Optimizing treatment positioning to achieve better heart sparing in a left-sided, whole-breast irradiation
Images

Case Winner June 2016
CASE SUMMARY
A 33-year-old, premenopausal, BRCA-negative woman with Adriamycin-induced cardiomyopathy and left ventricular dysfunction presented to our department in 2015 for adjuvant treatment of ductal carcinoma in situ (DCIS) of the left breast. Her medical history was significant for Wilm’s tumor diagnosed at age 10, treated with surgery followed by Adriamycin-based chemotherapy, which resulted in dilated cardiomyopathy. She remained on several medications for cardiomyopathy over many years, and has been followed closely by cardiologists. Her left ventricular ejection fraction, prior to initiating radiation, was estimated to be 37%.
Three months prior to presentation, the patient sustained an episode of congestive heart failure exacerbation, and was found to be 13 weeks pregnant. Cardiology assessed that the patient was at high risk for decompensating with pregnancy, and was advised to undergo an elective termination, which was subsequently performed without complication. She nevertheless continued to exhibit moderate heart failure symptoms, and was started on high-dose diuretics with ultimate symptom resolution.
With regard to the DCIS diagnosis, she initially presented with bloody left nipple discharge. Breast imaging studies were obtained, demonstrating suspicious microcalcifications in the retroareaolar region of the left breast. Core biopsy of the microcalcifications revealed intraductal papilloma. Lumpectomy was performed, with pathology consistent with a diagnosis of DCIS. Adjuvant radiation with 3D-conformal radiation therapy was recommended, following extensive discussion with her breast surgeon and cardiologist. Additionally, given the positivity of ER and PR hormone receptors, she was recommended a 5-year course of Tamoxifen upon completion of left breast irradiation.
IMAGING AND PATHOLOGY FINDINGS
Initial left breast diagnostic mammogram showed grouped microcalcifications in the retroareolar region. Diagnostic ultrasound of the left breast showed a 1-cm cyst cluster that was 4 cm from the nipple, and dilated retroareolar ducts with minimal intraluminal debris. Subsequent bilateral breast MRI demonstrated a suspicious 8-mm mass medial to the nipple of the left breast, and suspicious nonmass enhancement in the retroareolar region of the right breast, as well as an 8-mm enhancing mass in the superior mid portion of the right breast. Ultrasound-guided core biopsies of the subareolar axis of the left breast and of retroareolar axis of the right breast revealed intraductal papillomas. The patient underwent an additional core biopsy of the left breast inferior outer quadrant, which also revealed intraductal papilloma. She then underwent bilateral lumpectomies. Whereas the right breast surgical specimen contained intraductal papilloma, her left breast lumpectomy pathology revealed an 8-mm focus DCIS, EORTC intermediate grade, with negative margins, ER+, PR+.
DIAGNOSIS
The final diagnosis was AJCC stage 0 (Tis N0 M0) ductal carcinoma in situ, intermediate grade, ER+/PR+.
DISCUSSION
This report describes the case of a left-sided breast DCIS in a young woman with dilated cardiomyopathy. Considering the patient’s left-sided breast carcinoma, previous exposure to Adriamycin-based chemotherapy, dilated cardiomyopathy, left ventricular ejection fraction of 37%, and young age, choosing a radiotherapy treatment plan that maximized cardiac sparing was essential. In a recent population-based case-control study, Darby et al1 have demonstrated that exposure of the heart to ionizing radiation during radiotherapy for left-sided breast cancer significantly increases the risk of having a major coronary event, such as myocardial infarction, coronary revascularization, or death from ischemic heart disease.1 This risk is found to increase linearly with the mean heart dose (MHD). For every 1 Gy increase in the MHD, the rate of major coronary events increased by 7.4%. Moreover, because the study by Darby et al included few women who were younger than 40 years and received radiation for left breast cancer, it was cautioned that the risk of women younger than 40 is likely to be even higher than what they reported. Therefore, the aim of designing a treatment plan for this patient was to minimize the heart dose without compromising coverage to the breast tissue. The use of deep inspiration breath hold (DIBH) has been demonstrated as a highly effective technique for reducing cardiac dose.2,3,8 In this technique the patient takes a deep breath, trying to increase the distance between the chest wall and heart, allowing adequate treatment