IMRT, VMAT and image guidance: Changing the landscape of colorectal cancer treatment
Images

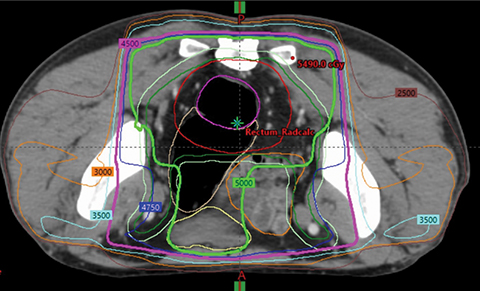
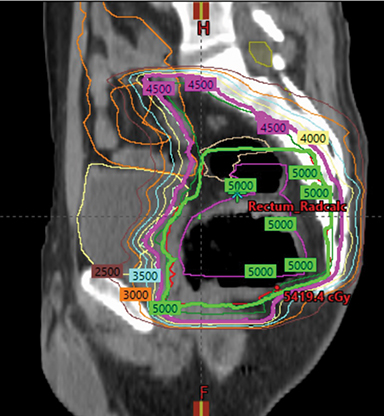
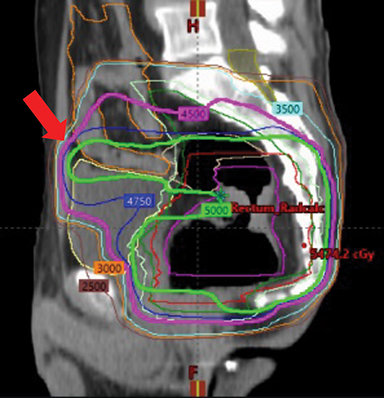
Surgery has long been a primary treatment for colorectal cancer. While it remains a fundamental pillar of curative therapy, it doesn’t come without potential consequences for a patient’s quality of life. For example, if the cancer is in the lower part of the rectum close to or involving the anus, an abdominal perineal resection (APR) is performed, resulting in removal of the anus and a permanent colostomy.1
Radiation therapy (RT), frequently in combination with chemotherapy, is often used as a neoadjuvant therapy prior to surgery, especially in rectal cancers (Figure 1). Although historically 3-dimensional conformal RT (3DCRT) has been used, intensity-modulated radiation therapy (IMRT) is now utilized more often when RT is included in a patient’s treatment. However, not all insurance plans in the US cover the cost of IMRT in colorectal cancer.
“Reimbursement is an issue. It is so frustrating that it often comes down to this because I want to do what is best for my patients and minimize the risk of toxicity associated with the treatment,” says Karyn A. Goodman, MD, MS, professor of radiation oncology and the David F. and Margaret Turley Grohne Chair in Clinical Cancer Research at the University of Colorado Denver School of Medicine. Dr. Goodman is also co-chair of the National Cancer Institute’s Gastrointestinal Cancer Steering Committee.
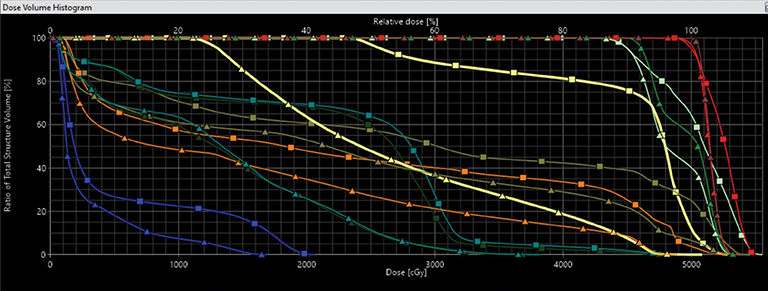
“If there was no cost difference in reimbursement between IMRT and 3D conformal, we would do all plans as IMRT,” Dr. Goodman says. (Figure 2 compares a dose volume histogram of a 3DCRT plan with an IMRT plan.) Her dosimetrist also prefers IMRT and, in most cases, VMAT (volumetric-modulated arc therapy) plans for rectal cancers. “They just feel more comfortable with IMRT than a plan that has 3 fields, uses wedges, etc.”
