Presentation of pituitary carcinoma as neck metastasis after irradiation of recurrent pituitary macroadenoma
Images

Case contest winner September 2015
CASE SUMMARY
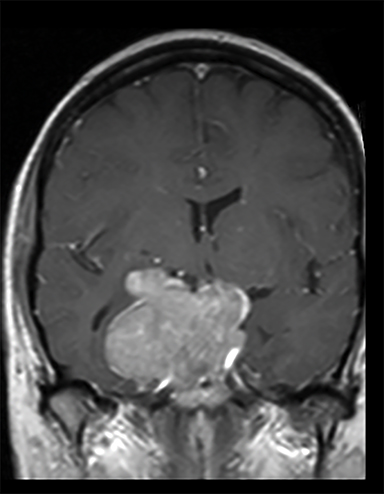
A 56-year-old female presented with acute-onset third cranial nerve palsy. Her history is significant for a pituitary macroadenoma (Figure 1A) treated with subtotal resection (STR) 6 years prior (Figure 1B). One year after STR, she developed progressive disease and underwent intensity-modulated radiotherapy (IMRT) (45 Gy/25 fractions).
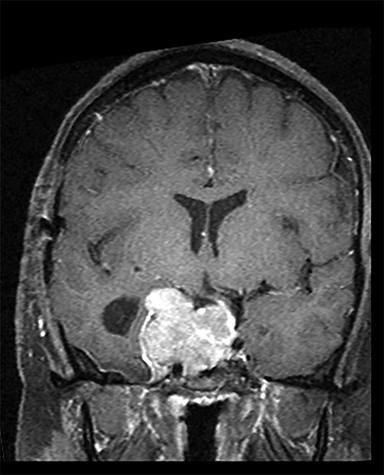
Three years after completing IMRT, MRI again demonstrated disease progression (Figure 2A) requiring repeat STR. Pathology dem onstrated pituitary adenoma with Ki-67 of 75%. Six weeks postoperatively, the patient required admission for rapidly progressive lethargy. MRI demonstrated further progression; given the aggressive nature of this lesion and her prior radiotherapy, she underwent concurrent pulsed low-dose rate IMRT (54 Gy/27 fractions) with temozolomide (TMZ) (Figure 2B).1,2
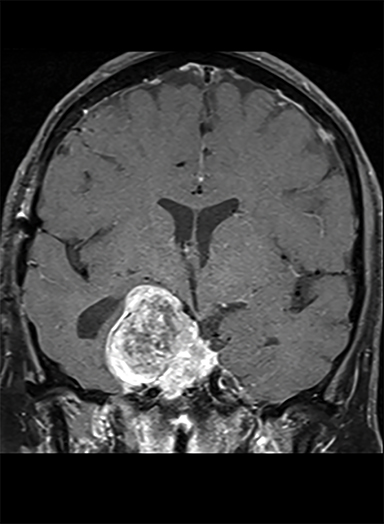
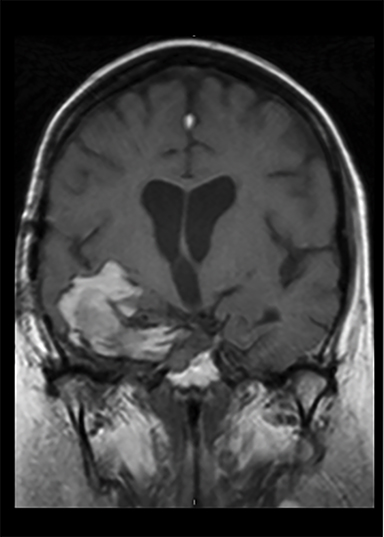
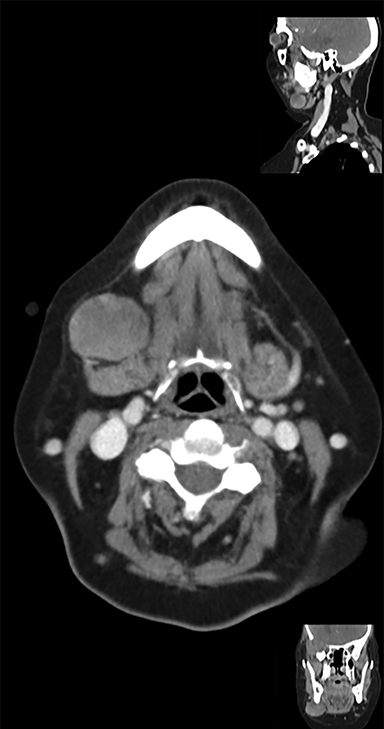
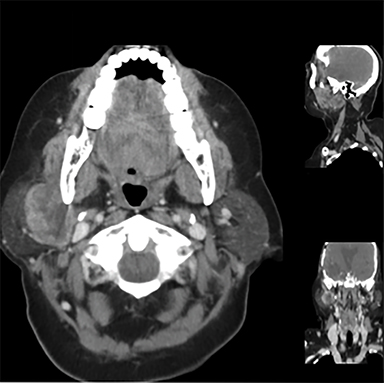
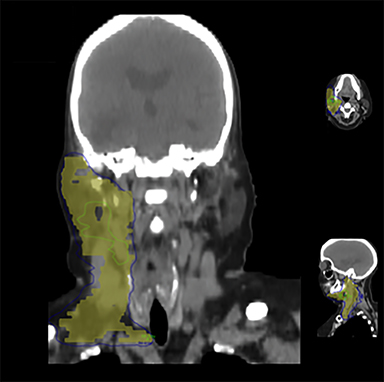
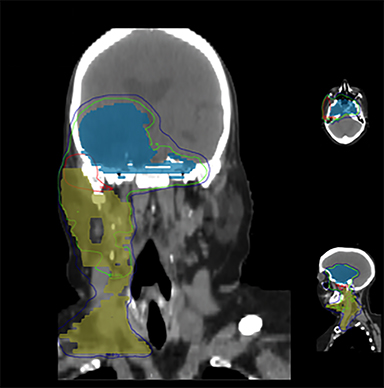
One month after completing chemoradiotherapy, repeat MRI demonstrated stable sellar disease with blood products in the right middle cranial fossa (Figure 3A). However, at this time she detected a right submandibular mass (Figure 3B). Biopsy was consistent with metastatic pituitary carcinoma and she underwent a resection of the right submandibular gland and lymph node. Three months later, she developed a right parotid mass (Figure 4A) requiring parotidectomy and supraomohyoid neck dissection, again consistent with metastasis. Postoperatively, she underwent unilateral neck IMRT (55 Gy/20 fractions) (Figure 4B) utilizing an accumulated dose plan (Figure 4C).
