Predicting close local failure after liver resection for hepatocellular carcinoma
Images
Hepatocellular carcinoma (HCC) is the 5th leading cause of cancer worldwide,1 and the 3rd most common cause of cancer-related death.2 In the United States, where risk factors of viral hepatitis and alcoholic cirrhosis are rising, the incidence of new HCC is also expected to increase.3 The age-adjusted incidence of HCC tripled from 1975-2005,4 and although overall survival improved, the 5-year survival is 40% to 50%.5 Partial hepatic resection remains the most common surgical management of HCC. However, even though advances in operative technique and postoperative care have reduced postoperative mortality in immediate hospital deaths to nearly zero,6 the prognosis remains grim. After surgery, many studies show the 3-year intrahepatic recurrence to be 60% to 70%.7-10 A proportion of these recurrences is in close proximity to the original surgical margin.8-10
Retrospective reviews have consistently identified numerous patient, tumor and treatment-related risk factors that increase the rates of recurrence, including microvascular invasion, satellite nodules and tumor size.11-15 Other factors, such as α-fetoprotein (AFP) level, cirrhosis, resection type and resection margin, have demonstrated a less consistent correlation with recurrence.
Historically, the use of radiotherapy for HCC has been restricted due to poor radiation tolerance of the liver.16,17 However, with advances in three-dimensional radiation planning, image-guided radiotherapy (IGRT), and high-dose stereotactic body radiation (SBR), radiotherapy has demonstrated its effectiveness as an adjuvant treatment as well as its utility in preventing intrahepatic HCC recurrence.18-25
A systematic radiological analysis of intrahepatic recurrence patterns was done at our institution to better understand where a failure will subsequently develop relative to the original surgical bed. In particular, we investigated factors associated with recurrences found within 2 cm of the original surgical site, a region that could be easily treated with local adjuvant or even intra-operative radiation therapy (IORT). This information can help in future selection of patients likely to benefit from additional radiotherapy.
Materials and methods
Internal Review Board approval from the Cleveland Clinic, Cleveland, Ohio, was obtained to perform a retrospective chart and radiographic review of all patients who underwent non-transplant surgical resection at our institution for primary HCC between September 1996 and May 2012. HCC diagnosis was either by characteristic radiological findings on a triphasic CT scan or through biopsy, according to the updated American Association for the Study of Liver Diseases (AASLD) guidelines.26 A total of 169 patients had hepatic resection; however, 22 were excluded due to insufficient available data regarding treatment and follow-up, or because they presented with metastatic disease. All patients were closely followed with computed tomography (CT) or magnetic resonance imaging (MRI) for recurrence.
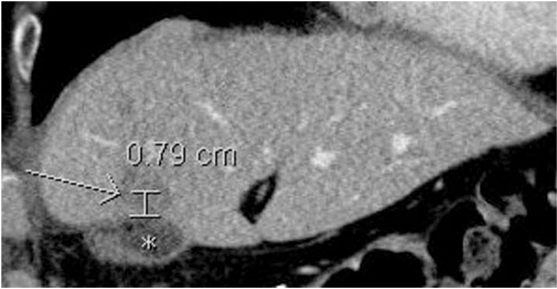
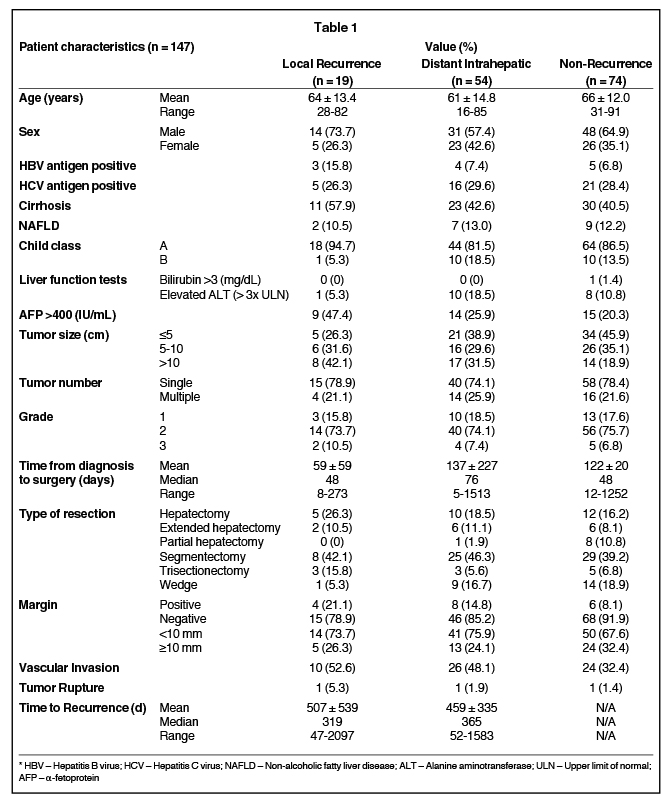
Patient demographic data were recorded, as well as details of pre-existing hepatic disease. Information regarding pre- and postoperative liver function and radiographic detail, method of diagnosis, tumor stage and grade, adjuvant treatment, surgical method and resulting pathology, tumor recurrence, further treatment, and date and cause of death were also gathered (Table 1). Tumors were graded based on the WHO 2010 criteria as outlined in the WHO Classification of Tumors of the Digestive system.27 Recurrence was defined as new radiographic, or biopsy-proven, evidence of HCC not visualized prior to previous resection. Distance to recurrence from the site of the excised primary lesion was defined as the shortest three-dimensional distance from the closest resection margin to the center of the new lesion(s) (Figure 1). The measurement tool and in some instances, the three-dimensional reconstruction software, were used on the hospital Impax image archiving system (AGFA, Mortsel, Belgium) to determine these distances.