Dr. Goodman believes the RTOG 0822 clinical trial, which compared the gastrointestinal toxicity in patients treated with IMRT combined with 5-FU and oxaliplatin to a historical control group of patients treated with 3DCRT with 5-FU and oxaliplatin, has made it difficult to justify the benefit of IMRT.2 Unfortunately, the oxaliplatin, a chemotherapy drug found to have more GI toxicity when combined with 5-FU and RT, probably contributed more to the GI toxicity than the radiation so the GI toxicity rates were not different in the IMRT group vs the 3DCRT group.3,4
“Typically, GI toxicity is much lower in our clinic than what the RTOG 0822 trial was studying. Unfortunately, the use of IMRT couldn’t make up for the added toxicity of oxaliplatin,” Dr. Goodman says. “An ideal study would be a randomized prospective study; however, that likely will not be funded.”
Instead, she says, the best way to overcome the RTOG 0822 trial results is for radiation oncologists to continue publishing their studies regarding toxicity when using IMRT in rectal cancer patients. She is also hopeful that the new recommendations from the Centers for Medicare & Medicaid Services (CMS) that may tie reimbursement to the type of cancer, not the type of RT used, may alleviate this issue altogether.
Dr. Goodman was co-author of a study that examined toxicity profiles and outcomes among rectal cancer patients treated with either IMRT or 3DCRT prior to surgery to identify predictive clinical factors tied to increased toxicity.5 The study reported that more patients suffered from grade 2 diarrhea in the 3DCRT group, which also had greater odds of a higher diarrhea score than IMRT. Additionally, the 3DCRT group had higher grade 2 genitourinary toxicity (13 percent) vs the IMRT group (6 percent), which also had a trend toward decreased grade 2 proctitis (22 percent for the IMRT group vs 32 percent for the 3DCRT group).5
Location Matters
At Memorial Sloan Kettering Cancer Center (MSKCC), RT is primarily used for rectal cancers. According to Marsha Reyngold, MD, PhD, 3DCRT is the most commonly used technique, although IMRT is used for specific clinical situations depending on the location of the primary tumor.
“The small bowel is the sensitive organ that we want to avoid,” Dr. Reyngold explains. “There are various positioning techniques, such as a belly board, that we can use to avoid the small bowel with 3D conformal.”
IMRT is typically employed at MSKCC when a significant volume of the patient’s small bowel is inside their pelvis or if they have already undergone prior surgery for pelvic disease (benign or malignant), resulting in a fixed small bowel. If the rectal cancer involves the prostate, bladder, uterus or vagina, then the treatment area is larger and IMRT may be more beneficial as it allows for more bowel sparing. Also, IMRT is very useful in treating tumors that are low in the rectum where avoidance of the genitalia is key to preventing sexual dysfunction, Dr. Reyngold explains.
A recent article co-authored by Dr. Reyngold and Dr. Goodman analyzed the use of IMRT for locally advanced rectal cancer at National Comprehensive Cancer Network (NCCN) centers. Using the NCCN Colorectal Cancer Outcomes Database, the authors looked at trends in the use of IMRT between 2005 and 2011, patient factors in selecting IMRT and acute toxicity of IMRT and 3DCRT. While increased use of IMRT was reported, it was not uniform across the 9 NCCN sites included in the analysis. Furthermore, a key predictor of the use of IMRT in the cohort was RT dose and not having undergone surgery. Patients receiving more than 5040 cGy had triple the likelihood of receiving IMRT. Also, not undergoing surgery as part of treatment was also a predictor of receiving IMRT vs 3DCRT.6
“As a field, we need to also look at long-term toxicity, including sexual dysfunction or the risk of hip and bone fractures,” adds Dr. Reyngold. “We don’t have very good data, and while we believe [toxicity] is low, it is not zero,” she said, noting that use of IMRT may reduce long-term toxicity.
“While we use 3D conformal at MSKCC for the majority of patients, IMRT is not inferior, it is just less cost effective at this time,” she adds. “IMRT may be superior to 3D conformal for a larger group of patients than we currently know, but more work needs to be done to identify what that group may be.”
