Locoregional Chemoradiation for a Patient with BRCA1 Stage IV Pancreatic Adenocarcinoma
Images

Abstract
Although the highest lifetime cancer risk in the setting of a BRCA 1 mutation is the development of a breast and ovarian malignancy, there is also a < 5% lifetime risk of pancreatic cancer.1 Recent recommendations suggest annual contrast-enhanced pancreatic MR imaging or endoscopic ultrasound for pancreatic cancer screening for these patients.2 In this case report, a patient undergoing MRI breast surveillance was incidentally found to have metastatic pancreatic cancer in the liver. The patient was treated with leucovorin, 5-fluorouracil, irinotecan, and oxaliplatin (FOLFIRINOX) alone and was rendered disease-free. Five years later, she developed an isolated nodal recurrence for which she received systemic gemcitabine and cisplatin chemotherapy with a partial response followed by consolidation chemoradiation to 50.4 Gy with intensity-modulated radiation therapy and concurrent capecitabine, with a complete response. This case highlights the potential for long disease-free intervals in the setting of BRCA1-metastatic pancreatic cancer and suggests an individualized role for locoregional radiation.
Keywords: BRCA1, ATM, pancreatic adenocarcinoma
Case Summary
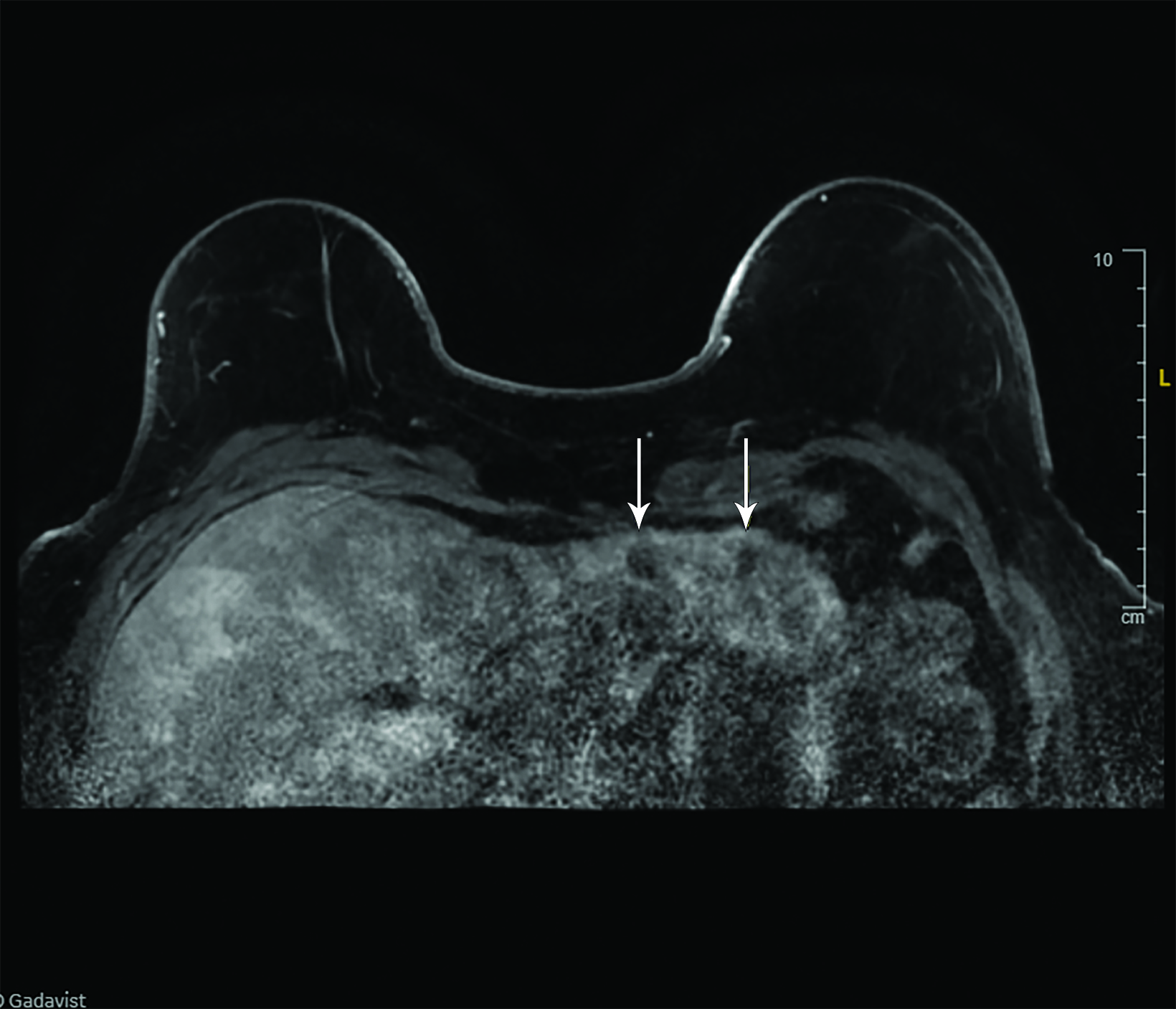
A 70-year-old White woman with no personal history of radiation therapy, but with a prior history of breast cancer following postbilateral mastectomy with transverse rectus abdominis muscle (TRAM) reconstructions, presented for her annual MRI in 2015. She was a heavy smoker with additional oncological history of stage III ovarian cancer 22 years prior and a family history of breast/ colon cancer. Multiple bilobar liver lesions were incidentally found on breast MRI and confirmed on triple-phase computed tomography (CT) and positron emission tomography (PET) scans, which also identified a hypermetabolic mass in the body of the pancreas with CA 19-9 > 400. Ultrasound-guided