Patients were divided into 1 of 3 groups based on the specific location of intrahepatic recurrence: local recurrence (defined as < 2 cm from surgical margin), non-local intrahepatic recurrence (>2 cm from surgical margin) and concurrent local and intrahepatic recurrence. Risk factors associated with each of the above recurrence patterns were characterized and quantified. A 2-cm radius around a given tumor bed was chosen to model the dose distribution characteristics of IORT.
Assessment of tumor resectability was based on AASLD guidelines.28,29 Types of resection included all of the following: right and left hepatectomy, extended or partial right and left hepatectomy, right and left trisectionectomy, segmentectomy, and sub-segment wedge resection of the tumor. Extended left hepatectomy included, in addition to Couinaud segments 2-4, an adjacent narrow portion of segments 5 and 8, and extended right hepatectomy included, in addition to segments 5-8, an adjacent narrow portion of segment 4. Partial right and left hepatectomy was defined as a left or right hepatectomy that did not extend to include the entirety of the segment(s) adjacent to the contralateral hemiliver. Resection types were described according to the Liver Resection Guidelines as reported by the International Hepato-Pancreato-Biliary Association.30 Segmentectomy was defined as resection of an entire Couinaud segment together with its portal vessels, while wedge resection implied removal of the tumor with margin without regard to segmental, sectional or lobar anatomy.
Analysis of patient demographic factors, tumor factors and treatment factors was undertaken. Time from diagnosis to surgery, surgery to recurrence, overall follow-up and overall survival was assessed. Initial diagnosis and recurrence were determined as defined above, and date of death was verified using an online social-security death index. The data was analyzed using logistical regression modeling and Kaplan-Meier survival analysis. A p value of <0.05 was considered statistically significant.
Results
Clinical and surgical data
There were 93 men and 54 women. The mean age of the studied group was 63.7 (range: 16-91) years. Seventy-four patients (50.3%) had histologically proven chronic liver disease: 64 (43.5%) with cirrhosis, 12 (8.0%) with hepatitis B virus (HBV), 42 (29.0%) with hepatitis C virus (HCV), and 18 (12.0%) with non-alcoholic fatty liver disease. The mean AFP level prior to surgery was 3,548.2 (median: 17.8, range: 0.9-88000) ng/mL. A total of 113 (76.9%) patients presented with a solitary lesion on radiological scan. The mean maximum diameter of lesions was 7.3 (range: 0.7-20) cm, and 110 (74.8%) patients had grade II tumors, while 26 (17.7%) and 11 (7.5%) had grades I and III respectively. Upon resection, 103 (72.0%) had a margin >10 mm, and the average margin taken was 6.6 (0-50) mm. Eighteen (12.6%) patients had positive margins, only 3 (0.02%) patients had tumor rupture upon resection, and 62 (42.2%) patients had histologic evidence of vascular invasion. The types of resection performed included right and left hepatectomy, including extended or partial (50 patients, 34.0%); segmentectomy (62 patients, 42.2%); right and left trisectionectomy (11 patients, 7.5%); and local wedge resection (24 patients, 16.3%).
