Is adjuvant radiation therapy an alternative to regional node dissection in select patients with lymph node-positive melanoma?
Images
Abstract
Objective: We report on regional lymph node irradiation for regional control of subclinical nodal disease in patients with node-positive melanoma.
Methods and Materials: We reviewed the medical records of 7 patients with biopsy-proven lymph node-positive melanoma treated with radiation therapy (RT) between 2007 and 2015 to assess treatment outcomes and toxicity. Patients who underwent completion lymph node dissection (CLND) or those with evidence of distant metastatic disease were excluded. Following sentinel lymph node biopsy (SLNB) or excision of a lymph node recurrence, subclinical regional disease was treated to 30 Gy in 5 fractions over 2.5 weeks. Two patients received adjuvant interferon. Median age at diagnosis was 70 years (range, 42-86 years). Median follow-up was 49 months (range, 10-114 months).
Results: No in-field or locoregional failures were observed. One patient was lost to follow-up 4 years after completing RT; at last follow-up, he was alive with no evidence of disease. One patient reported grade 1 extremity lymphedema after adjuvant RT to the inguinal lymph nodes. No other > grade 3 acute or late toxicities were recorded.
Conclusions: Based on our limited experience, adjuvant RT for subclinical regional disease in lymph node-positive melanoma may result in durable regional control without the potential added morbidity of a CLND. The risk of complications is likely lower than after a CLND and postoperative RT.
Standard-of-care treatment for lymph node-positive melanoma is a completion regional lymph node dissection (CLND). Routine CLND successfully identifies additional metastases in approximately 20% of patients who present with a positive sentinel lymph node biopsy.1-5 As a result, roughly 80% of patients undergo CLND with questionable survival benefit and the risk of potential anesthetic complications, postoperative wound problems, and chronic morbidities including lymphedema and paresthesias.6,7 Although hematogenous dissemination is the primary pattern of failure in patients with node-positive melanoma, the risk of locoregional failure after surgery alone is at least 20%, and increases with the number of positive lymph nodes and the presence of extracapsular extension.8-11 Furthermore, locoregional recurrence is often associated with significant morbidity.
Although adjuvant RT probably does not improve overall survival in patients with locally advanced melanoma, it has been associated with improved locoregional control in patients with subclinical regional disease.12 Most studies on RT for lymph node-positive melanoma have reported on the role of adjuvant RT following CLND.13-16 A single-institutional retrospective review of 36 patients with clinically apparent, nonsentinel parotid or cervical nodes treated with excision and postoperative RT alone reported a 5-year regional control rate of 93%.17
We undertook this study to examine whether adjuvant RT without CLND for subclinical regional disease in patients with sentinel node-positive melanoma or recurrent nodal melanoma after excision results in adequate regional control with minimal morbidity, obviating the need for CLND. Standard practice at our institution for patients with node-positive melanoma is CLND which, depending on the extent of disease, may be followed by postoperative RT. Seven patients were treated with excision and adjuvant RT without CLND at our institution in the last 20 years. Herein we report their outcomes.
