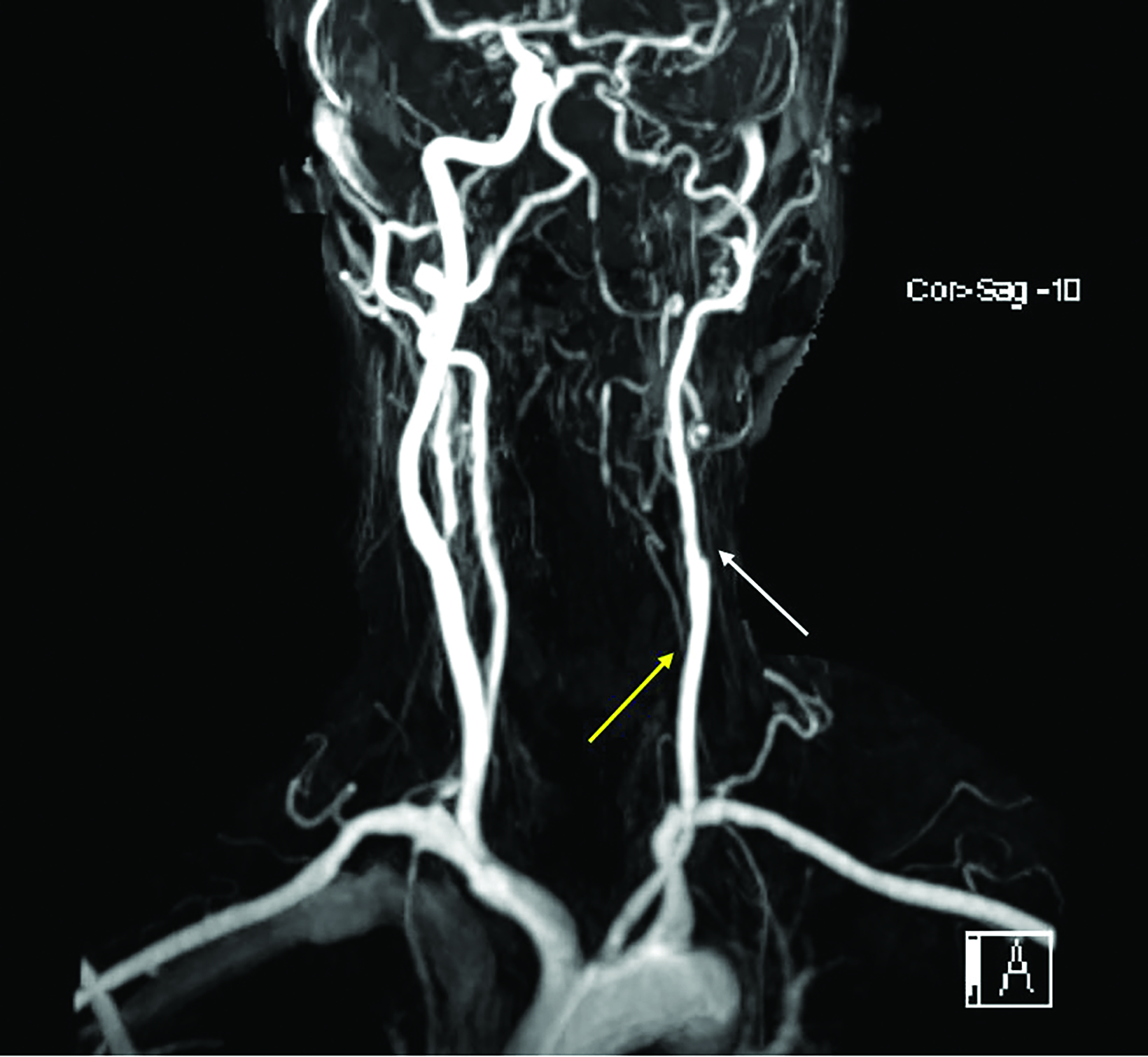
Under recommendation by cardiology, neurology, and interventional neurology, the patient underwent MR angiography, which redemonstrated complete occlusion of the left ICA with minimal flow 0.5 cm beyond the bifurcation of the common carotid but also found occlusion of the left vertebral artery at its origin, with muscular collaterals resulting in retrograde flow in the left V3 and V4 segments. (Figure 3). It also demonstrated compensatory flow from the right side with the posterior communicating artery to the middle cerebral artery.