of the breast while minimizing irradiated cardiac volume. In a large series of breast cancer patients receiving whole-breast radiation, it was shown that the MHD was reduced from 5.2 Gy with free breathing to 2.7 Gy with DIBH.3 However, DIBH requires the patient to hold her breath for ≥ 20 seconds, and its dosimetric benefit depends on the adequate expansion of the chest wall and the distance of the heart from it. As the chest wall expands anteriorly with deep inspiration, it pulls more lung into the treatment field.2 DIBH is known to treat more absolute volume of the lung than with free breathing (FB) while sparing the heart.3 Prone breast irradiation, on the other hand, has consistently provided lower lung doses but has shown varied results concerning cardiac dose.4-7 Since this position displaces the heart anteriorly toward the chest wall, it increases the likelihood of heart exposure to radiation.6 In a prospective trial comparing the prone vs. the supine position, while 87% of the patients had lower cardiac exposure in the prone position, 13% of the patients were noted to have a higher heart dose.4 Another comparative study of both these positions reported that patients with a whole-breast clinical target volume (WB CTV) ≥ 1000 cc benefited from the prone position when it came to heart sparing, whereas patients with WB CTV volume < 1000 cc had higher heart doses.5 Larger breast volume enabled the breast to be pulled under gravity anteriorly in relation to the chest wall, allowing the placement of shallower tangents to better spare the heart, proving prone to be better than supine positioning in these patients.
A comparison of the prone position using FB with supine using DIBH was recently performed in a prospective study.7 Noncontrast CT scans for 17 patients were acquired in the supine position with FB, supine position with DIBH and prone position with FB. For insignificant differences in planning target volume (PTV) coverage and homogeneity, the MHD was consistently highest in the prone position at 5.4 Gy (3.5 Gy to 6.2 Gy) and lowest with DIBH in the supine position at 1.6 Gy (1.2 Gy to 2.2 Gy). The ipsilateral lung V20 Gy was consistently lowest in the prone position at 2.3% (1.4% to 3.4%) and highest in the supine position with FB at 7.3% (5.7% to 9.7%). The study concluded that both treatment positions had advantages and disadvantages; prone being the best position to reduce dose to the lungs, and DIBH with supine being the best to reduce dose to the heart. It also recommended that a patient unfit to handle DIBH be treated supine using FB rather than in the prone position.
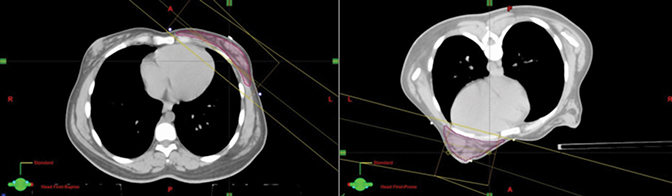
Since sparing the heart from radiation was of the highest priority in this case and since the major advantage of DIBH is to reduce dose to the heart, it first was decided that the patient should be simulated in the supine position for DIBH. The patient was positioned supine with both arms up using the C-Qual Breastboard (CIVCO, Orange City, Iowa). The breast tissue was palpated and outlined with a radio-opaque wire. Two scans were acquired, namely supine using FB and supine with DIBH. Upon comparing the FB and DIBH scans for this patient (Figure 1), the position of the heart with respect to the chest wall did not appear to differ remarkably. At this juncture, the patient’s efforts to breathe deeper—to allow for increased separation of the heart from the chest wall and to take a new scan with DIBH—were also diminishing. Due to these impediments, we decided to scan the patient in the prone position, the advantage being that the patient did not need to hold her breath for prolonged periods, which improved comfort and compliance in receiving radiation therapy. Studies have shown that setup errors are larger in the prone than in the supine position and that elderly and obese patients especially have difficulty positioning themselves on the prone board, leading to challenges in reproducing setup during treatment.9,10 In this case, the patient had a normal BMI (body mass index) of 24.4 at the time of therapy, raising less concern pertaining to reproducibility of setup. For this position, the Prone Breast System (Bionix, Toledo, Ohio) was used. The patient lay prone on the board with both arms raised above the head, which was turned to the contralateral side. The board has an adjustable aperture that allows the breast tissue to be treated to fall freely within the opening. The contralateral breast was held up and away using support cushions and wedges as needed to keep it out of the path of the treatment fields.10 The palpable breast tissue was again outlined with a radio-opaque wire and the patient was scanned with 3-mm slice thickness.
