Effect of radiation dose escalation on overall survival in ependymoma: A National Cancer Database analysis
Images

Abstract
Objective: Although adjuvant radiation therapy is the standard of care in treatment of localized grade II/III ependymoma, the appropriate dose to which to treat remains controversial. Excellent local control has been demonstrated after treatment to 5940 cGy, but there is no randomized evidence evaluating the effect of dose escalation on overall survival (OS). To address this question, we utilized the National Cancer Database (NCDB) to evaluate the effect of radiation dose escalation on OS in patients with localized ependymoma.
Materials and Methods: Patients > 2 years of age with localized World Health Organization (WHO) grade II and III ependymoma treated from 2010 to 2015 were identified from the NCDB and dichotomized into cohorts receiving 5400 cGy and ≥ 5940 cGy. OS was compared using the Kaplan-Meier estimator and multivariable Cox proportional hazards analysis.
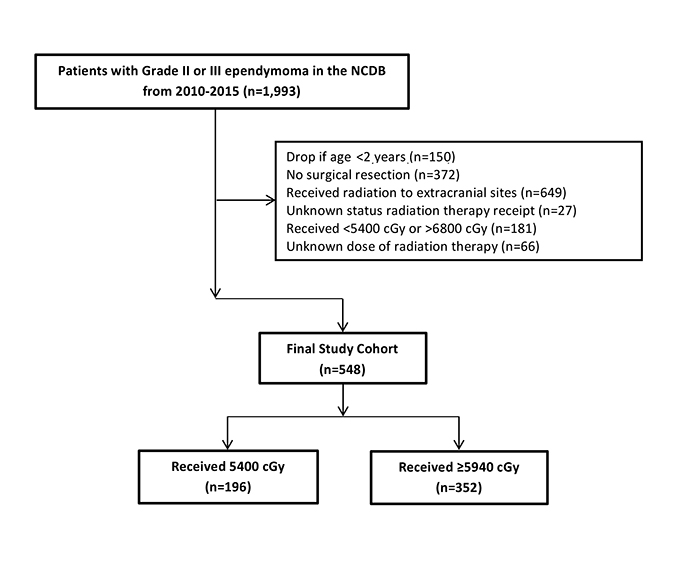
Results: A total of 548 patients met study criteria. Of these, 196 (36%) received 5400 cGy and 352 (64%) received ≥ 5940 cGy. Gross total resection was performed in 105 cases (54%) and 238 patients (43%) were ≤ 18 years of age. On multivariable survival analysis, there was no difference in OS between patients receiving 5400 cGy and those receiving ≥ 5940 cGy (hazard ratio [HR] = 0.74, 95% conformity index [CI]: 0.39-1.40, p = 0.36).
Conclusions: In this assessment of the NCDB, dose-escalated radiation of ≥ 5940 cGy was not associated with improved OS among patients with localized high-grade ependymoma. Further prospective study of the role of dose escalation in localized ependymoma is warranted.
Ependymoma is a rare primary malignancy of the central nervous system (CNS), which has an annual incidence of approximately 2-4 new cases per million with a peak age of approximately 5 years in children and 55 years in adults.1-3 The current standard of care for management of World Health Organization (WHO) grade II/III ependymoma includes maximal feasible resection followed by radiation therapy. While treatment to a dose of 5400 cGy is generally standard, progression-free survival (PFS) is only about 40% at 5 years.4-6 Therefore, dose escalation is an active area of interest in ependymoma management. A recent phase II study using dose-escalated radiation to 5940 cGy reported excellent local control, further supporting the potential benefits of higher doses.7
The benefits of dose escalation may be limited by late toxicities associated with radiation therapy to the CNS.8-10 In particular, risk of radiation necrosis may be correlated with increasing dose and volume delivered to the brainstem.11-13 Currently, there is no randomized evidence that evaluates the effect of dose escalation above 5400 cGy on overall survival (OS) in localized ependymoma. Therefore, we sought to evaluate the effect of radiation dose escalation on OS in patients with localized ependymoma utilizing the National Cancer Data Base (NCDB).