Diagnosis
Symptomatic vertebrobasilar insufficiency secondary to radiation-induced carotid and vertebral artery stenosis. Although initial workup did not determine vertebral artery occlusion, symptoms correlated with cranial nerve V and XII involvement and can be localized by decreased perfusion along the brainstem, which is supplied by the vertebral artery. In addition, both locations of occlusion are found within the patient’s neck irradiation volumes.
Follow-up
Because revascularization was considered very high-risk, medical therapy was recommended, and the patient began a regimen of aspirin (325 mg), clopidogrel (75 mg), and rosuvastatin (20 mg). Following 3 months of this regimen, the patient was transitioned to clopidogrel monotherapy. Since that time, he has been free of symptoms and is followed with serial ultrasound to monitor patency of the right ICA, which has shown stability and possible improvement over time.
Discussion
Adjuvant and definitive RT are components of standard of care in the management of head and neck SCC. However, radiation-induced carotid artery stenosis is a well-recognized late toxicity of radiation therapy for head and neck cancer, with incidence ranging from 9% to as high as 30%.1-6 Although the pathophysiology of radiation-induced arterial stenosis remains incompletely understood, it is thought to occur both by direct endothelial injury and by indirect immune-modulated changes, in which radiation induces an
upregulation of inflammatory cytokines, inflammation, and an intensified innate immune response.7 Together, these processes induce accelerated atherosclerosis and vascular fibrosis that accumulate in the years after initial exposure to ionizing radiation. Carotid and vertebral artery stenosis has been documented in the treatment of oropharyngeal malignancies that necessitate full inclusion of the carotid arteries in treatment fields using conventional 2-D and 3-D conformal radiation therapy. We present an uncommon case of symptomatic radiation-induced vertebral and carotid artery stenosis in a patient treated with IMRT; in doing so, we highlight the importance of continued vigilance and surveillance for this complication.
Among patients with prior neck irradiation, vascular stenosis is well-documented and occurs most commonly in the ICA.8 Despite limited median follow-up duration for this chronic complication, all-cause stroke in patients who have underwent radiation therapy occurred at actuarial risk levels higher than age-matched controls9-12 (Table 1). Hence, radiation-induced artery stenosis represents a serious and highly morbid complication. Surveillance for carotid artery stenosis has relied on carotid ultrasound, but surveillance
schedule has been highly variable with little standardization, with first imaging in the literature ranging from 12 months to nearly two decades after conclusion of RT. Although there are no clear guidelines, we would recommend screening 2 to 3 years after completion of radiation or when patients develop symptoms of vascular occlusion. However, duplex ultrasound may be less sensitive in detecting vertebral artery stenosis and angiogram is the gold standard.13 Once symptomatic patients or those with severe stenosis are detected, treatments can include surgical endarterectomy or angioplasty and stenting. Medical treatment includes combination antiplatelet regimens (such as aspirin and clopidogrel) as well as management of comorbid hypertension or hyperlipidemia.14,15 As radiation treatment for HPV-related tonsillar cancer is highly effective, long-term follow-up after curative treatment is necessary.
Much of the existing literature on radiation-induced carotid artery stenosis is informed by data from the conventional and 3-D eras of radiation therapy, that is, prior to the early 2000s.16 Since then, there has been insufficient data on the incidence of long-term carotid artery stenosis in patients previously treated with IMRT, in part due to limited follow-up since the implementation of IMRT.