Tumor recurrence
Of the 147 patients, 73 (49.7%) had clinical/radiological evidence of tumor recurrence. Median time to recurrence was 11.9 (range: 1.6-69.9) months. In 47 (64.4%) of these, the recurrence was confined to the hepatic remnant. Extrahepatic recurrence, in addition to intrahepatic failure, was found in 21 (28.8%) patients, and 5 (6.9 %) patients had exclusively extrahepatic tumors: lung (1 patient), omentum and abdominal wall (1 patient), ovary (1 patient), bi-lateral adrenals (1 patient), femoral head (1 patient). Nineteen (27.9%) of the patients who had intrahepatic recurrence failed within 2 cm of the primary surgical margin.
Local vs. distant intrahepatic recurrence
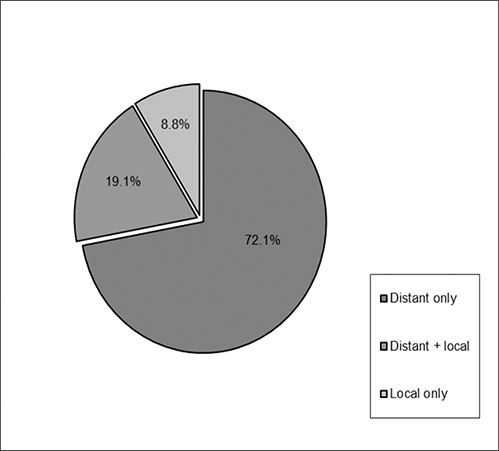
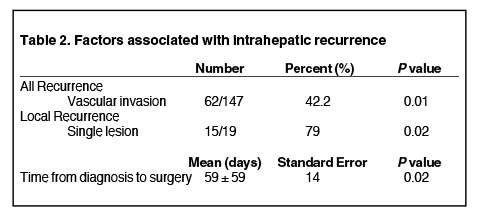
Among patients who had intrahepatic recurrence, 19 (27.9%) were local failures and 49 (72.1%) had recurrences elsewhere in the liver. Mean tumor size for local recurrence was 8.9 (range: 2.6-14.5) cm and 7.5 (range: 1.5-20) cm for distant intrahepatic recurrence. Of those with local recurrence, 6 (32%) had local failure exclusively, while 13 (68%) had both local and distant intrahepatic recurrence (Figure 2). Tumor rupture correlated with recurrence, but was not statistically significant (p = 0.09). While 48% of margin negative resections had recurrence (n = 60/125) compared with 67% with margin positive resections (n = 12/18), the difference was not statistically significant for recurrence (p = 0.16). Vascular invasion was significant for overall recurrence (p = 0.01), but did not specifically correlate with local or distant recurrence. Median time from diagnosis to surgery did correlate with local recurrence (p = 0.02), with a shorter delay associated with increased likelihood of local failure. The presence of a single lesion prior to initial resection also correlated with recurrence (p = 0.02) (Table 2). Tumor grade, tumor size, segmental location, and the concomitant existence of cirrhosis were not significant correlates.
Discussion
The purpose of this study was to review all patients who had partial hepatic resection for primary HCC between September 1996 and May 2012 to determine the patterns and risk factors for local recurrence. We found that, of the 47.9% of resected patients who had intrahepatic recurrence, 27.9% were within 2 cm of the original surgical margin. Multiple studies have distinguished between intra- and extrahepatic recurrence, and some report the number of recurrences close to the primary surgical margin;7 however, to our knowledge no studies have identified risk factors specifically for such close local recurrence.
Previous studies differ regarding the exact constellation of risk factors for HCC recurrence. Previous reports have included vascular invasion, cirrhosis and perioperative blood transfusion,12 tumor size, encapsulation and preoperative AFP,7 to merely preoperative AFP and close resection margin.9 In addition, Huang et al. added pathological grade and tumor thrombus,31 while still others propose 3 or more lesions as predictive of recurrence.32 Aside from vascular invasion, tumor size and tumor number,5 there does not seem to be a clear consensus in the literature regarding what clinical, surgical or pathological factors can be used to predict post-resection HCC recurrence. In our limited patient population, we corroborated vascular invasion as a risk factor for any recurrence. No other risk factors were identified.
