Proton therapy for pituitary adenoma
Images

Pituitary adenomas arise from the adenohypophysis and represent approximately 10% to 15% of all primary brain tumors. Tumor classification is divided by size and functional characteristics.1 Morbidities owing to tumor size include visual and neurological defects due to proximity to the optic chiasm and cavernous sinuses. Perhaps the most important distinction in classifying pituitary adenomas is functional capacity. Secretory adenomas may cause potentially fatal biochemical imbalances because of overproduction of pituitary hormones like prolactin, growth hormones, adrenocorticotropic hormones and, more infrequently, thyroid-stimulating hormones.2 Standard treatment for nonfunctioning macroadenomas is transsphenoidal resection, and functioning adenomas can be medically managed when indicated.
Radiation therapy (RT) is used in the adjuvant setting after a subtotal resection or as a primary treatment for symptomatic primary or recurrent gross disease that is not amenable to surgical excision and cannot be medically managed. External-beam RT (EBRT) results in excellent radiographic disease control rates, ranging from 80% to 98% in nonfunctioning adenomas and 67% to 89% in functioning adenomas.3 While photon-based RT has consistently produced high tumor control with low toxicity, room remains for improving the therapeutic ratio, especially in younger patients who may be at greatest risk from radiation-induced late effects. Hypopituitarism of 1 or more axes is by far the most common adverse effect, with a 20% 5-year incidence rising to nearly 80% within 15 years of follow-up. Less frequent toxicities include visual and neurological complications, secondary tumors, cerebral vascular accidents, and cerebral necrosis.4,5
The advantage of proton therapy over conventional RT is a potential for decreased late effects of radiation attributable to lower doses to adjacent normal tissues. While there is little hope that pituitary function will be spared, additional toxicities may be avoided given the more favorable dose distribution. Dosimetric studies comparing different radiotherapy modalities suggest proton therapy could improve the therapeutic ratio in pituitary adenoma treatment by reducing the dose to the retinas, optic nerves, brainstem, and temporal lobes compared with conventional photon techniques including intensity-modulated radiation therapy (IMRT).6,7 In addition, proton therapy reduces the dose to the hippocampi, thus lowering radiation exposure to the neural stem cells, which may lessen the neurocognitive impact of radiotherapy.8 To date, the literature regarding proton therapy for pituitary adenoma is sparse. We have conducted a retrospective review of patients treated at our institution with proton therapy for pituitary adenoma in an effort to contribute to the literature.
Patients and methods
In accordance with an institutional review board-approved protocol and the Health Insurance Portability and Accountability Act (HIPAA), we reviewed the medical records of 17 patients with pituitary adenomas treated between 2007 and 2013 at the University of Florida Proton Therapy Institute in Jacksonville. All patients were treated with curative intent using three-dimensional conformal proton therapy. All patients were radiographically evaluated with computed tomography (CT) and/or magnetic resonance imaging (MRI) before and after treatment. In addition, all patients’ pituitary adenoma diagnoses were histologically confirmed prior to RT. Only benign pituitary tumors were included in the study; pituitary carcinomas were excluded from analysis. Patients treated with modalities other than transsphenoidal or transcranial surgical resection, such as stereotactic radiosurgery (SRS), were also excluded from our study.