Methods and Materials
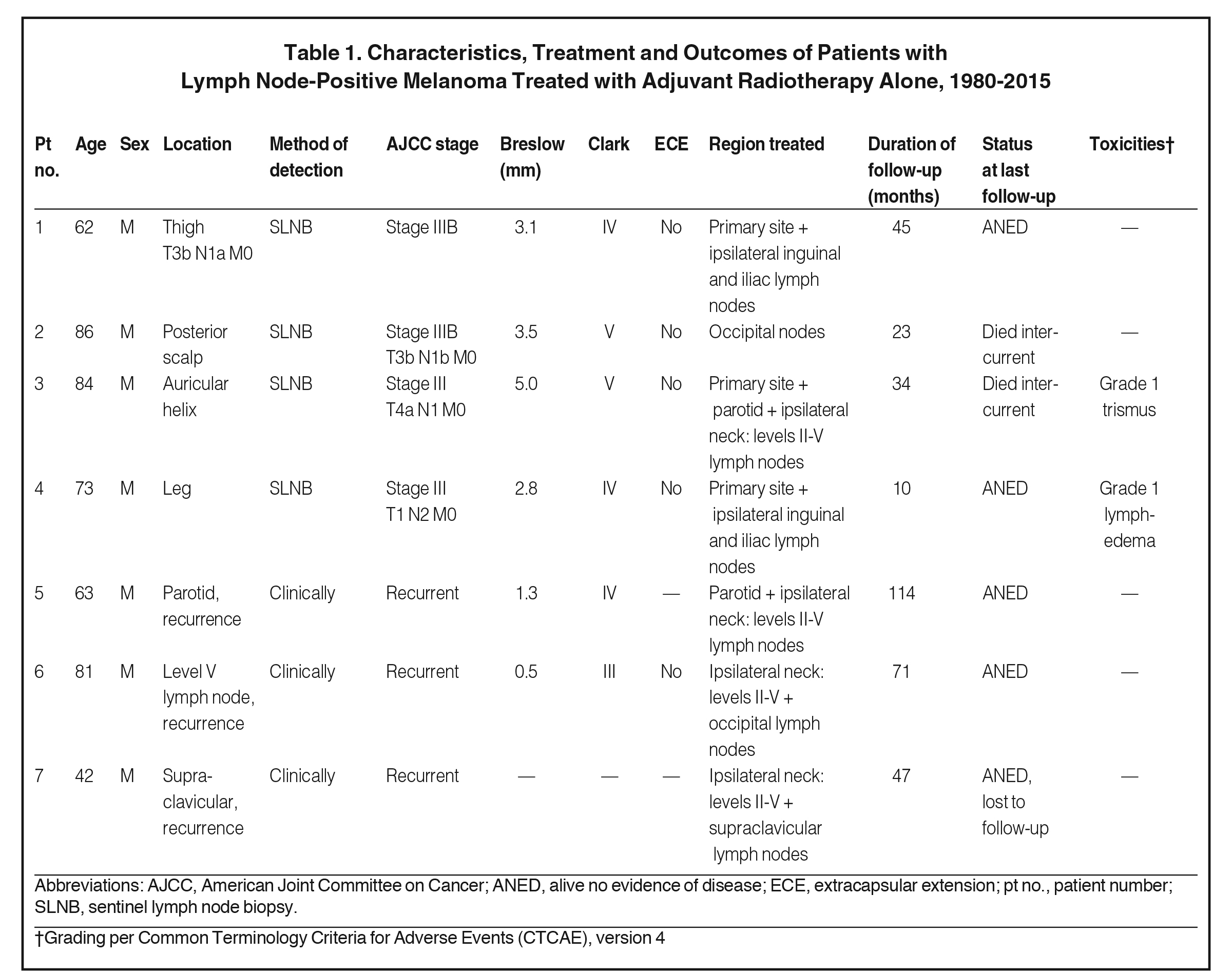
We reviewed the medical records of 7 patients with either sentinel lymph node-positive melanoma or melanoma recurrent to a single regional lymph node treated with excision and postoperative adjuvant RT at our institution between January 1986 and July 2015. Patients were excluded if they had undergone CLND, received previous RT to the involved lymph node basin, or had radiographic evidence on computed tomography (CT) or positron emission tomography (PET)-CT of residual or systemic disease following lymph node excision. Primary disease sites, which included the head and neck in 5 patients and an extremity in 2 patients, were treated with wide local excision +/- adjuvant RT. Four patients had sentinel lymph node-positive disease treated with adjuvant RT alone and 3 patients had a nodal recurrence of their previously excised primary melanoma treated with excision and adjuvant RT (Table 1
).
RT was delivered using either 3-dimensional conformal or intensity-modulated techniques. Beam orientation varied depending on the disease location and would encompass the entire lymph node region determined to be at high risk for subclinical disease. Regional lymph node basins were appropriate to the primary lesion (Table 1
). Head and neck nodal regions included cervical levels II-V with the addition of a low anterior neck field to include the supraclavicular nodes when appropriate. The parotid lymph nodes were included if thought to be at high risk. In patients with lower-extremity primaries, the inguinal and ipsilateral pelvic nodes were treated with intensity-modulated radiation therapy (IMRT). All patients were treated with a hypofractionated course of RT to a total dose of 30 Gy in 5 fractions over the course of 2.5 weeks as described by investigators at the University of Texas MD Anderson Cancer Center (Houston). Two patients were treated with adjuvant interferon, per the discretion of the treating medical oncologist.
Patients were seen in follow-up every 3 to 4 months during the first and second years and every 6 months thereafter. Toxicities were recorded and documented in accordance with the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0.18 Endpoints for the study were in-field locoregional control, disease-free survival, and overall survival.
Results
The median patient age at treatment was 70 years (range, 42-86 years). All patients were male. The median follow-up was 49 months (range, 10-114 months).
No in-field locoregional failures were observed. At last follow-up, no patient had developed distant disease. Two patients died of intercurrent disease at 2 and 3 years, respectively. The locoregional control, disease-free survival, and overall survival rates were 100%, 100%, and 71%, respectively.
No patient required a treatment break. There were no > grade 3 acute or late toxicities based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. One patient developed grade 1 lower extremity edema. Another patient developed grade 1 trismus.