It has been suggested that anatomical resection of HCC that includes an entire Couinaud segment with its portal vessels results in a decreased rate of recurrence compared to non-anatomical, or wedge, resection. By removing an entire hepatic segment, there is a chance of including in the resection any local micrometastases or disease extension. Some authors report improved 5-year and disease-free survival using segmentectomy, while others have failed to demonstrate a survival benefit over wedge resection. A large 2012 meta-regression analysis found significant advantages with segmentectomy in 5-year and disease-free survival, but noted that in the retrospective, observational studies analyzed, patients who underwent non-anatomical resection were also more likely to have poorer liver function and reserve.33 Cucchetti et al. recently reported fewer early recurrences and a survival advantage with anatomical resection, though limited to cases with poorly differentiated tumors and microvascular invasion, 2 known risk factors for tumor recurrence.34 In our study, there was little difference in outcomes between patients who had anatomical and non-anatomical resections (Table 1). This may support existing data that suggest there is little recurrence and survival advantage to segmentectomy. Since this study, as well as the vast majority of the relevant literature is retrospective in nature, one major potentially confounding factor is the variability in the practical distinction between segmentectomy and wedge resection. Non-anatomical resections can differ from one case to another, as well as between surgeons; therefore, prospective data would better elucidate whether segmentectomy is truly superior to wedge resection, and in which specific clinical scenarios.
The apparent inability to find a consistent set of risk factors may relate to the numerous potential mechanisms by which HCC can recur and spread. At the time of initial diagnosis, there may already have been multifocal hepatic disease, and in patients with cirrhosis of the liver, the tumor may have developed through a multicentric origin.35 Over time, there could be intrahepatic spread via the portal venous system, and during surgery, there may not have been an adequate margin taken.9 To try and account for multifocal disease on presentation, intraoperative ultrasound was performed on all patients in this series. In our patients with distant intrahepatic recurrences, 10 were detected more than 2 years post resection. This long interval and the significant distance from the primary surgical site, especially in cases with cirrhosis, suggests that these recurrences may have been a new primary lesion. Although a trend may have suggested this relationship, we did not find a statistically significant correlation between the presence of cirrhosis and distant intrahepatic or multicentric tumor recurrence.
In our series, patients who had a local recurrence were statistically more likely to have had a shorter time interval from diagnosis to surgery and only 1 radiographically apparent lesion at the time of surgery. In patients whose tumor recurred, these two factors conferred a 79% risk of the recurrence emerging within 2 cm of the original tumor. While these 2 factors may decrease the likelihood of recurrence overall, when patients with these characteristics recurred, it was often a close local failure. Vascular invasion correlated significantly with overall recurrence, although not with local or distant recurrence in particular. The lack of significance within this division may be reasonably explained by the small sample size of each group and, therefore, decrease in statistical power. Similarly, tumor rupture correlated with recurrence, as all 3 patients with tumor rupture had both local and distant intrahepatic recurrence, although due to the small sample size, it was not statistically significant. Patients who had local recurrence had a shorter overall survival from the time of resection, as well as from the time of recurrence. However, the wide survival time range and the relatively small sample size limited the ability to find statistical significance in overall survival time.
Nevertheless, these findings suggest that there may be a subset of patients who have a predictable recurrence pattern and associated worse prognosis and, therefore, may benefit from enhanced local control. Historically, a number of pre and post-operative adjuvant therapies have been attempted to minimize local recurrence with mixed results. In 2 prospective randomized trials, neoadjuvant transarterial chemoembolization (TACE) showed no statistically significant improvement over surgery alone, and in some cases, even worse overall survival.36,37 Postoperative regional chemotherapy has also been studied to reduce local recurrence, and while 2 randomized controlled trials yielded increased overall survival,38,39 others found little benefit to regional chemotherapy when combined with systemic chemotherapy.40,41 Studies of the use of oral chemotherapy alone have also shown minimal promise, and use is limited by systemic toxicity.42,43
A few small studies have reported the rate of intrahepatic recurrence after radiotherapy alone, or combined with TACE. In a series of 44 patients assessing survival in patients with unresectable HCC treated with radiotherapy, Liu et al. reported a 43.2% rate of intrahepatic recurrence.20 In another small series of 25 patients, Cheng et al. found that at 2 years, 56% of patients had either local or regional intrahepatic tumor recurrence after radiotherapy.25 Park et al. reported a 47.5% intrahepatic recurrence rate in a survival analysis of 59 patients.24 In the latter 2 of these studies, many of the patients had also been treated with TACE. No studies, however, specifically detail a pattern of close local intrahepatic failure, at best separating intrahepatic recurrence into general categories of local vs. regional recurrence. As such, there is currently no data on the potential utility of radiation to prevent such close local failure.