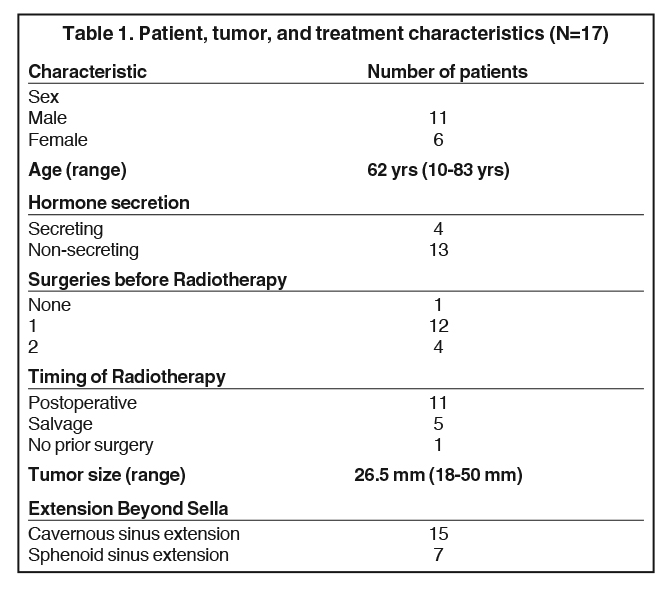
Follow-up was calculated from the date the patient initiated RT. Length of follow-up ranged from 0.3 to 5.7 years, with a median time of 3.0 years. Patient, tumor, and treatment characteristics are presented in Table 1. All but 1 patient underwent surgery before proton therapy. Of the 16 who received surgery, 15 underwent transsphenoidal resection, and 1 was resected via transcranial approach followed by a second transsphenoidal operation. In total, 5 patients received a second operation before proton therapy. All patients had measurable gross disease at the time of proton therapy so that no patient was classified as undergoing a gross total resection. The predominant reason for the proton therapy referral was locally invasive disease (15 patients had a cavernous sinus invasion). Proton therapy was delivered as adjuvant treatment in 11 patients, salvage therapy for a recurrence in 5 patients, and definitive treatment in 1 patient. For patients undergoing adjuvant therapy, the median interval from surgery to initiation of proton-based irradiation was 114 days (range, 45–283 days).
Radiation treatment
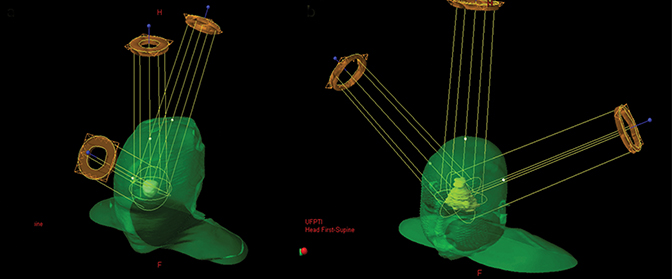
All 17 patients were treated with three-dimensional double-scattered conformal proton therapy (3DCPT) in a continuous course of 5 fractions per week at 1.8 Gy relative biological effectiveness (RBE) per fraction. For each patient, pre- and postoperative treatment planning MRIs were co-registered to the treatment planning CT. Target volumes were defined as both the pre- and postoperative gross tumor volumes, with the clinical target volume (CTV) adding a 5-mm margin off the gross tumor volume to account for tumor spread. The planning target volume (PTV) was defined as the CTV with an additional 3-mm margin. Two 3-field beam arrangements were used—either a mohawk (Figure 1A) or a 2- lateral oblique, superior-anterior oblique arrangement (Figure 1B). Beam-shaping apertures were designed based on a customized expansion of the PTV projection in the beam’s eye-view of approximately 5 to 7 mm. Customized beam compensators were individually designed to maximize dose conformality and reduce the effects of tissue heterogeneity on the dose distribution. Total dose ranged from 45 to 50.4 Gy (RBE) (median, 45 Gy [RBE]). Plans were normalized such that 99% of the CTV was covered by the prescription, which nearly always meant that 95% of the PTV received 95% of the prescription.
Statistical methods
JMP software (SAS Institute, Cary, NC) was used to compute the Kaplan-Meier product limit estimates for local control, progression-free survival, and cause-specific survival.
Results
Local control
The 3-year radiographic local control rate for both secreting and non-functional pituitary adenomas after treatment with proton therapy was 100%, meaning all patients exhibited either stabilization or regression in tumor size. Objective measures of biochemical control were not available for the 4 patients with secreting tumors. Of these, 3 patients reported no signs, symptoms or biochemical evidence that they remained hypersecretory. It was not evident at what time their baseline hypersecretion normalized. The patient with a tumor secreting growth hormones continues to visit her local endocrinologist and reports no endocrine-related symptoms.
Survival
The 3-year overall survival rate was 100%. There was 1 intercurrent death that occurred at 4.98 years after treatment due to cardiovascular disease.
