Implementing adaptive radiation therapy for pancreatic and pancreatobiliary cancers
Images
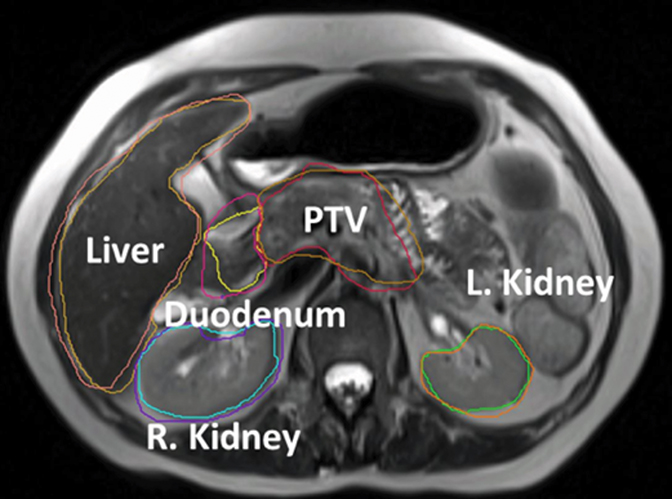
Image guidance using computed tomography (CT) and MRI has helped revolutionize the delivery of external-beam radiation therapy (EBRT). While imaging has been embraced for planning with most EBRT—intensity-modulated radiation therapy (IMRT), stereotactic body radiation therapy (SBRT), and 3D conformal—it is primarily used for initial treatment plans rather than for changes during the course of treatment. That, however, is changing thanks to the emergence of adaptive radiation therapy (ART).
There are two ways to adapt a treatment plan—“offline” and “online,” explains Parag J. Parikh, MD, associate professor of radiation oncology, at Siteman Cancer Center, affiliated with Washington University School of Medicine and Barnes Jewish Hospital in St. Louis, Missouri. “Most radiation therapy is administered day-by-day, fraction-by-fraction. After delivering some fractions, the oncologist may make a change in the plan and have it ready for future treatment delivery. Most sites do this type of offline adaption, reacting to a change in the patient or the tumor.”
While such changes do not require specialized technology, they do call for restarting the planning process, says Dr. Parikh.
“ART is a new way of thinking about the delivery of radiation therapy that requires new tools, software, processes, time and people to implement it,” he says. “What we are looking at is the ability to do online ART, meaning while the patient is on the table we are making a change in the plan based on what we see today.” That makes radiation therapy less like the classic description of EBRT and more like a series of operations.
Image Quality
At Froedtert Hospital and the Medical College of Wisconsin in Milwaukee, Beth Erickson, MD, FACR, FASTRO, and X. Allen Li, PhD, FAAPM, have been investigating the use of online ART for pancreatic and pancreatobiliary cancers for several years.
“Implementing online ART is technically challenging,” says Dr. Li, who uses ART for prostate cancer treatment. “Most sites use cone-beam CT for daily imaging; however, with CT there isn’t sufficient tissue contrast to delineate the pancreas from critical structures.” Image quality is the first challenge for ART of the pancreas. Another significant challenge is the QA process—verifying the plan to ensure safe delivery.
Protecting organs at risk is paramount as well. One subset of pancreatic cancer patients are those with locally advanced disease for whom surgery is not an option, explains Dr. Erickson. These patients receive a higher dose of radiation, which increases concerns regarding toxicity to the stomach and duodenum.
“We have a very fine line between what we need to deliver to the tumor and what the stomach and duodenum can handle. It is in this setting where ART can provide the biggest benefit,” she says. “With higher radiation, we have more local control for advanced disease. However, injury of the closely positioned GI tract can lead to the need for interventions, including surgery, so it’s a real concern.”
This is where MRI can provide higher quality images needed to delineate the tumor from the normal pancreas, she adds. While Froedtert has been using MR simulation for several years, Dr. Erickson plans to implement Elekta’s (Stockholm, Sweden) forthcoming integrated high-field 1.5T MR-guided linear accelerator to help better delineate the tumor and critical structures to enable plans with dose escalation.
For most pancreatic cancer patients, the traditional pre- or postoperative dose is approximately 50 Gy, Dr. Erickson explains. However, for locally advanced pancreatic cancer, the dose needs to be closer to or > 60-70 Gy to control the disease, she says. “That is a big gap between historical doses and where we would like to be,” she says. “We need to fill the gap with better imaging that enables us to pull the dose away from those critical structures while giving the tumor the dose that it needs.”
Drs. Li and Erickson are studying the ability to push the radiation dose close to 70 Gy with MR-based treatment planning, and have published several studies on ART for pancreatic patients. One study found that using ART with respiratory gating can reduce the dose to the duodenum from 69% to 18%.1 “If we can spare the duodenum, then we can escalate the dose,” Dr. Li says.
Image Registration
One roadblock to implementing online ART in the clinic is the need for a seamless, automated framework. Indrin J. Chetty, PhD, director of the Radiation Physics Division in the Department of Radiation Oncology at Henry Ford Hospital, Detroit, Michigan, has been working with his team in the area of deformable image registration and automation of the adaptive RT planning process for over a decade.
The automated process for estimating the dose-of-the-day involves: (A) deformable registration of the CBCT and planning CT datasets; (B) automatic deformable contour propagation of the target and normal organ contours from the CBCT to the planning CT, and email notification to the oncologist for review of these contours; and (C) computation of the dose on the deformed planning CT and accumulation of the dose for estimating the dose-of-the-day using tools such as DVH review.