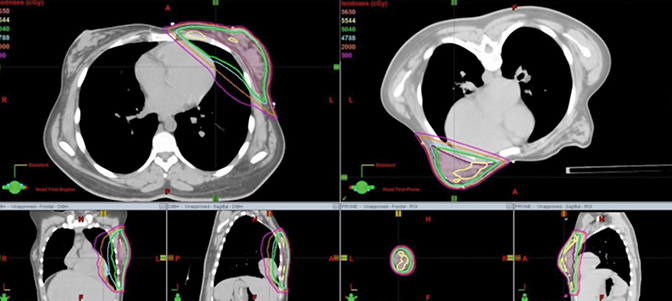
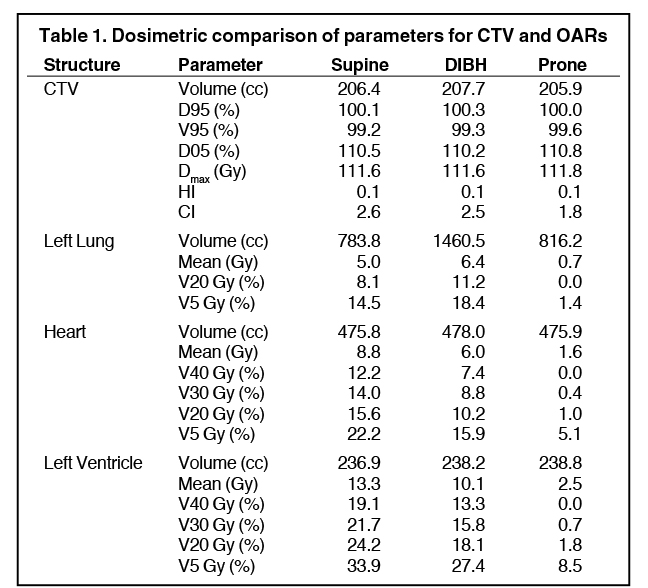
CT data from scans in all 3 positions (supine using FB, supine using DIBH, and prone with FB) were transferred to the Eclipse V2 (Varian Medical Systems, Palo Alto, California) treatment planning system for planning and dose calculations. The CTV and organs at risk (OARs) were delineated as the entire palpable breast tissue that was outlined by the radio-opaque wires plus any additional breast tissue that felt needed to be included as a part of the CTV. The radio-opaque markers were used to help define the superior, inferior, medial and lateral field borders. The posterior border of the CTV was defined by the pectoralis major muscle, and the anterior border was limited to 5 mm from the skin surface. Contouring of the heart and the left ventricle was according to published guidelines.11 Table 1 summarizes volumes of the CTVs and OARs. The volumes of the heart and the left ventricle are equivalent in the prone and the supine position (with FB or with DIBH) as were the volumes of the CTVs. However the ipsilateral lung volume in the supine position with FB in this study was found to be lower by 4% compared to the prone position. Similar results for the CTV and these OARs have been reported previously by Chen et al.12 For treatment planning, standard opposed tangential fields were used to cover the CTV as shown in Figure 2. The planning technique used was field-in-field. Each plan was optimized to cover the breast CTV such that the D95 and V95 were both ≥ 99% while maximizing heart and ipsilateral lung sparing and keeping the plan as homogeneous as possible. The dose prescribed was 50.4 Gy in 1.8 Gy fractions over 5 weeks using 6 MV photons. The normalization point was placed at 1 cm anterior from the lung chest wall interface and in a plane 1.5 cm inferior from the superior border of the tangential field. The dose calculation algorithm was AAA (analytical anisotropic algorithm) with a calculation grid size of 2.5 mm. Definitions for the homogeneity index (HI) and the conformity index (CI) were taken as described and utilized in the literature for these cases.12
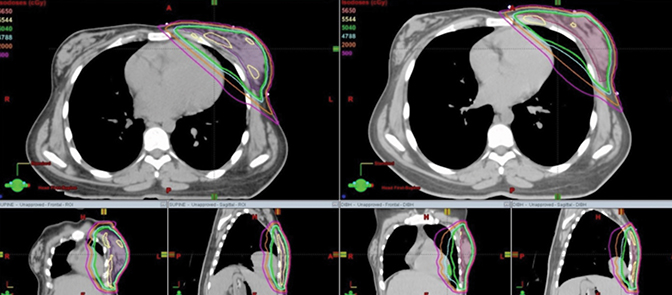
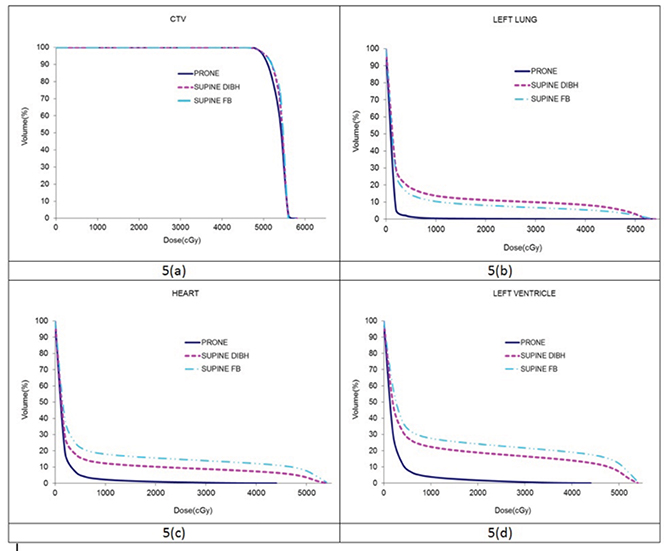
Dosimetric results comparing CTV coverage as well as the heart, left ventricle and the left (ipsilateral) lung have been tabulated in Table 1. Dose distributions comparing the supine DIBH plan vs. prone is shown in Figure 3, while that between supine FB vs. supine DIBH is shown in Figure 4. A comparison of the dose volume histograms (DVHs) in all 3 cases is shown in Figure 5. D95 and V95 of the CTV were comparable between the 3 cases as were the D05 and the maximum doses. All 3 plans were just as homogeneous as indicated by HI; however, the plan in the prone position was the most conformal as previously reported.12 The MHD was 9 Gy in the supine position with FB, and the use of DIBH was able to reduce the MHD to 6 Gy. Planning this patient in the prone position helped reduce the mean heart dose to < 2 Gy and the mean left