Complications
Several factors potentially lead to adverse neurological events, including surgery, radiation therapy, tumor mass effect, and hormonal secretion. The most commonly observed side effect was hypopituitarism, evident in 11 patients following RT. Table 2 shows the presence of pituitary dysfunction after both surgery and RT. All but 1 of the patients who were hormone-deficient after proton therapy had baseline pituitary dysfunction. In this series, no other major complications, such as cerebrovascular accidents, decline in visual function, ototoxicity, and second malignancies, have been observed as of the most recent follow-up. Objective neurocognitive function was not available for most patients, but all patients who were alive at the time of data collection report no signs or symptoms of significant cognitive deficits.
Dosimetric outcomes
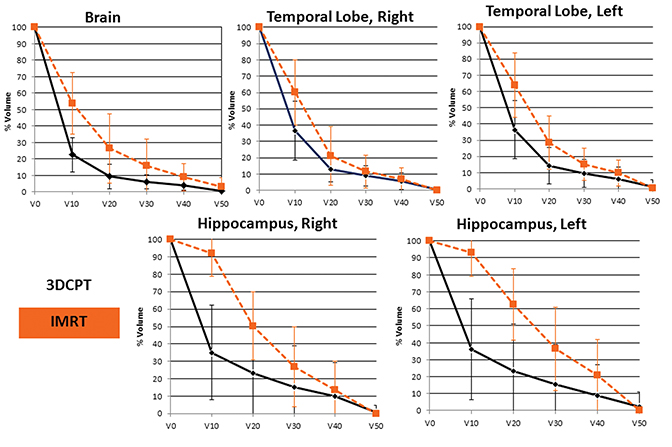
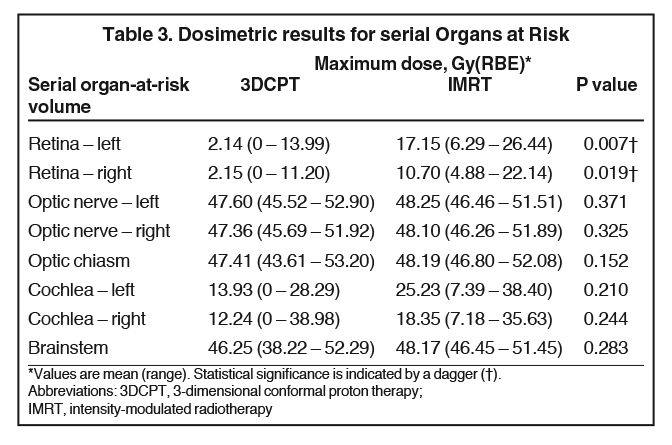
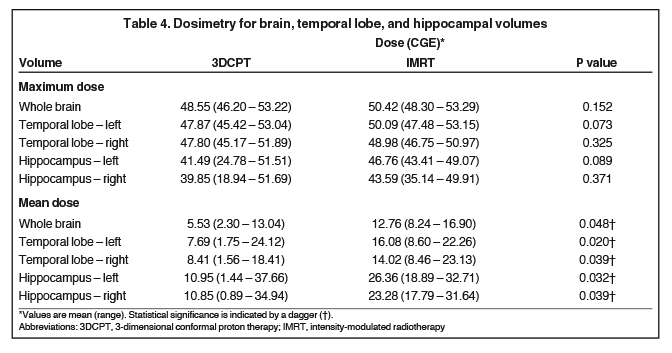
Dosimetric data were reviewed for all 17 proton plans. In addition, 4 IMRT comparison plans were generated, normalized to the same target coverage achieved with the proton plans. Table 3 shows the mean maximum doses to serial organs at risk (OARs) for both the 3DCPT and IMRT plans. Compared to the IMRT plans, the left and right retinae received lower doses with 3DCPT; however, none of the doses delivered to serial OARs with either technique are expected to result in significant normal tissue complications. Nevertheless, these data show that proton therapy did not result in any unacceptable physical dose heterogeneity within serial OARs. Comparison of both maximum and mean doses to the whole brain, temporal lobes, and hippocampi are presented in Table 4. On average, the proton plans produced lower doses to whole brain, temporal lobes and hippocampi. Average dose-volume histograms are shown in Figure 2, demonstrating that most of the benefits of proton therapy were seen from a reduction in the low and moderate doses to these organ-at-risk volumes (ORVs).
