Postradiation cardiac sarcoma and other secondary cancers more than 25 years after chemotherapy and mantle-field radiation for H
Images

CASE SUMMARY
In July 2013, a 48-year-old Caucasian female presented to the emergency department with rapid-onset fatigue, shortness of breath and palpitations. Her medical history was remarkable for early stage Hodgkin’s lymphoma (HL) diagnosed at age 19 (1984) for which she underwent doxorubicin, bleomycin, vinblastine and dacarbazine chemotherapy, then mantle-field radiation to 40 Gy (port films unavailable) with a complete response. During the late-1990s, she was diagnosed with hypothyroidism and a prolactinoma, both managed medically. She had regular surveillance of the breasts and thyroid. In early 2012, she was diagnosed with a Stage IA T1aN0M0 invasive tubular carcinoma in the upper outer quadrant of the right breast. After consideration of risks from re-irradiation as part of breast conservation therapy, she opted for bilateral mastectomy with flap reconstruction. In July, 2012, surveillance ultrasound of the thyroid revealed the growth of a nodule. Fine-needle aspirate was suspicious and she underwent total thyroidectomy, revealing a T1a follicular papillary microcarcinoma.
IMAGING FINDINGS
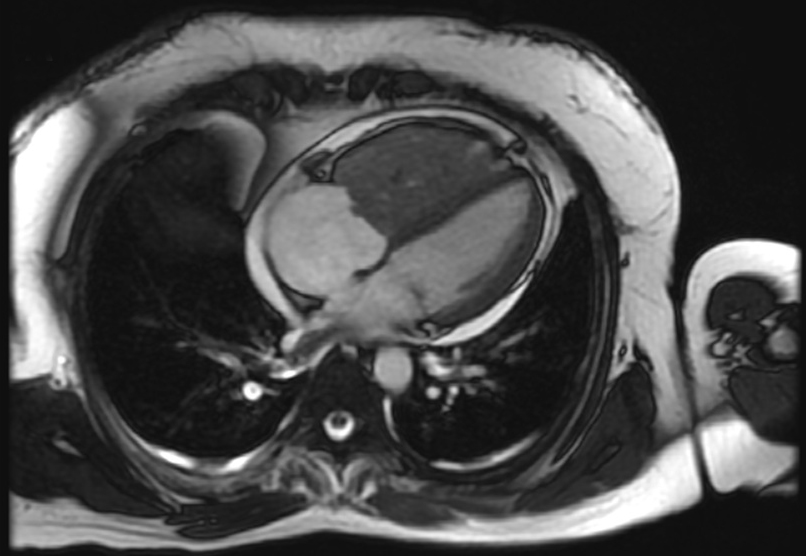
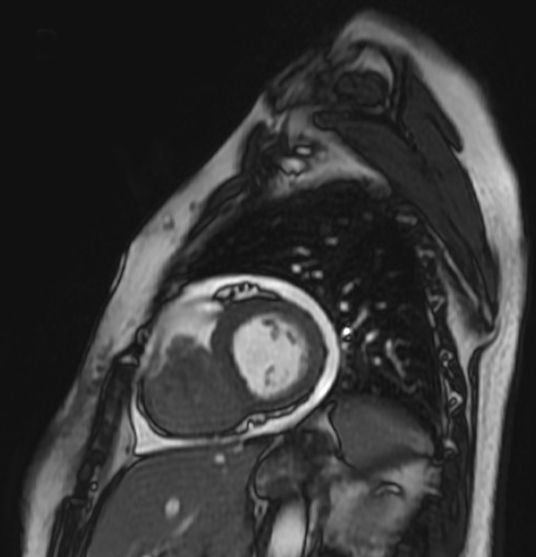
As part of her workup, chest computed tomography and angiography (CTA) was performed, and demonstrated a 6.7 cm mass filling the right ventricle (RV). Cardiac magnetic resonance imaging confirmed this mass, which seemed adherent to endocardial surface and tricuspid leaflets (Figure 1). Echocardiography showed severe RV inflow obstruction and dysfunction.
DIFFERENTIAL DIAGNOSIS
Primary sarcoma, metastasis, myxoma, papillary fibroelastoma, lipoma, paraganglioma
DISCUSSION
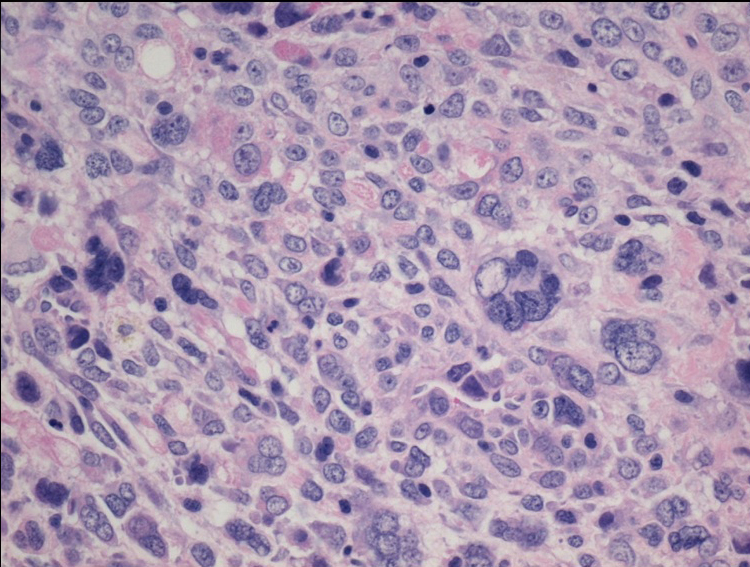
The patient underwent radical resection of the tumor, tricuspid bioprosthesis replacement with pacemaker implantation, and was placed on the cardiac transplant list. Pathology revealed undifferentiated pleomorphic sarcoma (Figure 2). Postoperative imaging showed residual tumor along the RV wall. She was started on liposomal doxorubicin but developed an anaphylactic reaction.