Discussion
A recent Cochrane review7 sought to assess the effects and safety of SLNB followed by CLND for the treatment of localized previously untreated cutaneous melanoma. Based on available clinical evidence, we concluded that there is no clear benefit in overall survival or melanoma-specific survival in patients undergoing SLNB followed by CLND. Although no randomized data address this specific issue at present, SLNB followed by CLND remains the standard of care at most clinical practices owing to the risk of additional positive lymph nodes if an SLNB is positive. Although the risk of additional pathologically positive residual nodes after CLND for positive SLNB ranges from 17% to 28%,2-5 a substantial proportion of patients undergoing CLND risk complications, including postoperative wound-healing problems, chronic lymphedema, paresthesias, and anesthetic complications without a proven survival benefit.6
Although we are limited by our small patient population, we believe that CLND may not be necessary in node-positive melanoma treated with excision of clinically positive nodes and adjuvant postoperative RT for subclinical regional disease. This may be especially appropriate for patients thought likely to require postoperative RT. Adjuvant RT for subclinical regional disease appears to yield good locoregional control as evidenced by the absence of in-field or locoregional recurrences in our limited study population with a median follow-up of nearly 5 years. In the absence of a survival benefit, CLND may expose patients to an unnecessary additional morbidity without improving the likelihood of regional control.
Conclusion
Based on our limited data as well as that reported by Ballo et al,17 postoperative adjuvant RT for subclinical regional disease in lymph node-positive melanoma may result in durable regional control without the potential added morbidity of a CLND. Additionally, the risk of complications is less likely with postoperative RT than after a CLND. Further research is needed before adjuvant RT may be considered an alternative to CLND.
References
- Holmes EC, Moseley HS, Morton DL, et al. A rational approach to the surgical management of melanoma. Ann Surg. 1977;186:481-490.
- Wagner JD, Gordon MS, Chuang TY, et al. Predicting sentinel and residual lymph node basin disease after sentinel lymph node biopsy for melanoma. Cancer. 2000;89:453-462.
- Vuylsteke RJ, van Leeuwen PA, Statius Muller MG, et al. Clinical outcome of stage I/II melanoma patients after selective sentinel lymph node dissection: long-term follow-up results. J Clin Oncol. 2003;21:1057-1065.
- Pearlman NW, McCarter MD, Frank M, et al. Size of sentinel node metastases predicts other nodal disease and survival in malignant melanoma. Am J Surg. 2006;192:878-881.
- Sabel MS, Griffith KA, Arora A, et al. Inguinal node dissection for melanoma in the era of sentinel lymph node biopsy. Surgery. 2007;141:728-735.
- Morton DL, Wanek L, Nizze JA, et al. Improved long-term survival after lymphadenectomy of melanoma metastatic to regional nodes. Analysis of prognostic factors in 1134 patients from the John Wayne Cancer Clinic. Ann Surg. 1991;214:491-499; discussion 499-501.
- Morton DL, Cochran AJ, Thompson JF, et al. Sentinel node biopsy for early-stage melanoma: accuracy and morbidity in MSLT-I, an international multicenter trial. Ann Surg. 2005;242:302-311; discussion 311-303.
- Shen P, Wanek LA, Morton DL. Is adjuvant radiotherapy necessary after positive lymph node dissection in head and neck melanomas? Ann Surg Oncol. 2000;7:554-559; discussion 560-551.
- Lee RJ, Gibbs JF, Proulx GM, et al. Nodal basin recurrence following lymph node dissection for melanoma: implications for adjuvant radiotherapy. Int J Radiat Oncol Biol Phys. 2000;46:467-474.
- Calabro A, Singletary SE, Balch CM. Patterns of relapse in 1001 consecutive patients with melanoma nodal metastases. Arch Surg. 1989;124:1051-1055.
- O’Brien CJ, Coates AS, Petersen-Schaefer K, et al. Experience with 998 cutaneous melanomas of the head and neck over 30 years. Am J Surg. 1991;162:310-314.
- Ballo MT, Bonnen MD, Garden AS, et al. Adjuvant irradiation for cervical lymph node metastases from melanoma. Cancer. 2003;97:1789-1796.
- Burmeister BH, Mark Smithers B, Burmeister E, et al. A prospective phase II study of adjuvant postoperative radiation therapy following nodal surgery in malignant melanoma-Trans Tasman Radiation Oncology Group (TROG) Study 96.06. Radiother Oncol. 2006;81:136-142.
- Ballo MT, Zagars GK, Gershenwald JE, et al. A critical assessment of adjuvant radiotherapy for inguinal lymph node metastases from melanoma. Ann Surg Oncol. 2004;11:1079-1084.
- Ballo MT, Strom EA, Zagars GK, et al. Adjuvant irradiation for axillary metastases from malignant melanoma. Int J Radiat Oncol Biol Phys. 2002;52:964-972.
- Stevens G, Thompson JF, Firth I, et al. Locally advanced melanoma: results of postoperative hypofractionated radiation therapy. Cancer. 2000;88:88-94.
- Ballo MT, Garden AS, Myers JN, et al. Melanoma metastatic to cervical lymph nodes: Can radiotherapy replace formal dissection after local excision of nodal disease? Head Neck. 2005;27:718-721.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) v4.0. 2009. Available from: http://evs.nci.nih.gov/ftp1/CTCAE/About.html.
Citation
SR N, C S, WM M. Is adjuvant radiation therapy an alternative to regional node dissection in select patients with lymph node-positive melanoma?. Appl Radiat Oncol. 2017;(2):20-22.
June 2, 2017