Discussion
In the management of pituitary adenomas, surgical resection alone yields control rates that substantially differ by tumor characteristics. A large series by Mortini and colleagues9 reported control rates of 55.5% in macroadenoma patients, compared to 78.9% for microadenomas. Much poorer outcomes were reported in tumors invading the cavernous sinuses, at 7.4%. While surgical resection is often indicated as a first line of treatment for these tumors, recurrence after surgery alone is 19% vs. 2% in patients receiving surgery and RT.10 RT is an effective treatment modality either postoperatively when the likelihood of recurrence is high, or definitively when tumors are unresectable and cannot be medically managed.
Long-term outcomes of patients treated with postoperative conventional RT have been well-documented in the scientific literature. In one of the largest and most-cited analyses, Brada et al. reported the outcomes of 411 patients, of which 252 had non-functioning adenomas, 131 had functional adenomas, and the remaining 28 were of unknown secretory status. At 10 years, the progression-free survival rate was 94%, and at 20 years it was 88% for all patients. The only factor affecting prognosis in this study was hormone secretion.11 In 2008, Chang et al. reported the outcomes of adjuvant RT in 663 patients with nonfunctioning pituitary adenomas, with progression-free survival rates of 93% at 5 years, 87% at 10 years, and 74% at 20 years. Out of these patients, cavernous sinus involvement was the only significant prognostic factor.12 Snead and colleagues reviewed the records of 100 patients with pituitary adenomas, 69 of which were nonfunctioning and 31 were functioning. Overall, the 10-year progression-free survival rate was 95% for nonfunctioning and 88% for functioning adenomas. No statistically significant variables influenced prognosis in this study. 2 A 2009 study by Erridge et al. reported the progression-free survival rate of 385 patients treated with RT to be 97% at 10 years, and 96% at 20 years. No identifiable factors affected control rates in this study.13
In contrast to conventional RT, the outcomes of patients treated with fractionated proton therapy either definitively or adjuvantly are less well-documented. While several studies have reported outcomes of both conventional and proton-based SRS for treating pituitary adenomas, SRS is best indicated for tumors < 3 cm in diameter and further than 5 mm from the optic chiasm.4 In the only other series reporting outcomes using fractionated proton therapy to date, Ronson and colleagues analyzed 47 patients treated with fractionated proton therapy. They observed 100% radiographic local control of all 41 patients who had available follow-up, with a median follow-up of 3.9 years.14 These control rates are consistent with our results (100% at 3 years). A recent review by Loeffler et al. estimates that RT achieves biochemical remission rates of approximately 50% at 10 years, with these rates enhanced by concomitant medical management.4 Our series reports treatment of 4 patients with functioning tumors—2 with prolactinomas, 1 with a growth hormone-secreting tumor, and 1 with an adrenocorticotropic hormone-secreting tumor. Unfortunately, objective endocrine follow-up was unavailable in these patients. Nevertheless, all 4 patients report no signs, symptoms or other evidence of hypersecretion.
The most common complication of RT, by far, is hypopituitarism of 1 or more hormonal axes. The literature suggests that this toxicity requires many years to develop. With fractionated RT, radiation-associated endocrinopathies is seen in roughly 20% of patients after 5 years of follow-up. Some studies have revealed pituitary decline to reach as high as 80% in patients after 10 years of follow-up data.4 In our series, only 5 patients had normal pituitary function before RT, while the remainder had existing postoperative pituitary dysfunction. We observed 1 of those 5 patients develop new-onset hypopituitarism associated with RT (Table 2). With our median follow-up of 3.9 years, this rate is consistent with the current literature.