fine-needle aspiration (FNA) of a liver lesion confirmed the diagnosis of T2N0M1, stage IV pancreatic adenocarcinoma. She received 16 cycles of FOLFIRINOX (5-FU at 1800 gm, oxaliplatin at 65 mg/m2, and irinotecan at 100 mg/m2 every 3 weeks) followed by no evidence of disease until 2021, when FNA confirmed a celiac nodal recurrence. Germline testing confirmed BRCA1 and ATM mutations. Chemotherapy with gemcitabine and cisplatin achieved a partial radiographic response.
Capecitabine chemoradiation with 50.4 Gy of intensity-modulated radiation therapy (IMRT) achieved a complete response. She did not experience any known early or late toxicity related to her radiation treatment.
Diagnosis
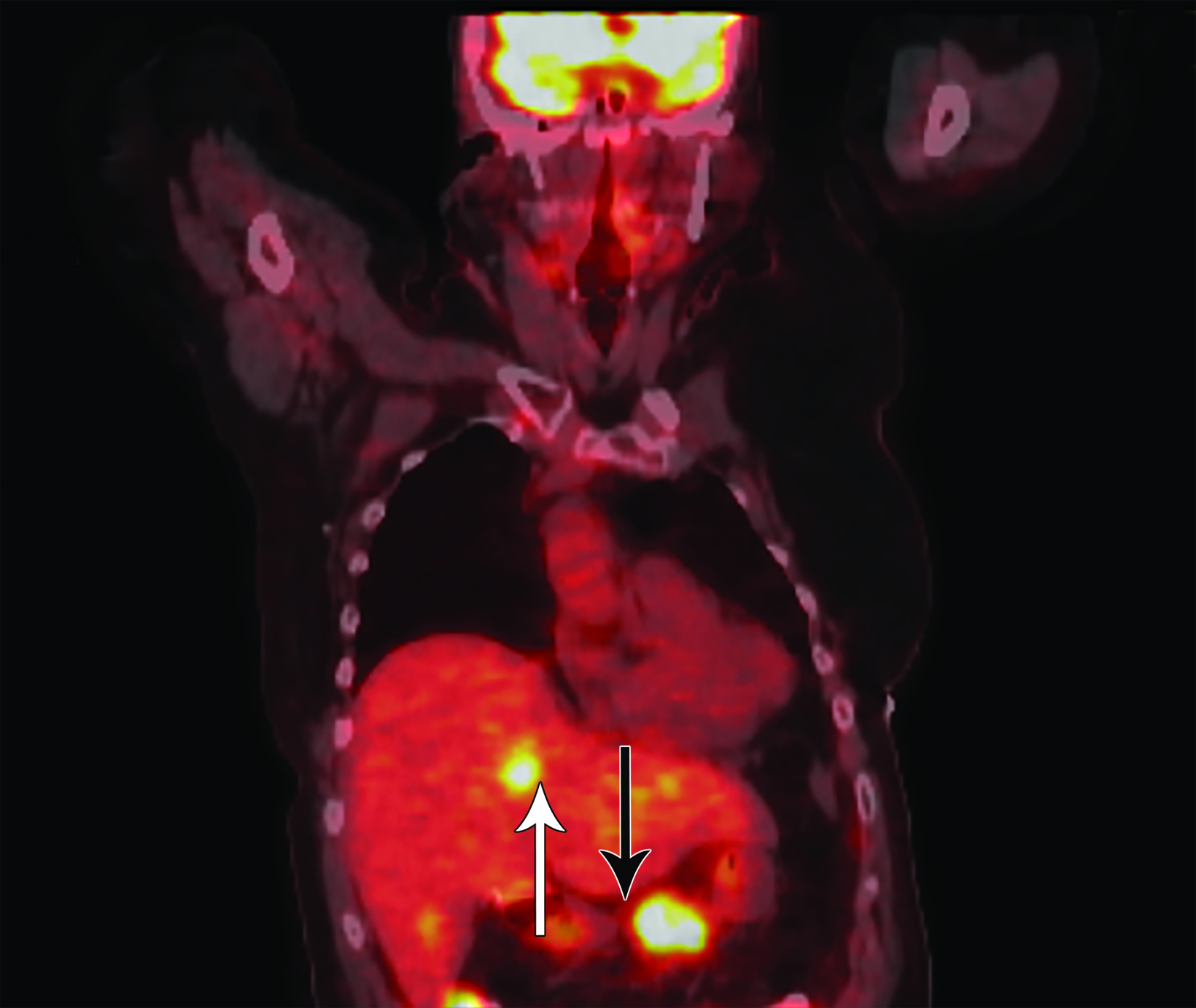
In this patient’s case, the workup confirmed she had developed a pancreatic body primary adenocarcinoma (Figure 1), which metastasized to multiple sites in the liver (Figure 2). There were no obvious involved abdominal lymph nodes on imaging at the time of initial diagnosis in 2015. The FNA of one of the liver lesions showed the cells were positive for CK7, CK19, CA125, CA19-9, pancy-tokeratin (AE1/AE3 CAM5.2), beta catenin, and GATA3.
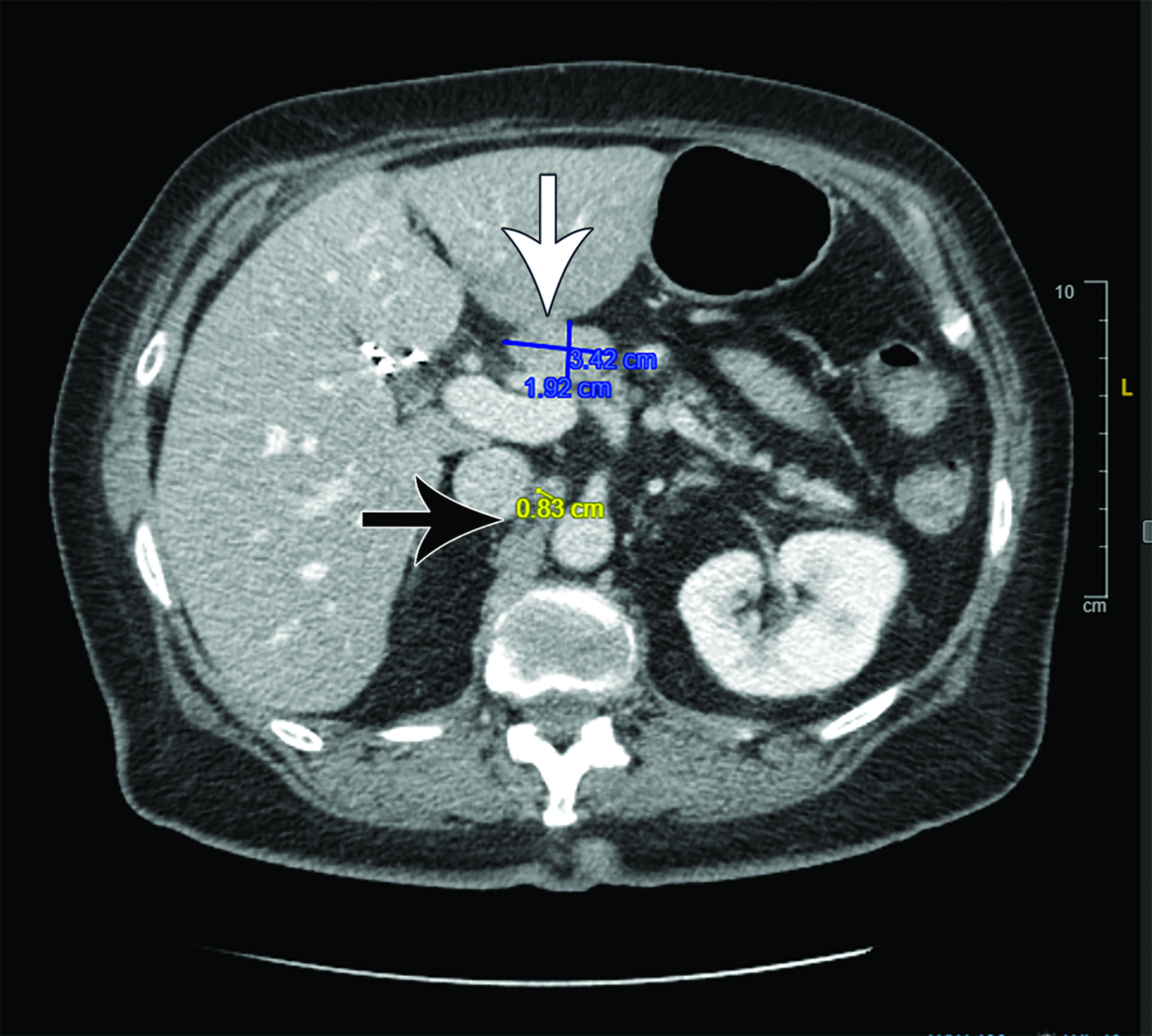
At the time of recurrence (Figure 3) in 2021, FNA of the celiac node was interpreted as poorly differentiated carcinoma consistent with the history of prior pancreatic cancer. After systemic chemotherapy with 3 months of gemcitabine and cisplatin every 21 days, the patient received chemoradiation with capecitabine and an IMRT plan to a dose of 45 Gy in 25 fractions to the clinical target volume followed by a boost of 5.4 Gy in 3 fractions (Figure 4) to the gross disease.
Discussion
Despite recent advances in cancer therapy, pancreatic cancer continues to have one of the lowest 5-year overall survival rates (11%).3 The majority of cases are sporadic but genomic evidence now suggests there is a heterogeneous landscape of molecular sub-types.4 Several genetic syndromes are associated