Materials and Methods
Data Source
The study population was identified from the NCDB, a national cancer registry sponsored by the American College of Surgeons and American Cancer Society that draws upon hospital registry data from Commission on Cancer-accredited facilities in the United States.14,15 Data are collected prospectively from cancer registries with nationally standardized data-coding definitions.16
Study Population
Inclusion criteria (Figure 1) consisted of patients >2 years of age at diagnosis with localized supratentorial or infratentorial WHO grade II and III ependymoma treated with surgical resection and adjuvant external-beam radiation therapy from 2010 to 2015. Dates were restricted to this time period to account for changes in practice based on a phase II dose-escalation study published in 2009.7 In addition, information regarding extent of resection was not available for intracranial tumors prior to 2010. Patients < 2 years old were excluded given that this group is often treated to lower dose due to concern for CNS toxicity.7 Those who received < 5400 cGy or > 6800 cGy were excluded as such doses may fall outside of the conventional dose range for treatment of ependymomas.17 Patients who received radiation to extracranial sites, had unknown dose data, or were not known to have received radiation therapy were also excluded.
Patient Cohorts and Variables
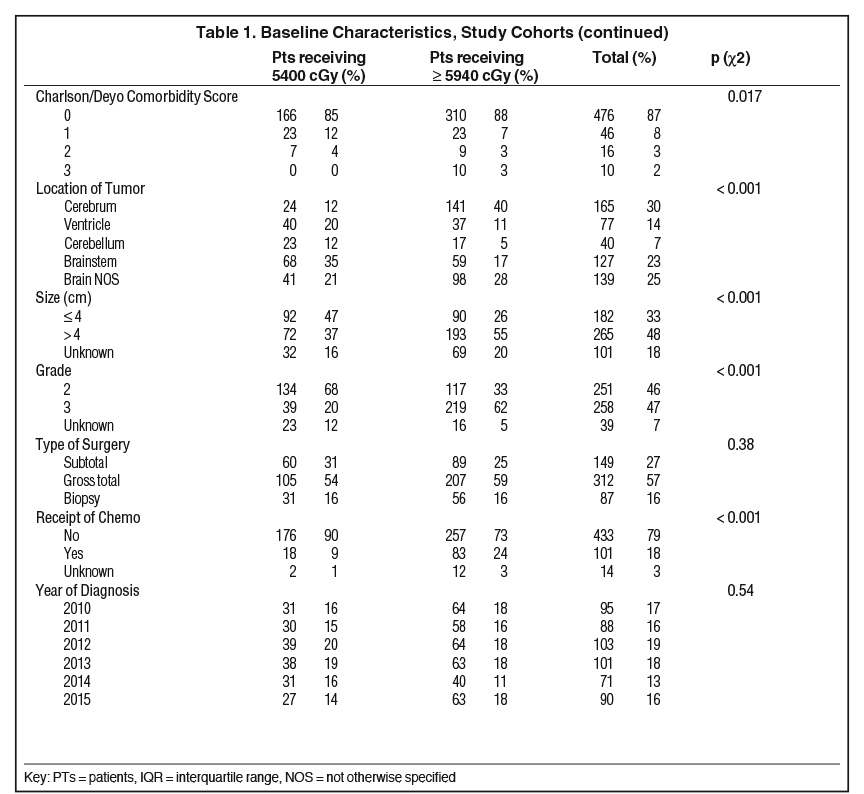
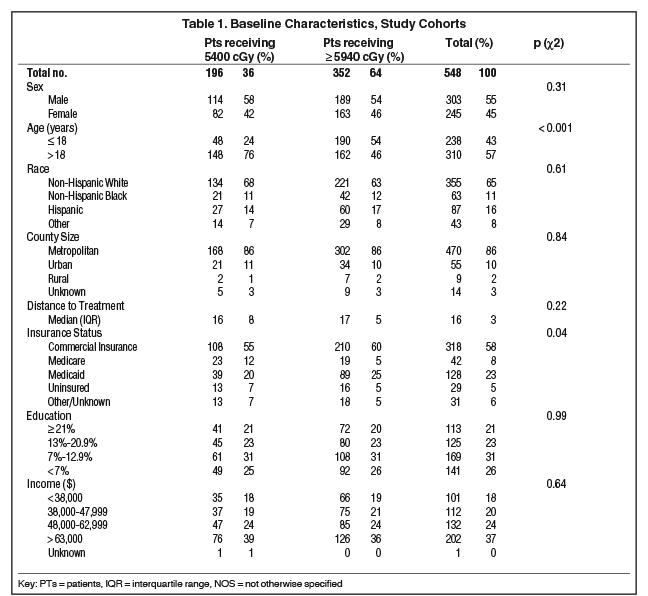
The overall cohort was divided into 1) a standard dose cohort that received 5400 cGy and 2) a dose escalated radiation cohort that received ≥ 5940 cGy. Covariates analyzed included gender, age, race, insurance status, treatment facility geographic location, distance to facility, education levels, income (median income in patients’ zip codes), Charlson-Deyo comorbidity score, tumor location (cerebrum, ventricle, cerebellum, brainstem, brain not otherwise specified), tumor size and grade, extent of surgical resection, receipt of chemotherapy, and year of diagnosis.