IMAGING FINDINGS
Imaging of the submandibular mass demonstrated a heterogeneous, mildly enhancing 3.4 cm lymph node adjacent to the right submandibular gland (Figure 3B). At the time of her parotid metastasis, CT of the neck demonstrated a 3.1 cm right parotid mass (Figure 4A) extending into the deep lobe of the parotid gland.
DIAGNOSIS
Pituitary macroadenoma with malignant transformation to a pituitary carcinoma accompanied by right parotid and submandibular lymph node metastases.
DISCUSSION
While pituitary adenomas are common benign tumors, they nevertheless may result in significant morbidity secondary to mass effect or secretory phenomena. Invasive adenomas, which account for 25-55% of adenomas, exhibit more aggressive and locally invasive behavior.3 Although the 2004 WHO classification of pituitary tumors is grounded in secretory products, a subset of “atypical” or “aggressive” invasive adenomas was also delineated.3,4 These adenomas are typically characterized by a high mitotic index, Ki-67 ≥ 3%, and extensive positive staining for p53. The significance of this delineation is apparent in the 2 proposed tumorigenesis models of pituitary carcinomas: the sequential and de novo models.4 While the first model reflects an adenoma-to-carcinoma sequence, the second suggests that aggressive adenomas form de novo with the potential for subsequent malignant transformation. In both models, the significance of the aggressive adenoma is paramount, as it represents the precursor lesion to pituitary carcinoma in the vast majority of cases.