with pancreatic cancer including mutations in DNA mismatch repair (Lynch syndrome), BRCA1 and BRCA2.5 The BRCA tumor suppressor genes are responsible for numerous functions regarding DNA-damage-dependent cellular checkpoints, DNA repair, and cell death.6 Mutations that alter the functions of these proteins may lead to targetable treatment. Given the rarity of BRCA-mutated pancreatic cancer (BMPC), it is unclear how much the prognosis differs from wildtype. However, there is agreement regarding the improved sensitivity of BMPC to platinum-based chemotherapies and poly (ADP-ribose) polymerase inhibitors, such as olaparib.7 The role of radiation therapy in BMPC is not clear, with some laboratory studies showing enhanced radiosensitivity, but clinical studies failing to show clearly improved outcomes.8
In addition to BRCA1, our patient had an ATM mutation, which is associated with ataxia telangiectasia and has been characterized by extreme radiosensitivity.9 Recent studies have shown that both heterozygous and homozygous ATM inactivation are associated with increased radiosensitivity10 and that patients with cancer and both BRCA and ATM mutations may have significantly increased radiosensitivity and an enhanced response to radiation therapies.11 ATM mutations have been of concern due to the potential of excessive radiation-associated toxicity particularly in breast cancer. Recent evidence indicates, however, that overall there is no excess in clinically significant acute toxicity, although caution is warranted as subvariants such as c5557G>A may have a higher risk of late toxicity or radiation-induced contralateral breast cancer.12,13
The clinical implications of germline genetic mutations illustrate the importance for genetic testing of all patients diagnosed with pancreatic cancer. Our patient declined BRCA testing after her first breast cancer diagnosis in her 30s, believing it was no longer important since she underwent bilateral mastectomy, only to be diagnosed