Aims/Endpoints
The primary endpoint was OS in patients treated to doses ≥ 5940 cGy compared to those treated with 5400 cGy. OS was defined as the time from diagnosis until death or last follow-up. As a secondary aim, we evaluated factors associated with receipt of dose-escalated radiation therapy.
Statistical Analysis
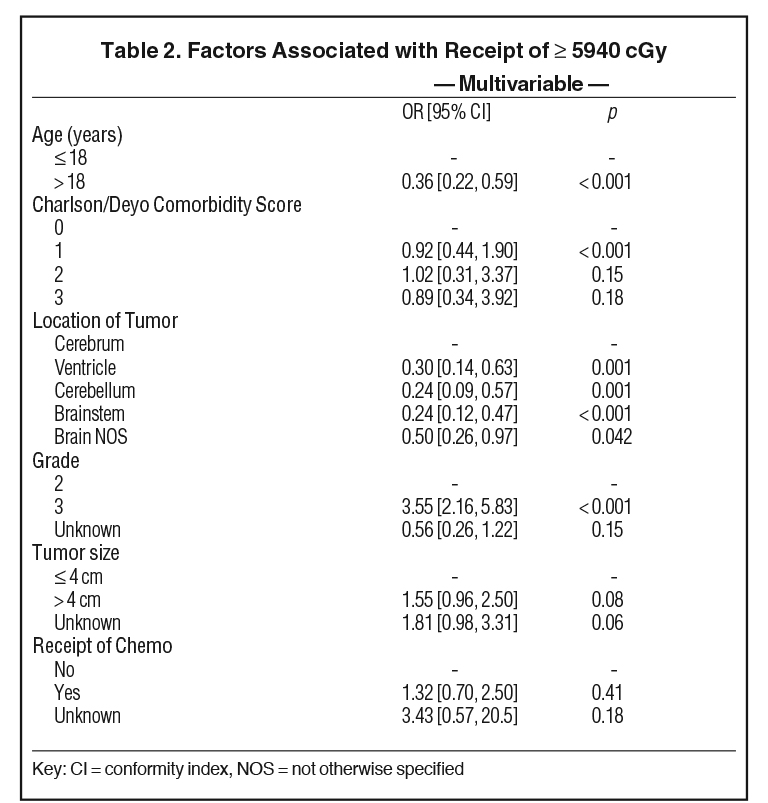
Baseline demographic and clinical characteristics between cohorts were compared using the chi-squared test for categorical variables and rank-sum tests for continuous variables. A multivariable logistic regression model was constructed using all baseline covariates reaching a univariable analysis threshold significance of p < 0.1 to assess the independent effect of each covariate on the likelihood of being treated with dose-escalated radiation therapy.
The Kaplan-Meier estimator and log-rank tests were used to assess OS between study cohorts. A Cox proportional hazards model was constructed using all variables achieving a threshold significance of p < 0.1 on univariable analysis to assess the independent effect of radiation dose on hazard of death. Patients diagnosed in 2015 were excluded from survival analysis due to insufficient follow-up data.