There are technical challenges to the deformable image registration and dose mapping process, says Dr. Chetty. These include validation of the deformable image-registration algorithms; dealing with scatter and artifacts on cone-beam CT, which degrades image quality; and other issues such as registration of the planning CT with truncated cone-beam CT datasets for some treatment sites.
“The primary goal of deformable dose accumulation is to determine whether the intended dose at the planning stage is actually being accurately delivered to the patient,” Dr. Chetty explains. “By comparing the deviations between dose-of-the-day and the planned dose on a daily basis, we can see if we are indeed delivering what we intended; if we are missing the target or exceeding normal organ tolerance doses, then the oncologist will decide on how to adjust the plan, if considered necessary.”
Daily volumetric imaging and online correction enables us to reduce treatment planning margins. Deformable dose accumulation allows us to determine whether the target is being properly covered, which is especially important when planning margins are reduced. With regard to normal tissues, deformable dose accumulation captures interfraction variation in normal organ volumes, which provides better estimates of the doses to these organs over the course of fractionated treatment. Using the dose-to-date information from deformable dose accumulation, and based on the clinical judgment of the team, the treatment plan can be reoptimized midtreatment to meet the goals of the initially planned dose distribution.
For routine use of ART in the clinic, Dr. Chetty says that automation of the workflow process is a key element. “If, with the click of a few buttons, daily cone-beam CT images can be automatically submitted to the treatment planning system, deformably registered with the planning CT along with propagated contours, and be ready for the physician review, then such an automated process will facilitate broader utilization of deformable dose accumulation technology.”
Other challenges that have been resolved to some degree involve the accuracy of the deformable registration algorithms for contour propagation, he adds. However, the issue of estimating accurate deformed dose on a voxel-by-voxel basis is complex and requires much more investigation. Among the various algorithms used at Henry Ford Hospital, Dr. Chetty notes that Velocity (Varian Medical Systems, Palo Alto, California) for multimodality image registration, has performed well against benchmarks.
Dr. Chetty points out that ART takes a clinical champion (often a physician) interested in pursuing prospective studies involving computation of the dose-of-the-day. However, even with a clinical champion, the ART-based process is resource-intensive, and may not be feasible in many clinics without appropriate staff resources to implement it properly. In this regard, automation of the various steps will certainly provide impetus for increased clinical utilization of these tools, he says.
ART as a QA Tool
Compared to other types of EBRT, SBRT delivers higher doses of targeted radiation to treat tumors. At the Cancer Treatment Centers of America at Western Regional Medical Center, Phoenix, Arizona, Benjamin Slane, MD, and Matt West, PhD, have been treating pancreatic patients with SBRT using offline ART to minimize risk of toxicity when delivering high doses near critical structures such as the bowel. “We run ART to ensure that we are delivering the dose that we signed off on,” says Dr. Slane. “If not, we can make those changes on the first fraction.”
The center began using ART to help determine the precise dose delivered when anatomy changes. It’s a second QA step that takes about 5 minutes, says Dr. Slane.
“Early on we realized we could evaluate patient set-up and the robustness of the plan, including the direction of the beams,” adds Dr. West, who over the last 7 years has used the TomoTherapy System (Accuray, Sunnyvale, California) with some form of adaptive planning, and is an advocate for both online and offline ART.
ART is also ideal if the patient needs to be re-planned, says Dr. Slane, noting that ART has reduced the need for re-planning and re-contouring because it allows him to see that critical structures are safe from toxicity. “ART is a great decision tree tool,” he says. “It brings us all together—the therapist and oncologist—to identify patients at risk and decide whether we involve the physicist or proceed with treatment.”
The Value of ART in GI Cancers
Dr. Parikh uses online ART for pancreatic and pancreatobiliary cancers with real-time MR guidance on the MRIdian system (ViewRay, Cleveland, Ohio). ART is an important tool for pancreatic—or other gastrointestinal—cancers not because the tumor changes, Dr. Parikh says, but because of the need to track daily changes that can occur in the gastrointestinal system.
“These changes in the stomach and intestine are not repeatable,” he explains. “It is hard to get EBRT into the pancreas; but if we can adjust every day we deliver radiation, then maybe we can use higher doses.”
Another concern is intrafraction motion, in which anatomy moves during treatment. In the abdomen, this is often caused by respiratory motion. “This is an area where it seems online ART may help us deliver better treatments,” Dr. Parikh says. The real impact, he adds, is the ability to treat certain areas in the abdomen with higher doses knowing that he can evaluate dose safety while the patient is on the treatment table.
“I can adjust that dose around the organ at risk, and although we are giving the patient a more aggressive dose, we aren’t seeing higher toxicity,” he says. “It’s a change from tumor-specific to toxicity-specific dose delivery.”
ART is also a patient-centered approach, much like in the surgical suite. “You have to schedule the patients around the physicians—the work cannot happen without the physician being there, so this requires a change in how we work.” He likens it to brachytherapy or radiosurgery, where the oncologist is not only present during therapy, but is delivering it.
Perhaps most important, Dr. Parikh adds, is what radiation oncologists do with the information from online ART: “ART is more than just having images available; the key is to act on them.”
Reference
- Li XA, Liu F, Tai A, et al. Development of an online adaptive solution to account for inter- and intra-fractional variations. Radiother Oncol. 2011;100:370–374.
Citation
MB M. Implementing adaptive radiation therapy for pancreatic and pancreatobiliary cancers. Appl Radiat Oncol. 2016;(2):18-20.
June 4, 2016