The Importance of Imaging
In addition to using standard linac-based IMRT plans, the University of California San Francisco (UCSF) Medical Center often uses VMAT integrated with image-guided radiation therapy (IGRT) to deliver precision radiation therapy.
“VMAT allows for a more rapid plan to be delivered that is more efficient and highly conformal,” says Mekhail Anwar, MD, PhD, assistant professor of radiation oncology at UCSF. Advances in treating rectal cancer have improved to the point where select patients with metastatic disease can be approached with curative intent. Dr. Anwar will also use stereotactic body radiation therapy (SBRT) in patients with metastases in the liver, bone or lung. Given the high doses used, image-guidance is an essential component in treatment planning for patients with colorectal cancers at UCSF.
“We capture cone-beam CTs to ensure we are aligning the patient precisely but also to look at internal changes, such as the bladder filling or bowel gas, that may impact dose to the internal organs,” Dr. Anwar explains. He can visualize unexpected changes prior to treatment, especially if the small bowel falls into the beam or if the bladder is not in the same condition—empty or full—as the simulation and plan. “Our goal is to deliver precise and safe treatments.”
Additionally, some cancers in the pelvis experience dramatic shrinkage while on therapy. This can be seen in advanced anal cancers, which are radiosensitive and can experience significant changes in size during treatment.
Although the decision to use IMRT or 3DCRT is guided by insurance, when IMRT is reimbursed Dr. Anwar and his colleagues typically use a VMAT plan.
Motivated by the challenges his patients face and tapping his PhD in electrical engineering, Dr. Anwar also conducts research on cancer imaging using microfabricated sensors and integrated circuits to evaluate patient response to treatment and increase personalized therapy.
IMRT and IGRT are complementary technologies that enable Dr. Anwar to deliver a high dose to the primary tumor as well as a meaningful yet lower dose to the surrounding areas at risk, using smaller margins on the volumes than a standard plan.
“IMRT and VMAT require more attention to detail as opposed to treating a large target area with a uniform dose,” Dr. Anwar explains. “These techniques involve more contouring precision and knowledge of the patient’s anatomy. We fuse not only our planning CT but also other clinical information, such as MRI and PET [positron emission tomography], to best interpret what areas are at risk for tumor development and to better understand patterns of spread.”
Dr. Goodman will also use VMAT at the University of Colorado because it delivers a good conformal dose and leads to more areas of low dose in the patient compared to a static IMRT plan.
“With static IMRT fields, we have a higher dose delivered from each field, but there are areas of the body with no entry dose and areas with less dose,” Dr. Goodman explains. “One area where we need to refine the planning technique with VMAT is in anal cancer or very low rectal cancer, which requires that we treat lymph nodes in the groin, and can increase the skin toxicity.”
She has noted that special attention must be given to sculpt around the genitalia.
Nonoperative Management
While an emerging trend is to avoid surgery in treating low rectal cancers, more clinical evidence is needed. There is growing interest as to whether resection of low-grade tumors—particularly in elderly patients or when the tumor is low or in the anus—can be treated only with RT, chemotherapy or other targeted agents.
“Part of this emerging paradigm is to look at dose escalation and radiosensitizing systemic agents,” says Dr. Anwar. “Both strategies are focused on delivering the maximal dose to the tumor while preserving the surrounding tissue. It is widely accepted that a permanent colostomy bag is not the most optimal outcome, although it is better than cancer returning. For, now the most concerted efforts are in determining who the right patient is for this approach.”
Dr. Reyngold was also a co-author of a study that investigated the relationship between a pathologic complete response to neoadjuvant therapy in locally advanced rectal cancer and the distance to the anal verge. Using clinical data from MSKCC, the authors reported “a bimodal association between the distance to the anal verge and pathologic complete response with low tumors (<4 cm) and higher tumors (8-10 cm and >10 cm) less likely to have a complete pathologic response.”7
“We want to identify patients with a complete response to chemo and chemoradiation who have a tumor that is close to the anal verge,” Dr. Reyngold explains. “Surgery in these patients means their sphincter will be removed, and that is a quality-of-life issue for them. If we can avoid surgery, we are giving something back to the patient.