However, our case suggests that even in the IMRT era, unintended focal doses of radiation to the carotid and vertebral arteries may be sufficient to induce the atherosclerosis and stenosis secondary to conventional 3-D RT; this reinforces the importance of continued surveillance for vascular stenosis in head and neck cancer patients. Early diagnosis of mild carotid artery stenosis in these patients — and early, or even prophylactic medical therapy in high-risk patients — may ameliorate morbidity and mortality from full-blown occlusion at symptomatic presentation.17,18 With the continued development of vessel-sparing RT treatment plans and the use of more advanced RT techniques, including carotid sparing IMRT for early glottic cancers and vertebral artery sparing intensity-modulated proton therapy (IMPT)19,20, vascular injury and subsequent sequelae may decrease in incidence. Concurrently, evidence suggesting favorable outcomes of dose and target volume de-escalation regimens for elective regions in the treatment of HPV-positive oropharyngeal carcinoma may also decrease the incidence of future radiation-induced sequelae.21 However, this case highlights that at present, continued active surveillance remains necessary.
Future studies will need to examine the effects of IMRT planning and dose thresholds on carotid and vertebral arteries and how they may affect risk of stenosis and stroke. Furthermore, we need recommendations for survivorship and noninvasive screening in this specific population. Studies on how to integrate prevention of cerebrovascular disease including prophylactic use of angiotensin-converting enzyme inhibitors and statins are important for radiation oncologists and general practitioners alike.
Conclusion
Carotid artery stenosis is a well-documented late complication of radiotherapy for head and neck cancers. However, vertebral artery stenosis is a similarly serious long-term sequelae that is less commonly documented. Despite innovations in radiation delivery technique that spare uninvolved adjacent structures from undue radiation dose, routine screening for arterial changes in postradiation therapy head and neck cancer survivors in the IMRT era remains necessary.
References
- Chang YJ, Chang TC, Lee TH, Ryu SJ. Predictors of carotid artery stenosis after radiotherapy for head and neck cancers. J Vasc Surg. 2009;50(2):280-285. doi:10.1016/J.JVS.2009.01.033
- Cheng SWK, Ting ACW, Lam LK, Wei WI. Carotid stenosis after radiotherapy for nasopharyngeal carcinoma. Arch Otolaryngol Neck Surg. 2000;126(4):517-521. doi:10.1001/ARCHOTOL.126.4.517
- Cheng SWK, Wu LLH, Ting ACW, Lau H, Lam LK, Wei WI. Irradiation-induced extracranial carotid stenosis in patients with head and neck malignancies. Am J Surg. 1999;178(4):323-328. doi:10.1016/S0002-9610(99)00184-1
- Moritz MW, Higgins RF, Jacobs JR. Duplex imaging and incidence of carotid radiation injury after high-dose radiotherapy for tumors of the head and neck. Arch Surg. 1990;125(9):1181-1183. doi:10.1001/ARCHSURG.1990.01410210107017
- Carmody BJ, Arora S, Avena R, et al. Accelerated carotid artery disease after high-dose head and neck radiotherapy: is there a role for routine carotid duplex surveillance? J Vasc Surg. 1999;30(6):1045-1051. doi:10.1016/S0741-5214(99)70042-X
- Brown PD, Foote RL, McLaughlin MP, et al. A historical prospective cohort study of carotid artery stenosis after radiotherapy for head and neck malignancies. Int J Radiat Oncol. 2005;63(5):1361-1367. doi:10.1016/J.IJROBP.2005.05.046