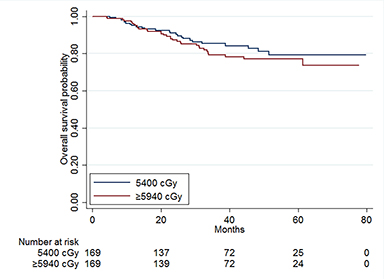
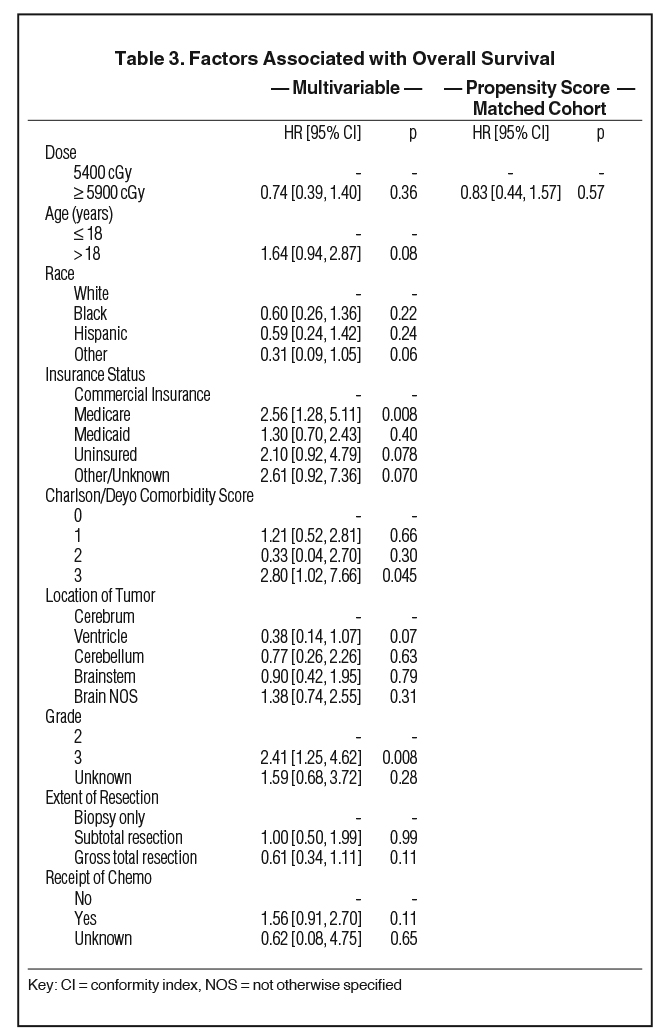
To more robustly account for baseline differences between study cohorts, a matched cohort of 338 patients (all 169 patients who received 5400 cGy matched with 169 patients who received ≥ 5940 cGy) was identified using 1-to-1 nearest neighbor propensity score-matching without replacement.18 Propensity scores were derived using multivariable logistic regression methods and denoted the probability of any patient receiving 5400 cGy. An absolute standardized difference of < 0.1 was accepted as a measure of adequate balance between matched covariates.19 Cox survival analysis was then repeated in the matched cohort to assess the robustness of the traditional multivariable analysis.
A two-tailed p-value < 0.05 was considered statistically significant. All statistical analyses were performed using Stata SE, version 14.0 (College Station, Texas).
Results
Patient Characteristics
A total of 548 patients met study inclusion criteria (Table 1). Of these, 196 (36%) were treated with standard dose radiation therapy and 352 (64%) were treated with dose-escalated radiation. Most patients were >18 years old (n = 310, 57%) and the median age of the cohort was 24 years (interquartile range [IQR], 8-24). Most patients had tumors in the cerebrum (n = 165, 30%), brainstem (n = 127, 23%), or unspecified location (n = 139, 25%). Tumors were most commonly > 4 cm (n = 265, 48%) and grade 3 (n = 258, 47%). Most patients underwent gross total resection (n = 312, 57%) and did not receive chemotherapy (n = 433, 79%).
Factors Associated with Receipt of Dose-escalated Radiation Therapy
On multivariable analysis, grade III disease was associated with receipt of dose-escalated radiation therapy (odds ratio [OR] 3.55, 95% conformity index [CI] 2.16-5.83, p < 0.001) (Table 2). Factors that predicted for a decreased likelihood of dose-escalated radiation therapy included age >18 years (OR = 0.36, 95% CI 0.22-0.59, p < 0.001), and tumor location outside of the cerebrum. Notably, extent of resection was not associated with dose-escalated radiation therapy.