ventricle dose to < 3 Gy. As found in the Darby study, a 40-year-old woman receiving radiation for breast cancer with an MHD of < 2 Gy and at least one cardiac risk factor has an absolute risk of death of 0.5% from radiation-related ischemic heart disease (IHD) by age 80 years. For an MHD of 6 Gy and 9 Gy, this risk is 1.4% and 2.1%, respectively. The absolute risk of at least 1 radiation-related acute coronary event (ACE) by age 80 years is < 1.1% for an MHD of < 2 Gy, while the risk is 3.3% and 4.9% for MHD of 6 Gy and 9 Gy, respectively. Using the prone position for this patient has helped lower these risks compared to the supine position with or without the use of DIBH. Also, contrary to reports of a dosimetric study by Kirby et al,5 although the patient’s whole-breast volume in this case study was ~200 cc, this patient benefited from the prone position compared to supine with regard to cardiac sparing. The prone position also best spared the ipsilateral lung with a V20 Gy of 0% and a mean dose of < 1 Gy.
Recently, a decision-making flow chart has been proposed for WBI, which recommends treating a left-sided breast cancer patient that is unfit for DIBH with FB in the supine position.7 In the case study presented here, the patient was not a good candidate for DIBH. Moreover with DIBH in the supine position, the MHD was at 9 Gy, making the plan nonviable for treatment. The prone position best spared both the heart as well as the lung. The choice of the treatment technique and optimal beam arrangement to cover the target depends on how the patient is set up at the time of simulation as well as on the patient anatomy for that particular simulated position. This case study is an example of a situation in which other treatment positions may need to be explored in addition to recommendations made by studies.7
The patient successfully completed radiation therapy as planned without any cardiac episodes. The increase in the rate of major coronary events per 1 Gy depends on the length of time after completing radiotherapy.1 Within 0 to 4 years, the rate is 16.3%, and between 5 to 9 years it is 15.5%. The current follow-up time for the patient in our case report is too short for manifestation of cardiac injury; however, by reducing the MHD from 9 Gy to 1.6 Gy, we were able to minimize the risk of a major coronary event in this patient. She is currently maintained on anti-estrogen therapy with Tamoxifen and follows up with her cardiologist regularly. At her 1-month follow-up appointment she reported feeling well overall and denied chest pain, pressure, palpitations, dyspnea on exertion or orthopnea. She also did not experience any shortness of breath, wheezing, coughing or hemoptysis. She denied any breast pain, swelling or palpable masses. She had normal range of motion in the bilateral upper extremities without any edema and denied any weight changes, fatigue or appetite disturbances post treatment, and was able to resume working fulltime. She will continue to receive regular mammograms and follow-up with her multidisciplinary team of physicians.