“In the analysis, we found that distance was a factor that strongly correlated to response,” Dr. Reyngold adds.
The excitement surrounding nonoperative management of colorectal cancers is also supported by surgeons, says Dr. Goodman.
“Fifteen years ago, it was unheard of for a colorectal cancer patient to not have surgery. However, we know that in upwards of 25 percent of the cases, the patient has a pathological response after chemoradiation,” she adds. “In these patients, the prospect of avoiding surgery and still curing them is exciting.”
Dr. Goodman is studying options such as induction chemotherapy followed by chemoradiation and new combinations of targeted agents with chemoradiation to increase the rate of pathological response with chemo and RT.
“We can see over a 35 percent response rate where there is no cancer left,” Dr. Goodman explains. “We are using MRI and endoscopy to help identify these patients and make this decision prior to surgery.”
The idea is to do more focal dose painting with IMRT and then utilize a novel agent, such as a PARP inhibitor that prevents DNA damage by RT, or with a DNA-PK inhibitor that is radiosensitive and enhances the effect of RT, potentially improving the patient’s response to therapy.
“We are looking to move the field to enhance radiation in the pre-op setting to improve outcomes in select groups of patients and allow for nonoperative management,” Dr. Goodman adds.
The growing use of MRI and MR-based linear accelerators may also help assess response and personalize patient care in colorectal cancer. “We can now obtain a good anatomic look at the tumor using an onboard MRI in an MR-based linac,” says Dr. Anwar.
Dr. Anwar is also examining treatment of anal cancer in challenging populations, such as HIV-positive patients who may be more vulnerable to toxicities, as well as quantifying changes in patients, such as skin reaction and blood counts, before the toxicity of treatment leads to difficult side effects.
Integrating multiparametric MRI and molecular PET imaging with machine learning may also help radiation oncologists identify subtle changes, he adds. Genetic-based biomarkers will hopefully guide clinicians on which patients will respond to radiation with chemotherapy or radiation with a targeted therapy or immunotherapy, although development of better biomarkers are greatly needed.
“As we move to personalized therapy, daily imaging will be a big contributor for assessing response,” adds Dr. Anwar. “We can look more in depth at the response as it is actually happening to determine if we are on the right track and who is responding and may not need surgery.”
References
- American Cancer Society. Treatment of Colon Cancer, by Stage. https://www.cancer.org/cancer/colon-rectal-cancer/treating/by-stage-colon.html. Accessed July 17, 2019.
- Hong TS, Moughan J, Garofalo MC, et al. NRG Oncology Radiation Therapy Oncology Group 0822: a phase 2 study of preoperative chemoradiation therapy using intensity modulated radiation therapy in combination with capecitabine and oxaliplatin for patients with locally advanced rectal cancer. Int J Radiat Oncol Biol Phys. 2015;93(1):29-36.
- Boughton B. Oxaliplatin has few benefits, high toxicity in rectal cancer. Medscape, January 31, 2012. https://www.medscape.com/viewarticle/757785. Accessed July 17, 2019.
- Di Francia R, Siesto RS, Valente D, et al. Current strategies to minimize toxicity of oxaliplatin: selection of pharmacogenomic panel tests. Anticancer Drugs. 2013;24(10):1069-1078.
- Ng SY, Colborn KL, Cambridge L, et al. Acute toxicity with intensity modulated radiotherapy versus 3-dimensional conformal radiotherapy during preoperative chemoradiation for locally advanced rectal cancer. Radiother Oncol. 2016;121(2):252-257.
- Reyngold M, Niland J, Ter Veer A, et al. Trends in intensity modulated radiation therapy use for locally advanced rectal cancer at National Comprehensive Cancer Network centers. Adv Radiat Oncol. 2017;3(1):34-41.
- Patel SV, Roxburgh CS, Vakiani E, et al. Distance to the anal verge is associated with pathologic complete response to neoadjuvant therapy in locally advanced rectal cancer. J Surg Oncol. 2016;114(5):637-641.
Citation
MB M. IMRT, VMAT and image guidance: Changing the landscape of colorectal cancer treatment. Appl Radiat Oncol. 2019;(3):38-42.
September 4, 2019