with ovarian and pancreatic cancer over the next 40 years. Early genetic testing would have also revealed her ATM H231fs mutation earlier, which is significant given the associated increased cancer predisposition to malignancies of the lung, thyroid, pancreas and other areas, which could have led to more frequent screening intervals. In addition, the ATM mutation has relevance to her sensitivity to platinum chemotherapy agents since this mutation can be associated with increased response rates.14 The current standard of care is to offer genetic testing to all patients diagnosed with pancreatic cancer.15
In our case, the patient had a clinical complete response of her pancreatic primary and metastatic liver disease to FOLFIRINOX. Our patient has vastly exceeded the average prognosis of her condition with over 6 years of survival to date and 5 years of remission prior to recurrence.16 Interestingly, our patient relapsed in the celiac and adjacent nodes only without any recurrence in the pancreas primary site or liver. This was confirmed with an MRI scan of the abdomen before treatment initiation. After 3 months of gemcitabine
and cisplatin chemotherapy, the MRI showed a persistent nodal viable tumor. Since multidisciplinary tumor board evaluation centered around her current age of 76 and her comorbidities, focusing on treatments to maintain her quality of life were preferred, leading to the decision to proceed with consolidation to all sites of nodal activity with chemoradiation (CRT).
For gastrointestinal tumor sites with adenocarcinoma histology, there is a paucity of literature supporting radiation therapy for stage IV disease. In metastatic cancers of the esophagus, there is a potential survival benefit associated with CRT, suggesting that patients with chemotherapy intervals of 3 months or longer have improved outcomes, approaching 20% at 5 years for gastroesophageal junction tumors.17 In colorectal cancer patients, after immune checkpoint blockade, oligoprogression can be a frequent pattern of failure and local therapy strategies that include radiation may improve clinical outcomes.18 In pancreatic cancer, there is as yet no literature to support the role of consolidation to nodal targets post-chemotherapy for stage IV disease.
