FDA Clears Tumor Auto-contouring Solution for Radiation Therapy
 Vysioneer has announced US FDA 510(k) clearance for VBrain, reportedly the first-ever AI-powered tumor auto-contouring solution enabling a quicker response time for performing radiation therapy with more precision in targeting the tumor. Manual contouring time that may take several hours by clinicians is completed within a few minutes with the assistance of VBrain. As the first of its kind, the fully automated solution also ensures precision mapping of brain tumors with closer cuts and the ability to identify additional lesions that may be missed by the human eye. This is the first time the FDA has cleared an AI device for tumor auto-contouring in radiation therapy. Devices to receive FDA clearance prior to VBrain are specific to normal organ auto-contouring.
Vysioneer has announced US FDA 510(k) clearance for VBrain, reportedly the first-ever AI-powered tumor auto-contouring solution enabling a quicker response time for performing radiation therapy with more precision in targeting the tumor. Manual contouring time that may take several hours by clinicians is completed within a few minutes with the assistance of VBrain. As the first of its kind, the fully automated solution also ensures precision mapping of brain tumors with closer cuts and the ability to identify additional lesions that may be missed by the human eye. This is the first time the FDA has cleared an AI device for tumor auto-contouring in radiation therapy. Devices to receive FDA clearance prior to VBrain are specific to normal organ auto-contouring.
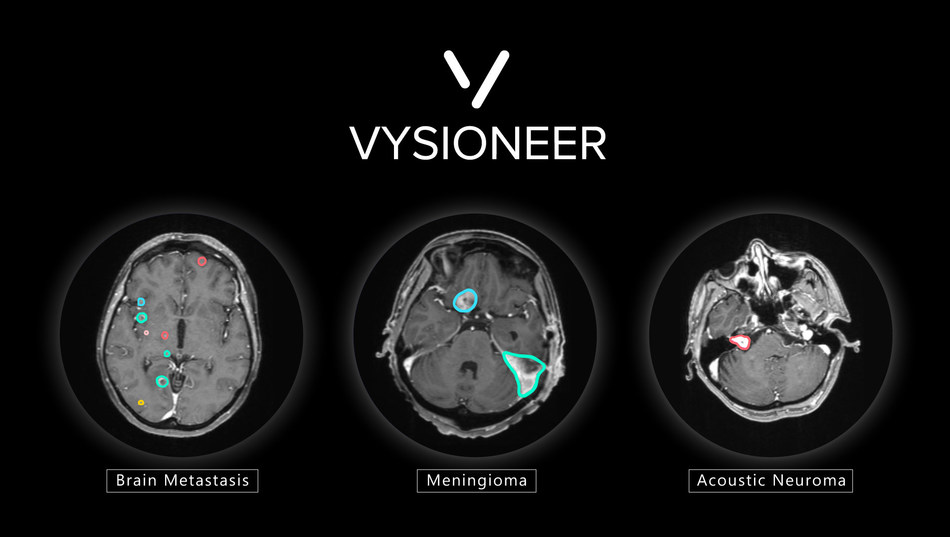
VBrain was tested at multiple sites throughout the US and Taiwan prior to receiving FDA clearance. The solution applies auto-contouring to the three most common types of brain tumors: brain metastasis, meningioma and acoustic neuroma. Additionally, the solution was extensively and rigorously tested through an 18-month clinical integration at National Taiwan University Hospital (NTUH), a leader in medical care for the country, with results published on the leading medical journal, Neuro-Oncology.
Clinical findings indicated clinicians assisted by VBrain demonstrated 12.2 percent higher sensitivity for lesion detection, and less experienced clinicians improved contouring accuracy with the added help. The efficiency in AI assistance also decreased treatment planning time at a median of 30.8 percent.
"There were distinct accuracy and efficiency improvements for clinicians of all skill levels," says Jason Chia-Hsien Cheng, MD, PhD, FASTRO, former director of radiation oncology at NTUH. "VBrain has a unique opportunity to influence future treatment on a global scale as a cloud-based software. Clinicians around the world, including areas lacking in resources, could utilize VBrain to achieve the world class standard of contouring."
VBrain has been integrated through Vysioneer's partnership with Mary Bird Perkins Cancer Center, a cancer care organization headquartered in Baton Rouge, Louisiana.
"I am thrilled to bring VBrain to our partners across the U.S. and Taiwan," says Jen-Tang Lu, CEO of Vysioneer. "Receiving FDA clearance for this solution allows Vysioneer to further its commitment of transforming radiotherapy workflows through developing full body auto-contouring solutions. The future of AI is near, bringing a second set of eyes and hands to assist clinicians in analyzing and segmenting medical scans and further improving patient cancer care."