Other documented complications of RT include visual decline, cerebrovascular accidents, ototoxicity, temporal lobe necrosis, and secondary brain tumors. These toxicities are fortunately rare, and often do not manifest until many years after treatment. Perhaps the most documented of these extra-pituitary events is injury to the optic pathways, with a 1.5% likelihood at 20 years after RT, and radiation-induced tumors, likely in 1.9% at 20 years.11,13 Ronson and colleagues reported 1 case of temporal lobe necrosis 19 months after treatment. Several factors may have contributed to this event, but it is noteworthy that this patient received 54 Gy (RBE) in 2 Gy fractions.14 While, fortunately, we report none of these complications in our series, continued follow-up is required to adequately assess such toxicities, as the incidence of these events slowly rises over time.
The rationale for particle therapy treatment such as fractionated proton therapy stems from a phenomenon known as the Bragg peak, which allows dose escalation to a target volume while sparing adjacent peripheral structures. Proton therapy has garnered particular interest in the treatment of intracranial tumors, especially as the importance of neuroprotection in radiation therapy is becoming increasingly realized. Neural stem cells serve a central role in neuroplasticity, with reserves located primarily in the subventricular zone as well as the subgranular layer of the hippocampal dentate gyrus.15,16 Conventional radiation therapies that do not spare these areas have been shown to damage hippocampal neurogenesis, contributing to the neurocognitive decline in patients treated for many intracranial tumors.17,18 Dosimetric comparisons have established that proton-based modalities have the potential to better spare these structures vs. conventional techniques in treating intracranial tumors.8,9
Recent prospective data support these hypotheses; increased doses to the temporal lobes and hippocampi significantly impair patients’ performances on standardized neurocognitive tests.20 Dose-cognitive effect models have also been applied in dosimetric comparisons to estimate the improved preservation of IQ in patients who receive proton therapy.21 But the relationship between brain irradiation and neurocognition is not entirely agreed upon. Some cross-sectional studies have found no significant differences in cognitive performance between patients with pituitary adenomas receiving surgery plus postoperative conventional RT, and patients receiving surgery alone.22,23 These studies analyzed patients with median ages between 55 and 61, whereas the Redmond et al. study included only pediatric patients.20 The patients in our study have a median age of 63 years, yet ages range from 10-83; thus, our findings may be difficult to apply uniformly. In addition, ongoing clinical trials such as RTOG 0933 aim to further assess the potential benefits of hippocampal avoidance and the relationship between radiation dose and cognitive function.
Our dosimetric analysis aimed to assess dosage differences in several serial OARs by comparing the 17 proton treatment plans used in our patients to 4 equivalent IMRT plans generated from our series. Of note, temporal lobe and hippocampal avoidance were objectives in the IMRT planning process. Despite specific goals to avoid these structures in IMRT planning, the whole brain, both temporal lobes, and both hippocampi were spared using 3DCPT. Dose-volume histograms of the 5 aforementioned structures also show significant decreases in the volume receiving up to 10 Gy (RBE; V10) in all 5 structures, as well as the V20 of the whole brain. Reducing doses to structures outside the tumor volume may potentially mitigate the unwanted effects of therapy on surrounding tissues. While we used no objective measures of neurocognitive function during follow-up of our patients, the dosimetric advantages characterized in our series may be of further interest given our growing understanding of RT doses to specific brain structures and cognitive impairment.
Conclusion
Our study demonstrates the feasibility of delivering proton therapy for pituitary adenoma. The high conformality of proton therapy does not appear to compromise local control and there is no increased early toxicity. Given the results of RTOG 0933, the lower dose to the hippocampi and temporal lobes should reduce the neurocognitive impact of radiotherapy. This greatest benefit will likely be in younger patients who are expected to have long-term survival.
References
- Ezzat S, Asa SL, Couldwell WT, et al. The prevalence of pituitary adenomas: A systematic review. Cancer. 2004;101:613-619.
- Snead FE, Amdur RJ, Morris CG, et al. Long-term outcomes of radiotherapy for pituitary adenomas. Int J Radiat Oncol Biol Phys. 2008;71:994-998.
- Becker G, Kocher M, Kortmann RD, et al. Radiation therapy in the multimodal treatment approach of pituitary adenoma. Strahlenther Onkol. 2002;178:173-186.