In addition to neoadjuvant TACE, chemotherapy and external beam radiotherapy (EBRT), radiofrequency ablation (RFA) combined with resection has shown promise as part of multimodality treatment of HCC. In a recent retrospective study, Prassas et al. found moderate improvement in overall and disease-free survival with an RFA-assisted liver resection method. Of note, they reported no close local recurrences with this technique.44 Liu compared the efficacy of RFA in HCC vs. metastatic disease, concluding that RFA was beneficial for small, localized tumors.45 Yi et al. prospectively compared RFA plus TACE with RFA alone and reported a significant survival benefit with combined treatment.46 While not yet applied to HCC, IORT to reduce local recurrence has shown to be a feasible, beneficial and low-risk treatment option for other cancer types. IORT has resulted in improved survival outcomes and treatment adherence in breast, colon, pancreatic, head and neck, sarcoma and gynecological cancers.47-55 This technique allows the delivery of an increased and concentrated dose of radiation in a precise, focused manner to specifically target the tumor bed and closely associated at-risk regions. Still other potential, though yet uncorroborated, methods to decrease local recurrence of HCC include segmental resection, earlier liver transplant, and expansion of planned surgical margins by 2 cm.
Conclusion
Based on the analysis of our experience with local and distant-intrahepatic recurrence of HCC after resection, we believe a subgroup of patients, namely those with only 1 radiographically apparent lesion at the time of diagnosis and who underwent resection soon after diagnosis, are more likely to have tumor recurrence within 2 cm of the original surgical bed. Identifying the ideal treatment method by which to substantially reduce the risk of local failure for HCC patients with high-risk features is ongoing. Perhaps, using time from diagnosis to surgery and number of lesions at diagnosis as exclusion criteria, the application of IORT for this subgroup at greater risk for local recurrence can be studied in the future.
References
- Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69-90.
- Ferlay J, Autier P, Boniol M, et al. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007;18(3):581-592.
- Sherman M. Hepatocellular carcinoma: Epidemiology, risk factors, and screening. Semin Liver Dis. 2005;25(2):143-154.
- Altekruse SF, McGlynn KA, Reichman ME. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol. 2009;27(9):1485-1491.
- Poon RT, Fan S, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232(1):10-24.
- Fan ST, Lo CM, Liu CL, et al. Hepatectomy for hepatocellular carcinoma: Toward zero hospital deaths. Ann Surg. 1999;229(3):322-330.
- Llovet JM, Schwartz M, Mazzaferro V. Resection and liver transplantation for hepatocellular carcinoma. Semin Liver Dis. 2005;25:181-200.
- Wu CC, Cheng SB, Ho WM, et al. Liver resection for hepatocellular carcinoma in patients with cirrhosis. Br J Surg. 2005;92(3):348-355.
- Chen MF, Hwang TL, Jeng LB, et al. Postoperative recurrence of hepatocellular carcinoma. Two hundred five consecutive patients who underwent hepatic resection in 15 years. Arch Surg. 1994;129(7):738-742.
- Poon RT, Fan ST, Lo CM, et al. Long-term survival and pattern of recurrence after resection of small hepatocellular carcinoma in patients with preserved liver function: Implications for a strategy of salvage transplantation. Ann Surg. 2002;235(3):373-382.
- Kozyreva ON, Chi D, Clark JW, et al. A multicenter retrospective study on clinical characteristics, treatment patterns, and outcome in elderly patients with hepatocellular carcinoma. Oncologist. 2011;16(3):310-318.
- Chen Z, Ni JL, Liu LY. Analysis of prognostic factors in patients with huge primary liver cancer after surgical resection. Zhonghua Zhong Liu Za Zhi. 2011;33(9):710-713.
- Ji SP, Li Q, Dong H. Therapy and prognostic features of primary clear cell carcinoma of the liver. World J Gastroenterol. 2010;16(6):764-769.
- Ng KK, Poon RT, Lo CM, et al. Analysis of recurrence pattern and its influence on survival outcome after radiofrequency ablation of hepatocellular carcinoma. J Gastrointest Surg. 2008;12(1):183-191.
- Yan K, Chen MH, Yang W, et al. Radiofrequency ablation of hepatocellular carcinoma: Long-term outcome and prognostic factors. Eur J Radiol. 2008;67(2):336-347.