For our patient, the decision to offer her consolidation CRT was made after extrapolation from esophageal adenocarcinoma outcomes, recognizing that her radiosensitivity was likely increased secondary to her underlying BRCA1 and ATM mutational status.
IMRT was incorporated to maximize her normal tissue sparing since the potential to enhance organ at risk (OAR) sensitization was unknown. The patient tolerated her CRT well and did not have any high-grade toxicities. At nearly 1 year post-therapy, her CA 19-9 and imaging have not shown evidence of recurrence. The multidisciplinary tumor board has discussed the possibility of olaparib as maintenance therapy. Due to the results of the recent POLO trial,19 the patient prefers to consider this if she recurs and not as maintenance therapy, given the lack of a survival benefit.
Conclusion
This case highlights the potential for long disease-free survival in BRCA-mutated metastatic pancreatic cancer. Although enhanced radiosensitivity of BRCA-mutated tumors has been described in the laboratory, correlative clinical outcomes are lacking. The resolution of primary and metastatic disease on systemic FOLFIRINOX therapy for 5 years postdiagnosis supports the efficacy of platinum-based chemotherapy in this patient population. After regional nodal recurrence, salvage CRT was delivered to the abdomen, and nearly 1 year post-treatment, the patient remains radiographically and biochemically without evidence of disease. As expert consensus opinions recommend all patients with pancreatic cancer obtain genetic testing, more patients may be identified with BMPC, and a personalized strategy that includes radiation may be warranted for patients that respond to systemic therapy.