Given the rarity of metastasis in patients with primary pituitary lesions, it has historically been difficult to distinguish pituitary carcinoma from aggressive adenoma.5 Moreover, most patients with pituitary carcinoma exhibit histories such as that described here, with sequential progression from benign adenomas with low proliferative indices to aggressive adenomas with the ultimate development of metastasis.3 Despite this challenge, pituitary carcinomas are exceedingly rare. While pituitary tumors account for 10-15% of central nervous system neoplasms, pituitary carcinomas represent 0.1-0.2% of pituitary tumors and require evidence of craniospinal or systemic metastasis for diagnosis.3,6,7
The natural history of pituitary carcinomas differs with respect to the degree of local invasion and sites of metastasis. The clinical features of invasion and mass effect include pain and cranial nerve palsies. In general, patients with macroadenomas may classically experience bitemporal hemianopsia, decreased visual acuity, ptosis, diplopia and facial numbness.3 While this report is of a nonsecretory carcinoma, the majority of pituitary carcinomas are secretory; as such, patients may present with signs and symptoms of Cushing’s disease, hyperprolactinemia, or acromegaly.7 Although the 5-year overall survival for invasive adenomas is approximately 80%, pituitary carcinoma confers a 30% 5-year survival, with systemic metastasis more common than craniospinal metastasis.8
Radiologic differentiation of pituitary carcinomas from aggressive adenomas is generally not possible in the absence of observed metastasis.8,9 Both lesions may reveal invasion of the clivus, sellar floor, and cavernous sinus with intracranial extension.3,4 Following definitive or adjuvant therapy, recurrence is not only more common but also more rapid among carcinomas, with a median time to recurrence of 6-12 months and a median time to metastasis of 5 years.3,7 Metabolic imaging with positron emission tomography/computed tomography (PET/CT) offers higher sensitivity than MRI for detection of metastasis; moreover, octreotate-bound nuclides such as 68Ga may be superior to 18F-FDG for detection of metastasis and local recurrence.10
Pathologic differentiation between aggressive adenomas and carcinomas is similarly challenging. Normal pituitary tissue exhibits a low rate of cellular division and, thus, adenomas generally exhibit Ki-67 ≤ 2%.4 Staining above 3% has been suggested to offer reasonable sensitivity (73%) and specificity (97%) for distinguishing invasive from noninvasive behavior.9,11 Nuclei for high-grade lesions are hyperchromatic and accompanied by prominent nucleoli. Immunohistochemical staining to p53 offers excellent sensitivity for malignant lesions: While 15% of invasive adenomas are p53 positive, 100% of carcinomas are positive.3,12 Benign and malignant histologies may both stain with antibodies to pituitary hormones; as such, this is less helpful for identifying carcinoma, but is crucial for medically directed therapies.7 Although no single test may diagnose pituitary carcinoma before observed metastasis, certain clinicopathologic prognostic criteria have been proposed, including a combination of Ki-67 > 3%, > 2 mitoses per high-powered field, and p53 positivity.9
Unfortunately, many patients with aggressive pituitary lesions will suffer multiple recurrences, which may require repeat radiotherapy. This presents a challenging situation due to nearby critical structures including the optic nerves, optic chiasm, brainstem and the carotid arteries. A repeat course of fractionated radiation therapy is a possibility for some patients after careful consideration of treatment options, interval since first course of radiation therapy, and details of prior radiation treatment.13
In this case, we extrapolated from the glioblastoma literature and utilized a pulsed low-dose rate technique in hopes of minimizing late toxicity.2,14 This technique relies on the radiobiological advantage of normal tissue repairing sublethal damage when exposed to dose-rates between 0.01 and 1.00 Gy/min. Furthermore, recent literature has indicated that cytotoxic chemotherapy with temozolomide (TMZ) can provide clinical and radiographic response rates of 60-70%.1 While MGMT (O-6-methylguanine-DNA methyltransferase) methylation is prognostic for glioblastoma, there appears to be no correlation between response to TMZ and MGMT methylation status in patients with aggressive pituitary tumors.15,16
In the present report, pathologic data highlighted this challenge in distinguishing aggressive adenoma from carcinoma. Tissue from the first stage of repeat STR demonstrated a Ki-67 index focally in excess of 75%, with 15-20% staining with p53. In the second stage, Ki-67 index was 3-4% focally and p53 staining was < 5%. One month later, correlation with MRI demonstrated a heterogeneously enhancing mass suggestive of marked recurrence, followed shortly thereafter by clinically evident metastasis.
CONCLUSION
In this report, a 56-year-old female exhibited an adenoma-to-carcinoma tumorigenesis sequence with metastasis to submandibular lymph nodes and the parotid gland. While differentiating aggressive adenomas from carcinomas is challenging, certain clinicopathologic criteria may offer insight into the risk for malignant transformation. This differentiation is clinically significant, as a diagnosis of pituitary carcinoma confers a 5-year survival of 30%.