- Ingold JA, Reed GB, Kaplan HS, et al. Radiation hepatitis. Am J Roentgenol Radium Ther Nucl Med. 1965;93:200-208.
- Lawrence TS, Robertson JM, Anscher MS, et al. Hepatic toxicity resulting from cancer treatment. Int J Radiat Oncol Biol Phys. 1995;31(5):1237-1248.
- Thomas E, Chapet O, Kessler ML, et al. Benefit of using biologic parameters (EUD and NTCP) in IMRT optimization for treatment of intrahepatic tumors. Int J Radiat Oncol Biol Phys. 2005;62(2):571-578.
- Zeng ZC, Tang ZY, Yang BH, et al. Radiation therapy for the locoregional lymph node metastases from Hepatocellular carcinoma, Phase I clinical trial. Hepatogastroenterology. 2004;51(55):201-207.
- Liu MT, Li SH, Chu TC, et al. Three-dimensional conformal radiation therapy for unresectable hepatocellular carcinoma patients who had failed with or were unsuited for transcatheter arterial chemoembolization. Jpn J Clin Oncol. 2004;34(9):532-539.
- Herfarth KK, Debus J, Wannenmacher M. Stereotactic radiation therapy of liver metastases: Update of the initial Phase- I/II trial. Front Radiat Ther Oncol. 2004;38:100-105.
- Goitein M. Organ and tumor motion: An overview. Semin Radiat Oncol. 2004;14(1):2-9.
- Seong J, Park HC, Han KH, et al. Clinical results and prognostic factors in radiotherapy for unresectable Hepatocellular carcinoma: A retrospective study of 158 patients. Int J Radiat Oncol Biol Phys. 2003;55(2):329-336.
- Park W, Lim DH, Paik SW, et al. Local radiotherapy for patients with unresectable Hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2005;61(4):1143-1150.
- Cheng JC, Chuang VP, Cheng SH, et al. Local radiotherapy with or without transcatheter arterial chemoembolization for patients with unresectable hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2000;47(2):435-442.
- Bruix J, Sherman M. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: An update. Hepatol. 2011;53(3):1020-1022.
- Bosman FT, Carneiro F, Hruban RH, et al. WHO classification of tumours of the digestive system (4th ed). France, Lyon: IARC Press.
- Bruix J, Sherman M. Practice Guidelines Committee, American Association for the Study of Liver Disease. Management of hepatocellular carcinoma. Hepatol. 2005;42(5):1208-1236.
- de Lope CR, Tremosini S, Forner A, et al. Management of HCC. J Hepatol. 2012;56 Suppl 1:75-87.
- Liver Resection Guidelines. http://www.ihpba.org/92_Liver-resection-Guidlines.html. Accessed July 1, 2013.
- Huang X, Wei W, Ya N, et al. A mathematical model to predict short-term recurrence and metastasis of primary hepatocellular carcinoma larger than 10cm in diameter. Hepatogastroenterology. 2012;60(122). [Epub ahead of print]
- Chan KM, Chou HS, Wu TJ, et al. Characterization of hepatocellular carcinoma recurrence after liver transplantation: Perioperative prognostic factors, patterns, and outcome. Asian J Surg. 2011;34(3):128-134.
- Cucchetti A, Cescon M, Ercolani G, et al. A comprehensive meta-regression analysis on outcome of anatomic resection versus non-anatomic resection for hepatocellular carcinoma. Ann Surg Oncol. 2012;19(12):3697-3705.
- Cucchetti A, Qiao GL, Cescon M, et al. Anatomic versus nonanatomic resection in cirrhotic patients with early hepatocellular carcinoma. Surg. 2014;155(3):512-521.
- Belli L, Romani F, Belli LS, et al. Reappraisal of surgical treatment of small hepatocellular carcinomas in cirrhosis: Clinicopathological study of resection or transplantation. Dig Dis Sci. 1989;34(10):1571-1575.
- Wu KT, Wang CC, Lu LG, et al. Hepatocellular carcinoma: Clinical study of long-term survival and choice of treatment modalities. World J Gastroenterol. 2013;19(23):3649-3657.
- Zhou WP, Lai EC, Li AJ, et al. A prospective, randomized, controlled trial of preoperative transarterial chemoembolization for resectable large hepatocellular carcinoma. Ann Surg. 2009;249(2):195-202.