References
- Thompson D, Easton DF, Breast Cancer Linkage Consortium. Cancer incidence in BRCA1 mutation carriers. J Nat Cancer Inst. 2002;94(18):1358-1365. doi:10.1093/jnci/94.18.1358
- Roch AM, Schneider J, Carr RA, et al. Are BRCA1 and BRCA2 gene mutation patients underscreened for pancreatic adenocarcinoma? J Surgl Oncol. 2019;119(6):777-783. doi:10.1002/jso.25376
- Siegel, RL, Miller, KD, Fuchs, HE, Jemal, A. Cancer statistics, 2022. CA Cancer J Clin. 2022. https://doi.org/10.3322/caac.21708
- Lomberk G, Dusetti N, Iovanna J, Urrutia R. Emerging epigenomic landscapes of pancreatic cancer in the era of precision medicine. Nat Commun. 2019;10(1):3875. doi:10.1038/s41467-019-11812-7
- Goral V. Pancreatic cancer: pathogenesis and diagnosis. Asian Pac J Cancer Prev. 2015;16(14):5619-5624. doi:10.7314/APJCP.2015.16.14.5619
- Wu J, Lu LY, Yu X. The role of BRCA1 in DNA damage response. Protein & Cell. 2010;1(2):117-123. doi:10.1007/s13238-010-0010-5
- Leung K, Saif MW. BRCA-associated pancreatic cancer: the evolving management. J Pancreas. 2013;14(2):149-151. doi:10.6092/1590-8577/1462
- Bernier J, Poortmans P. Clinical relevance of normal and tumour cell radiosensitivity in BRCA1/BRCA2 mutation carriers: a review. Breast. 2015;24(2):100-106. doi:10.1016/j.breast.2014.12.003
- Ayars M, Eshleman J, Goggins M. Susceptibility of ATM-deficient pancreatic cancer cells to radiation. Cell Cycle. 2017;16(10):991-998. doi:10.1080/1538 4101.2017.1312236
- Aghamohammadi A, Akrami SM, Yaghmaie M, et al. Individual radiosensitivity assessment of the families of ataxia-telangiectasia patients by g2-checkpoint abrogation. Sultan Qaboos Univ Med J. 2018;18(4):e440-e446. doi:10.18295/squmj.2018.18.04.003
- Kim KH, Kim HS, Kim SS, et al. Increased radiosensitivity of solid tumors harboring ATM and BRCA1/2 mutations. Cancer Res Treat. 2022;54(1):54-64. doi:10.4143/crt.2020.1247
- Modlin LA, Flynn J, Zhang Z, et al. Tolerability of breast radiotherapy among carriers of ATM germline variants. JCO Precis Oncol. 2021;5:PO.20.00334. doi:10.1200/PO.20.00334
- McDuff SGR, Bellon JR, Shannon KM, et al. ATM variants in breast cancer: implications for breast radiation therapy treatment recommendations. Int J Radiat Oncol Biol Phys. 2021;110(5):1373-1382. doi:10.1016/j.ijrobp.2021.01.045
- Choi M, Kipps T, Kurzrock R. ATM mutations in cancer: therapeutic implications. Mol Cancer Ther. 2016;15(8):1781-1791. doi:10.1158/1535-7163.MCT-15-0945
- Mohindroo C, De Jesus-Acosta A, Yurgelun MB, Maitra A, Mork M, McAllister F. The evolving paradigm of germline testing in pancreatic ductal adenocarci- noma and implications for clinical practice. Surg Pathol Clin. 2022;15(3):491-502. doi:10.1016/j.path.2022.05.004
- Azar I, Virk G, Esfandiarifard S, Wazir A, Mehdi S. Treatment and survival rates of stage IV pancreatic cancer at VA hospitals: a nation-wide study. J Gastroin- test Oncol. 2019;10(4):703-711. doi:10.21037/jgo.2018.07.08
- Mizrak Kaya D, Wang X, Harada K, et al. 101 Long-term survivors who had metastatic gastroesophageal cancer and received local consolidative therapy. On- cology. 2017;93(4):243-248. doi:10.1159/000475550
- Marmorino F, Boccaccino A, Germani MM, Falcone A, Cremolini C. Immune checkpoint inhibitors in pMMR metastatic colorectal cancer: a tough challenge. Cancers. 2020;12(8):2317. doi:10.3390/cancers12082317
- Golan T, Hammel P, Reni M, et al. Overall survival from the phase 3 POLO trial: maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. J Clin Oncol. 2021;39(3_suppl):378. doi:10.1200/JCO.2021.39.3_suppl.378
Citation
P S, J A, S C, M A, J M, L B, R L, D WK, R P, J F, S H. Locoregional Chemoradiation for a Patient with BRCA1 Stage IV Pancreatic Adenocarcinoma. Appl Radiat Oncol. 2023;(1):44-48.
March 21, 2023