- Loeffler JS, Shih HA. Radiation therapy in the management of pituitary adenomas. J Clin Endocrinol Metab. 2011;96:1992-2003.
- Brada M, Ashley S, Ford D, et al. Cerebrovascular mortality in patients with pituitary adenoma. Clin Endocrinol (Oxf). 2002;57:713-717.
- Bolsi A, Fogliata A, Cozzi L. Radiotherapy of small intracranial tumours with different advanced techniques using photon and proton beams: A treatment planning study. Radiother Oncol. 2003;68:1-14.
- Winkfield KM, Niemierko A, Bussiere MR, et al. Modeling intracranial second tumor risk and estimates of clinical toxicity with various radiation therapy techniques for patients with pituitary adenoma. Technol Cancer Res Treat. 2011;10:243-251.
- Boehling NS, Grosshans DR, Bluett JB, et al. Dosimetric comparison of three-dimensional conformal proton radiotherapy, intensity-modulated proton therapy, and intensity-modulated radiotherapy for treatment of pediatric craniopharyngiomas. Int J Radiat Oncol Biol Phys. 2012;82:643-652.
- Mortini P, Losa M, Barzaghi R, et al. Results of transsphenoidal surgery in a large series of patients with pituitary adenoma. Neurosurg. 2005;56:1222-1233; discussion 1233.
- Chun M, Masko GB, Hetelekidis S. Radiotherapy in the treatment of pituitary adenomas. Int J Radiat Oncol Biol Phys. 1988;15:305-309.
- Brada M, Rajan B, Traish D, et al. The long-term efficacy of conservative surgery and radiotherapy in the control of pituitary adenomas. Clin Endocrinol (Oxf). 1993;38:571-578.
- Chang EF, Zada G, Kim S, et al. Long-term recurrence and mortality after surgery and adjuvant radiotherapy for nonfunctional pituitary adenomas. J Neurosurg. 2008;108:736-745.
- Erridge SC, Conkey DS, Stockton D, et al. Radiotherapy for pituitary adenomas: Long-term efficacy and toxicity. Radiother Oncol. 2009;93:597-601.
- Ronson BB, Schulte RW, Han KP, et al. Fractionated proton beam irradiation of pituitary adenomas. Int J Radiat Oncol Biol Phys. 2006;64:425-434.
- Eriksson PS, Perfilieva E, Bjork-Eriksson T, et al. Neurogenesis in the adult human hippocampus. Nat Med. 1998;4:1313-1317.
- Doetsch F, Caille I, Lim DA, et al. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell. 1999;97:703-716.
- Monje ML, Vogel H, Masek M, et al. Impaired human hippocampal neurogenesis after treatment for central nervous system malignancies. Ann Neurol. 2007;62:515-520.
- Dietrich J, Monje M, Wefel J, et al. Clinical patterns and biological correlates of cognitive dysfunction associated with cancer therapy. Oncologist. 2008;13:1285-1295.
- Blomstrand M, Brodin NP, Munck Af Rosenschold P, et al. Estimated clinical benefit of protecting neurogenesis in the developing brain during radiation therapy for pediatric medulloblastoma. Neuro Oncol. 2012;14:882-889.
- Redmond KJ, Mahone EM, Terezakis S, et al. Association between radiation dose to neuronal progenitor cell niches and temporal lobes and performance on neuropsychological testing in children: a prospective study. Neuro Oncol. 2013;15:360-369.
- Merchant TE, Hua CH, Shukla H, et al. Proton versus photon radiotherapy for common pediatric brain tumors: comparison of models of dose characteristics and their relationship to cognitive function. Pediatr Blood Cancer. 2008;51:110-117.
- van Beek AP, van den Bergh AC, van den Berg LM, et al. Radiotherapy is not associated with reduced quality of life and cognitive function in patients treated for nonfunctioning pituitary adenoma. Int J Radiat Oncol Biol Phys. 2007;68:986-991.
- Brummelman P, Elderson MF, Dullaart RP, et al. Cognitive functioning in patients treated for nonfunctioning pituitary macroadenoma and the effects of pituitary radiotherapy. Clin Endocrinol. (Oxf). 2011;74:481-487.