Overall Survival
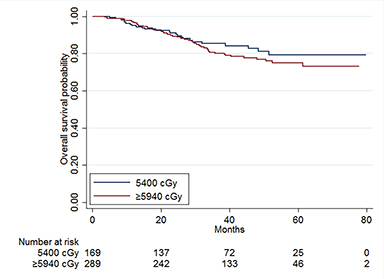
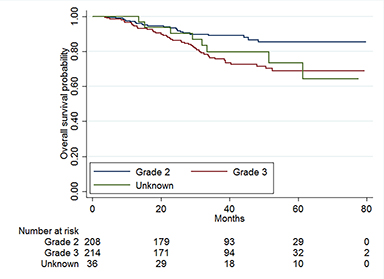
The median follow-up time for the entire cohort was 36.1 months (IQR, 24.4-51.8 months). The median 5-year OS was 79.6% for the standard dose cohort and 74.9% for the dose-escalated cohort (p = 0.86, Figure 2). No significant differences in OS were observed after propensity matching (p = 0.86, Figure 3). On univariable analysis, gross total resection (GTR) was associated with improved survival compared to biopsy alone (OR = 0.53, 95% CI 0.31-0.93, p = 0.026). On multivariable analysis, there was no difference between treatment with dose-escalated as compared to standard dose radiation therapy in the overall cohort (hazard ratio [HR] = 0.74, 95% [conformity index] CI 0.39-1.40, p = 0.36) or after propensity score matching (HR = 0.83, 95% CI 0.44-1.57, p = 0.57). GTR was not associated with OS (HR = 0.61, 95% CI 0.34-1.11, p = 0.11) (Table 3). Grade III disease was significantly associated withdecreased OS (HR = 2.41, 95% CI 1.25-4.62, p = 0.008) (Figure 4, p = 0.005)
Discussion
In this analysis of 548 patients with localized WHO grade II or III ependymoma identified from a national cancer registry, we found no difference in OS after receipt of dose-escalated radiation therapy.
Radiation therapy has been shown to improve OS and PFS in patients with localized ependymoma.20 While there have been no randomized comparisons of radiation dose, dose response has been observed in select retrospective studies. In an initial report from the Children’s Hospital of Philadelphia, local control was 32% for patients who received > 4500 cGy as compared to 0% for those who received lower doses.19 A subsequent study from the same institution demonstrated improved PFS after receipt of ≥ 5400 cGy.4 However, even after receipt of postoperative radiation therapy, patients remain at risk of local failure. In a recent study, patients who received radiation had a 58% risk of isolated local recurrence.5
Dose escalation has been evaluated in numerous studies in order to improve local control and survival outcomes. On the Pediatric Oncology Group protocol 9132, the potential benefits of dose escalation were evaluated using a hyperfractionated regimen of 6960 cGy (120 cGy twice daily). Patients had a 5-year event-free survival (EFS) of 52%, which compared favorably to historical controls.22 On an Italian Association of Pediatric Hematology Oncology (AEIOP) protocol, patients without residual disease were treated to 7040 cGy (110 cGy twice daily).23 Those with residual disease were treated with systemic therapy followed by the same radiation therapy. The 5-year PFS was 56% (95% CI 41-70%), 65% for those without residual disease (95% CI 49-82%) and 35% for those with residual disease (95% CI 10-61%). A subsequent AEIOP protocol attempted to improve results for patients with residual disease.17 Patients who underwent gross total resection received adjuvant radiation to 5940 cGy using standard fractionation while those with residual disease after surgery received chemotherapy followed by 5940 cGy to the resection cavity and an 800 cGy boost in 2 fractions of 400 cGy each to any residual disease. For the whole series, 5-year EFS and OS were 65.4% (95% CI 57.7-74.0%) and 81.1% (95% CI 74.6%-88.2%). The 5-year probability of local relapse was 20.7% (95% CI 14.8-29.1%).