- Yang EH, Marmagkiolis K, Balanescu D V., et al. Radiation-induced vascular disease—a state-of-the-art review. Front Cardiovasc Med. 2021;8:223. doi:10.3389/ FCVM.2021.652761/BIBTEX
- Tallarita T, Oderich GS, Macedo TA, et al. Reinterventions for stent restenosis in patients treated for atherosclerotic mesenteric artery disease. J Vasc Surg. 2011;54(5). doi:10.1016/J.JVS.2011.06.002
- Lam T, Choi H, Yeung I. Progressive increase in the risk of stroke among survivors of nasopharyngeal cancer treated with radical radiation therapy: 13-year follow-up of the 1998-2004. J Rad Onc Bio Phys. 2019; 105(1): Supp 212-213. Accessed December 9, 2021 via https://www.redjournal.org/article/S0360-3016(19)31124-1/abstract
- Haynes JC, Machtay M, Weber RS, Weinstein GS, Chalian AA, Rosenthal DI. Relative risk of stroke in head and neck carcinoma patients treated with external cervical irradiation. Laryngoscope. 2002;112(10):1883-1887. doi:10.1097/00005537-200210000-00034
- Dorresteijn LDA, Kappelle AC, Boogerd W, et al. Increased risk of ischemic stroke after radiotherapy on the neck in patients younger than 60 years. J Clin Onc. 2002; 20(1): 282-288. doi:10.1200/JCO.2002.20.1.282
- Hong JC, Kruser TJ, Gondi V, et al. Risk of cerebrovascular events in elderly patients after radiation therapy versus surgery for early-stage glottic cancer. Int J Radiat Oncol. 2013;87(2):290-296. doi:10.1016/J.IJROBP.2013.06.009
- Khan S, Cloud GC, Kerry S, Markus HS. Imaging of vertebral artery stenosis: a systematic review. J Neurol Neurosurg Psychiatry. 2007;78(11):1218. doi:10.1136/JNNP.2006.111716
- Xu J, Cao Y. Radiation-induced carotid artery stenosis: a comprehensive review of the literature. Interv Neurol. 2014;2(4):183-192. doi:10.1159/000363068
- Kocak B, Korkmazer B, Islak C, Kocer N, Kizilkilic O. Endovascular treatment of extracranial vertebral artery stenosis. World J Radiol. 2012;4(9):391. doi:10.4329/WJR.V4.I9.391
- Hong TS, Ritter MA, Tomé WA, Harari PM. Intensity-modulated radiation therapy: emerging cancer treatment technology. Br J Cancer. 2005;92(10):1819. doi:10.1038/SJ.BJC.6602577
- Trojanowski P, Sojka M, Trojanowska A, Wolski A, Roman T, Jargiello T. Management of radiation induced carotid stenosis in head and neck Cancer. Transl Oncol. 2019;12(8):1026. doi:10.1016/J.TRANON.2019.05.001
- Straub JM, New J, Hamilton CD, Lominska C, Shnayder Y, Thomas SM. Radiation-induced fibrosis: mechanisms and implications for therapy. J Cancer Res Clin Oncol. 2015;141(11):1985. doi:10.1007/S00432-015-1974-6
- Rosenthal DI, Fuller CD, Barker JL, et al. Simple carotid-sparing intensity-modulated radiotherapy technique and preliminary experience for T1-2 glottic cancer. Int J Radiat Oncol Biol Phys. 2010;77(2):455-461. doi:10.1016/J.IJROBP.2009.04.061
- Lin YH, Cheng JY, Huang BS, et al. Significant Reduction in Vertebral Artery Dose by Intensity Modulated Proton Therapy: A Pilot Study for Nasopharyngeal Carcinoma. J Pers Med. 2021;11(8). doi:10.3390/JPM11080822
- Tsai CJ, McBride SM, Riaz N, et al. Evaluation of substantial reduction in elective radiotherapy dose and field in patients with human papillomavirus-associated oropharyngeal carcinoma treated With definitive chemoradiotherapy. JAMA Oncol. 2022;8(3):364-372. doi:10.1001/JAMAONCOL.2021.6416
Citation
E L, S G, L C. Radiation-induced Carotid and Vertebral Artery Stenosis in the Intensity-modulated Radiation Therapy Era: A Case Report. Appl Radiat Oncol. 2022;(2):30-34.
July 15, 2022