Despite prognostic differences, both aggressive adenomas and pituitary carcinomas require multidisciplinary care. Emerging diagnostic and therapeutic technologies in the form of metabolic imaging and targeted therapies may provide additional benefit in the monitoring and treatment of these lesions.
REFERENCES
- Raverot G, Sturm N, de Fraipont F, et al. Temozolomide treatment in aggressive pituitary tumors and pituitary carcinomas: a French multicenter experience. J Clin Endocrinol Metab. 2010;95(10):4592-4599. doi:10.1210/jc.2010-0644.
- Adkison JB, Tomé W, Seo S, et al. Reirradiation of large-volume recurrent glioma with pulsed reduced-dose-rate radiotherapy. Int J Radiat Oncol Biol Phys. 2011;79(3):835-841. doi:10.1016/j.ijrobp.2009.11.058.
- Chatzellis E, Alexandraki KI, Androulakis II, Kaltsas G. Aggressive pituitary tumors. Neuroendocrinology. 2015;101(2):87-104. doi:10.1159/000371806.
- Di Ieva A, Rotondo F, Syro LV et al. Aggressive pituitary adenomas—diagnosis and emerging treatments. Nat Rev Endocrinol. 2014;10(7):423-435. doi:10.1038/nrendo.2014.64.
- Heaney AP. Clinical review: Pituitary carcinoma: difficult diagnosis and treatment. J Clin Endocrinol Metab. 2011;96(12):3649-3660. doi:10.1210/jc.2011-2031.
- Hansen TM, Batra S, Lim M, et al. Invasive adenoma and pituitary carcinoma: a SEER database analysis. Neurosurg Rev. 2014;37(2):279-285; discussion 285-286. doi:10.1007/s10143-014-0525-y.
- Ragel BT, Couldwell WT. Pituitary carcinoma: a review of the literature. Neurosurg Focus. 2004;16(4):E7.
- Pernicone PJ, Scheithauer BW, Sebo TJ, et al. Pituitary carcinoma: a clinicopathologic study of 15 cases. Cancer. 1997;79(4):804-812.
- Trouillas J, Roy P, Sturm N, et al. A new prognostic clinicopathological classification of pituitary adenomas: a multicentric case-control study of 410 patients with 8 years post-operative follow-up. Acta Neuropathol (Berl). 2013;126(1):123-135. doi:10.1007/s00401-013-1084-y.
- Zhao X, Xiao J, Xing B, et al. Comparison of (68)Ga DOTATATE to 18F-FDG uptake is useful in the differentiation of residual or recurrent pituitary adenoma from the remaining pituitary tissue after transsphenoidal adenomectomy. Clin Nucl Med. 2014;39(7):605-608. doi:10.1097/RLU.0000000000000457.
- Thapar K, Kovacs K, Scheithauer BW, et al. Proliferative activity and invasiveness among pituitary adenomas and carcinomas: an analysis using the MIB-1 antibody. Neurosurgery. 1996;38(1):99-106; discussion 106-107.
- Thapar K, Scheithauer BW, Kovacs K, et al. p53 expression in pituitary adenomas and carcinomas: correlation with invasiveness and tumor growth fractions. Neurosurgery. 1996;38(4):765-770; discussion 770-771.
- Suh JH, Chao ST, Weil RJ. Pituitary Tumors. Clinical Radiation Oncology. Vol 3rd ed. (Gunderson L, Tepper J, eds.). Churchill Livingstone; 2012.
- Müller K, Henke G, Compter I, et al. External validation of a prognostic model estimating the survival of patients with recurrent high-grade gliomas after reirradiation. Pract Radiat Oncol. 2015;5(3):e143-150. doi:10.1016/j.prro.2014.10.001.
- Bush ZM, Longtine JA, Cunningham T, et al. Temozolomide treatment for aggressive pituitary tumors: correlation of clinical outcome with O(6)-methylguanine methyltransferase (MGMT) promoter methylation and expression. J Clin Endocrinol Metab. 2010;95(11):E280-290. doi:10.1210/jc.2010-0441.
- Hegi ME, Diserens A-C, Gorlia T, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005;352(10):997-1003. doi:10.1056/NEJMoa043331.