Dose escalation to 5940 cGy using standard fractionation was also evaluated in a phase II study from St. Jude’s utilizing conformal radiation therapy.7 Given concern for toxicity, patients < 18 months after gross total resection were treated to 5400 cGy. The estimated 7-year local control and OS were 83.7% (95% CI 73.9-93.5%) and 81.0% (95% CI 71.0-91.0%), respectively. Based on these impressive results, dose-escalated radiation therapy to 5940 cGy has become a common treatment regimen and has been adopted in an ongoing national pediatric ependymoma protocol for patients ≥ 18 months in age, or < 18 months with subtotal resection.24
Increased radiation dose, however, may result in several late side effects of CNS radiation which can be debilitating or fatal. Pediatric patients may experience a decline in intelligence quotient which has been associated with total dose and dose per fraction.25-26 Radiation damage to critical structures can also result in endocrine and sensory changes.27-31 Radiation necrosis is of significant concern given proximity of many infratentorial tumors to the brainstem and has been reported in 2.5% of patients with ependymoma treated with dose-escalated radiation therapy.7 This risk has been correlated with dose and volume, especially the maximal dose and dose to 50% and 10% of the brainstem.11,12 In general, the entire brainstem may be treated to 5400 cGy using conventionally fractionated photon therapy without significant risk of permanent neurologic deficits. Smaller volumes may be irradiated to maximum doses of 5900 cGy and risk significantly increases with maximal dose > 6400 cGy.13 Given concern for increased risk of brainstem necrosis using proton radiation, more stringent brainstem constraints have been proposed.11
Other factors associated with OS in pediatric and adult ependymoma include greater extent of resection, low-grade disease, and supratentorial location.4,5,7 In this series, grade III disease was associated with decreased OS and increased likelihood of receiving dose-escalated radiation therapy. In multiple studies, GTR compared to subtotal resection has been associated with significantly improved OS.5,7 While GTR was associated with OS in univariable analysis, no association with extent of resection and OS was observed on multivariable analysis.
Several shortcomings limit the generalizability of the current study. First, there is inherent selection bias that cannot be controlled in the setting of a retrospective, observational study. In particular, bias toward treating patients with higher grade disease with dose-escalated radiation may have influenced outcomes seen on this study. Factors such as extent of resection, tumor location, and histopathologic grade are determined by the treating facility and not subject to central review, which may alter baseline patient characteristics given the relatively subjective nature of these features. Genetic information, which may affect the overall prognosis, was not available for inclusion in this analysis.
In addition, given the available data within the NCDB, no assessment of other treatment-related outcomes including local control, PFS, or toxicity could be made. While there are risks of dose-escalated radiation therapy, toxicities from local failure and salvage therapies may justify use of higher dose radiation even in the absence of an OS benefit. Furthermore, the limited follow-up of this study may not be enough to observe a meaningful difference in OS. Finally, although the NCDB captures a large volume of cancer cases, there is a lack of participation of many large independent children’s hospitals, which may bias results. This lack of pediatric cases is evident in our data where the majority of patients are ≥ 18 years, which is not representative of the epidemiology of this disease.
In conclusion, we show no OS benefit to dose-escalated radiation therapy in a population of pediatric and adult patients with localized, high-grade ependymoma identified from the NCDB. Given possible toxicities associated with dose-escalated radiation in the CNS, additional study to determine which patients may benefit from dose-escalated therapy is warranted.
References
- Gilbert MR, Ruda R, Soffietti R. Ependymomas in adults. Curr Neurol Neurosci Rep. 2010;10:240-247.
- Reni M, Gatta G, Mazza E, et al. Ependymoma. Crit Rev Oncol Hematol. 2007;63:81-89.
- McGuire CS, Sainani KL, Fisher PG. Incidence patterns for ependymoma: a Surveillance, Epidemiology, and End Results study. J Neurosurg. 2009;110:725-729.
- Shu HKG, Sall WF, Maity A, et al. Childhood intracranial ependymoma: Twenty-year experience from a single institution. Cancer. 2007;110:432-441.
- Marinoff AE, Ma C, Guo D, et al. Rethinking childhood ependymoma: a retrospective, multi-center analysis reveals poor long-term overall survival. J Neurooncol. 2017;135:201-211.
- Pollack IF, Gerszten PC, Martinez AJ, et al. Intracranial ependymomas of childhood: long-term outcome and prognostic factors. Neurosurgery. 1995;37:655-667.
- Merchant TE, Li C, Xiong X, et al. Conformal radiotherapy after surgery for paediatric ependymoma: A prospective study. Lancet Oncol. 2009;10:258-266.
- Armstrong GT, Liu Q, Yasui Y, et al. Long-term outcomes among adult survivors of childhood central nervous system malignancies in the childhood cancer survivor study. J Natl Cancer Inst. 2009;174:840-850.
- Indelicato DJ, Flampouri S, Rotondo RL, et al. Incidence and dosimetric parameters of pediatric brainstem toxicity following proton therapy. Acta Oncol. (Madr). 2014;53:1298-1304.