CONCLUSION
In conclusion, we highlight a case of a 33-year-old woman treated with Adriamycin-based chemotherapy for Wilm’s tumor at age 10 years, and recently diagnosed with DCIS of the left breast, requiring whole-breast radiation. Due to Adriamycin exposure, she suffers from dilated cardiomyopathy. Considering her young age and pre-existing cardiac risk factors at the time of radiation therapy, sparing her heart as much as possible from exposure to radiation was the highest priority while planning this case. While DIBH is known to reduce dose to the heart, this patient was not a good candidate. The prone position has shown inconsistent results with respect to sparing of the heart. While other studies recommend not treating in prone position if the patient is unfit for DIBH or has a smaller breast volume, our case required a comparison of both positions before deciding on the optimal plan.
REFERENCES
- Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. New Engl J Med. 2013;368(11):987-998.
- Sixel KE, Aznar MC, Ung YC. Deep inspiration beath hold to reduce irradiated heart volume in breast cancer patients. Int J Radiat Oncol Biol Phys. 2001;49(1):199-204.
- Nissen HD, Appelt, AL. Improved heart, lung and target dose with deep inspiration breath hold in a large clinical series of breast cancer patients. Radiother Oncol. 2013;106:28-32.
- Lymberis SC, deWyngaert JK, Parhar P, et al. Prospective assessment of optimal individual position (prone versus supine) for breast radiotherapy: volumetric and dosimetric correlations in 100 patients. Int J Radiat Oncol Biol Phys. 2012;84(4):902-909.
- Kirby AM, Evans PM, Donovan EM, et al. Prone versus supine positioning for whole and partial-breast radiotherapy: a comparison of non-target tissue dosimetry. Radiother Oncol. 2010;96:178-184.
- Chino JP, Marks LB. Prone positioning causes the heart to be displaced anteriorly within the thorax: implications for breast cancer treatment. Int J Radiat Oncol Biol Phys. 2008;70(3):916-920.
- Verhoeven K, Sweldens C, Petillion S, et al. Breathing adapted radiation therapy in comparison with prone position to reduce the doses to the heart, left anterior descending coronary artery, and contralateral breast in whole breast radiation therapy. Pract Radiother Oncol. 2014;4:123-129.
- Vikstrom J, Hjelstuen MH, Mjaaland I, et al. Cardiac and pulmonary dose reduction for tangentially irradiated breast cancer, utilizing deep inspiration breath-hold with audio-visual guidance, wihtout comproimising target coverage. Acta Oncol. 2011;50:42-50.
- Kirby AM, Evans PM, Helyer SJ, et al. A randomised trial of supine versus prone breast radiotherapy (SuPr study): comparing set-up errors and respiratory motion. Radiother Oncol. 2011;100:221-226.
- Stegman LD, Beal KP, Hunt MA, et al. Long-term clinical outcomes of whole-breast irradiation delivered in the prone position. Int J Radiat Oncol Biol Phys. 2007;68(1):73-81.
- Feng M, Moran JM, Koelling T, et al. Development and validation of a heart atlas to study cardiac exposure to radiation following treatment for breast cancer. Int J Radiat Oncol Biol Phys. 2011;79(1):10-18.
- Chen JL, Cheng JC, Kuo SH, et al. Prone breast forward intensity-modulated radiotherapy for Asian women with early left breast cancer: factors for cardiac sparing and clinical outcomes. J Rad Res. 2013;54:899-908.
Citation
VA D, S L, RD S, YC L, S G. Optimizing treatment positioning to achieve better heart sparing in a left-sided, whole-breast irradiation. Appl Radiat Oncol. 2016;(2):22-26.
June 4, 2016