In August 2013, the patient developed profound shortness of breath with chest pain and fatigue. Echocardiography confirmed interval mass growth with near-complete RV obstruction. Extensive multidisciplinary discussion yielded recommendations for palliative radiation for a bridge to heart transplant. Combined modality therapy was not advocated due to her fragile clinical status. After a single 300 cGy fraction of radiation, however, she developed progressive multi-organ failure. She decided to pursue comfort care measures, and radiation was discontinued. She was transferred to hospice care and died shortly after.
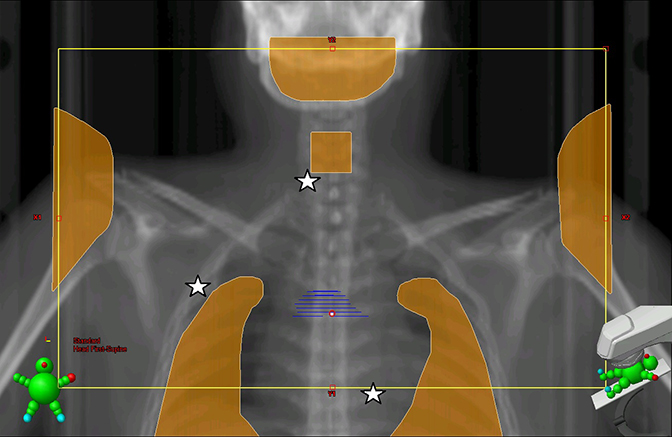
In the early decades of combined modality therapy for HL, large radiation fields (eg., mantle field, Figure 3) were routinely used to cover all at-risk nodal stations. The combined effects of large-field radiation and chemotherapy have resulted in considerable long-term risk for secondary cancers (SC), with over 20% risk for developing a solid tumor at 25 years after HL diagnosis.1,2 The risks are highest for survivors of younger age-at-diagnosis and older attained-age.3
The majority (≥ 75%) of SC after radiation for HL develop within or adjacent to previously irradiated fields with increasing risks of development associated with increased dose, especially ≥ 35 Gy.4,5 Over 20% of these tumors are lung cancers, > 10% are breast cancer, with thyroid cancers (2%) and sarcomas (1.3%) occurring more rarely.1
Postradiation sarcomas are defined by a history of radiation exposure, no prior sarcoma, and histologic features distinct from any previously treated primary. These tumors have significantly worse prognosis than sporadic sarcomas regardless of histologic type.>6 Postradiation cardiac sarcomas (PCS) are exceedingly rare and have yet to be reported in HL survivors.
For sporadic cardiac sarcomas (SCS), complete resection is the only curative treatment, with reported median survival of up to 23.5 months after definitive resection,7 but many cases are unresectable due to extensive myocardial involvement. Multimodality therapy (palliative debulking surgery, anthracycline-based chemotherapy, and/or radiation) has been advocated in cases of unresectable and recurrent tumors, although the value of this treatment approach is unknown due to the paucity of data.8 There is limited evidence for orthotopic cardiac transplantation in otherwise unresectable SCS, providing survival of up to 3 years in selected patients.9 Due to the scarcity of PCS cases, management strategies are extrapolated from evidence for SCS.
CONCLUSION
We report a 48-year-old woman with history of HL at age 18, treated with chemotherapy and mantle-field radiation to 40 Gy, who subsequently developed early stage breast cancer, thyroid cancer, and ultimately died of a cardiac sarcoma. The mantle field used to treat this patient to 40 Gy would have covered her thyroid, upper outer quadrants of bilateral breasts, and heart, likely contributing to each of her secondary malignancies. This case reflects the importance of early screening and management of radiation-induced secondary malignancies in long-term HL survivors and is, to our knowledge, the first reported case of cardiac sarcoma following mantle-field radiation.
REFERENCES
- Dores GM, Metayer C, Curtis RE, et al. Second malignant neoplasms among long-term survivors of Hodgkin’s disease: a population-based evaluation over 25 years. J Clin Oncol. 2002;20(16):3484-3494.
- Mudie NY, Swerdlow AJ, Higgins CD, et al. Risk of second malignancy after non-Hodgkin’s lymphoma: a British Cohort Study. J Clin Oncol. 2006;24(10):1568-1574.
- Hodgson DC, Gilbert ES, Dores GM, et al. Long-term solid cancer risk among 5-year survivors of Hodgkin’s lymphoma. J Clin Oncol. 2007;25(12):1489-97.
- Constine LS, Tarbell N, Hudson MM et al. Subsequent malignancies in children treated for Hodgkin’s disease: associations with gender and radiation dose. Int J Radiat Oncol Biol Phys. 2008;72(1):24-33.
- Nyandoto P, Muhonen T, and Joensuu H. Second cancer among long-term survivors from Hodgkin’s disease. Int J Radiat Oncol Biol Phys. 1998;42(2):373-378.
- Gladdy RA, Qin LX, Moraco N et al. Do radiation-associated soft tissue sarcomas have the same prognosis as sporadic soft tissue sarcomas? J Clin Oncol. 2010;28(12):2064-2069.
- Bakaeen FG, Jaroszewski DE, Rice DC, et al. Outcomes after surgical resection of cardiac sarcoma in the multimodality treatment era. J Thorac Cardiovasc Surg. 2009; 137(6):1454-1460.
- Butany J, Nair V, Naseemuddin A, et al. Cardiac tumours: diagnosis and management. Lancet Oncol. 2005;6(4):219-228.