- Ruben JD, Dally M, Bailey M, et al. Cerebral radiation necrosis: incidence, outcomes, and risk factors with emphasis on radiation parameters and chemotherapy. Int J Radiat Oncol Biol Phys. 2006;65:499-508.
- Haas-Kogan D, Indelicato D, Paganetti H, et al. National Cancer Institute workshop on proton therapy for children: considerations regarding brainstem injury. Int J Radiat Oncol Biol Phys. 2018;101:152-168.
- Nanda RH, Ganju RG, Schreibmann E, et al. Correlation of acute and late brainstem toxicities with dose-volume data for pediatric patients with posterior fossa malignancies. Int J Radiat Oncol Biol Phys. 2017;98:360-366.
- Mayo C, Yorke E, Merchant TE. Radiation associated brainstem injury. Int J Radiat Oncol Biol Phys. 2010;76:S36-41.
- American College of Surgeons, National Cancer Database. https://www.facs.org/quality-programs/cancer/ncdb. Accessed February 13, 2018.
- Bilimoria KY, Stewart AK, Winchester DP, et al. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15:683-690.
- Philips J, Stewart A. Facility oncology registry data standards (FORDS): revised for 2013. Chicago, IL: American College of Surgeons; 2013.
- Massimino M, Miceli R, Giangaspero F, et al. Final results of the second prospective AIEOP protocol for pediatric intracranial ependymoma. Neuro Oncol. 2016;18:1451-1460.
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424.
- Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med. 2009;28(25):3083-3107.
- Rousseau P, Habrand JL, Sarrazin D, et al. Treatment of intracranial ependymomas of children: review of a 15-year experience. Int J Radiat Oncol Biol Phys. 1994;28:381-386.
- Goldwein JW, Leahy JM, Packer RJ, et al. Intracranial ependymomas in children. Int J Radiat Oncol Biol Phys. 1990;19:1479-1502.
- Kovnar E, Curran W TT. Hyperfractionated irradiation for childhood ependymoma: early results of a phase III Pediatric Oncology Group Study. J Neurooncol. 1997;33:268.
- Massimino M, Gandola L, Giangaspero F, et al. Hyperfractionated radiotherapy and chemotherapy for childhood ependymoma: final results of the first prospective AIEOP (Associazione Italiana di Ematologia-Oncologia Pediatrica) study. Int J Radiat Oncol Biol Phys. 2004;58(5):1336-1345.
- NIH U.S. National Library of Medicine. CinicalTrials.gov. Maintenance Chemotherapy or Observation Following Induction Chemotherapy and Radiation Therapy in Treating Younger Patients With Newly Diagnosed Ependymoma. https://clinicaltrials.gov/ct2/show/NCT01096368 Accessed August 10, 2018.
- Ullrich NJ, Embry L. Neurocognitive dysfunction in survivors of childhood brain tumors. Semin Pediatr Neurol. 2012;19:35-42.
- Padovani L, André N, Constine LS, et al. Neurocognitive function after radiotherapy for paediatric brain tumours. Nat Rev Neurol. 2012;8:578-588.
- Anderson DM, Rennie KM, Ziegler RS, et al. Medical and neurocognitive late effects among survivors of childhood central nervous system tumors. Cancer. 2001;92:2709-2719.
- Samaan NA, Vieto R, Schultz PN. Hypothalamic, pituitary and thyroid dysfunction after radiotherapy to the head and neck. Int J Radiat Oncol Biol Phys. 1982;8:1857-1867.
- Darzy KH, Shalet SM. Hypopituitarism following radiotherapy revisited. Endocr Dev. 2009;15:1-24.
- Merchant TE, Gould CJ, Xiong X, et al. Early neuro-otologic effects of three-dimensional irradiation in children with primary brain tumors. Int J Radiat Oncol Biol Phys. 2004;58:1194-1207.
- Jiang GL, Tucker SL, Guttenberger R, et al. Radiation-induced injury to the visual pathway. Radiother Oncol. 1994;30:17-25.
Citation
J V, S V, S S, RA L, C H, JE S. Effect of radiation dose escalation on overall survival in ependymoma: A National Cancer Database analysis. Appl Radiat Oncol. 2019;(3):29-36.
September 4, 2019