- Nagano H, Kobayashi S, Marubashi S, et al. Combined IFN-α and 5-FU treatment as a postoperative adjuvant following surgery for hepatocellular carcinoma with portal venous tumor thrombus. Exp Ther Med. 2013;5(1):3-10.
- Tanaka S, Shimada M, Shirabe K, et al. A novel intrahepatic arterial chemotherapy after radical resection for advanced hepatocellular carcinoma. Hepatogastroenterology. 2005;52(63):862-865.
- Ikai I, Arii S, Ichida T, et al. Report of the 16th follow-up survey of primary liver cancer. Hepatol Res. 2005;32(3):163-172.
- Ono T, Yamanoi A, Nazmy El Assal O, et al. Adjuvant chemotherapy after resection of hepatocellular carcinoma causes deterioration of long-term prognosis in cirrhotic patients: Meta-analysis of three randomized controlled trials. Cancer. 2001;91(12):2378-2385.
- Hasegawa K, Takayama T, Ijichi M, et al. Uracil-tegafur as an adjuvant for hepatocellular carcinoma: A randomized trial. Hepatol. 2006;44(4):891-895.
- Yamamoto M, Arii S, Sugahara K, et al. Adjuvant oral chemotherapy to prevent recurrence after curative resection for hepatocellular carcinoma. Br J Surg. 1996;83(3):336-340.
- Prassas E, Petrou A, Kontos M, et al. Radiofrequency ablation assisted resection for hepatocellular carcinoma: Morbidity, mortality and long term survival. J BUON. 2014;19(1):256-262.
- Liu J, Qian LX. Therapeutic efficacy comparison of radiofrequency ablation in hepatocellular carcinoma and metastatic liver cancer. Exp Ther Med. 2014;7(4):897-900.
- Yi Y, Zhang Y, Wei Q, et al. Radiofrequency ablation or microwave ablation combined with transcatheter arterial chemoembolization in treatment of hepatocellular carcinoma by comparing with radiofrequency ablation alone. Chin J Cancer Res. 2014;26(1):112-118.
- Fisher ER, Anderson S, Redmond C, et al. Ipsilateral breast tumor recurrence and survival following lumpectomy and irradiation: Pathological findings from NSABP protocol B-06. Semin Surg Oncol. 1992;8(3):161-166.
- Vaidya JS, Joseph DJ, Tobias JS, et al. Targeted intraoperative radiotherapy versus whole breast radiotherapy for breast cancer (TARGIT-A trial): An international, prospective, randomised, non-inferiority phase 3 trial. Lancet. 2010;376(9735):91-102.
- Alektiar KM, Zelefsky MJ, Paty PB, et al. High dose rate intraoperative brachytherapy for recurrent colorectal cancer. Int J Radiat Oncol Biol Phys. 2000;48(1):219-226.
- Kusters M, Holman FA, Martijn H, et al. Patterns of local recurrence in locally advanced rectal cancer after intraoperative radiotherapy containing multimodality treatment. Radiother Oncol. 2009;92(2):221-225.
- Nuyttens JJ, Kolkman-Deurloo IK, Vermaas M, et al. High-dose-rate intraoperative radiotherapy for close or positive margins in patients with locally advanced or recurrent rectal cancer. Int J Radiat Oncol Biol Phys. 2004;58(1):106-112.
- Bachireddy P, Tseng D, Horoschak M, et al. Orthovoltage intraoperative radiation therapy for pancreatic adenocarcinoma. Radiat Oncol. 2010;5:105-110.
- Tran PT, Hara W, Su Z, et al. Intraoperative radiation therapy for locally advanced and recurrent soft-tissue sarcomas in adults. Int J Radiat Oncol Biol Phys. 2008;72(4):1146-1153.
- Zeidan YH, Shiue K, Weed D, et al. Intraoperative radiotherapy for parotid cancer: A single-institution experience. Int J Radiat Oncol Biol Phys. 2012;82(5):1831-1836.
- Tran PT, Su Z, Hara W, et al. Long-term survivors using intraoperative radiotherapy for recurrent gynecologic malignancies. Int J Radiat Oncol Biol Phys. 2007;69(2):504-511.
Editor’s note: Part of this work was previously published as an abstract with the following reference: Fredman E, Kumar A, El-Gazzaz G, et al. Local failure after liver resection for hepatocellular carcinoma. J Clin Oncol. 2013 May; 31(suppl; abstr):e15015.