Craniospinal Irradiation for Leptomeningeal Disease in Recurrent Breast Cancer
Images


CASE SUMMARY
We present a case of leptomeningeal disease (LMD) as the first recurrence of breast cancer in a 40-year-old woman with BRCA2 germline mutation. Initially diagnosed in 2017 with left-sided cT2N1 ER-positive, PR-negative, HER-2-negative invasive mammary carcinoma, she received neoadjuvant FEC-D chemotherapy and bilateral mastectomy with immediate reconstruction with near complete pathologic response (ypT1bN0). She then received adjuvant chest wall and regional nodal irradiation, followed by tamoxifen.
She remained in remission for 22 months before developing progressive headaches, neck cramps, bilateral extremity paresthesias, myalgias, arthralgias and memory impairment. In the absence of focal neurologic deficit, tamoxifen was discontinued in October 2019. Re-staging computed tomography (CT) scans and a bone scan showed no evidence of recurrent disease.
She presented a month later with severe headache, nausea, vomiting, pulsatile tinnitus and visual blurring. Neurological examination identified bilateral sensory loss in the C8 distribution and marked papilledema. Enhanced MRI of the brain and cervical spine showed no evidence of LMD. Lumbar puncture revealed elevated opening pressure, high cerebrospinal fluid (CSF) protein and mild pleocytosis. High-volume (10 cc) CSF cytologic analysis confirmed metastatic breast cancer. Dexamethasone 8 mg twice daily was initiated with symptomatic improvement but with side effects including increased appetite, insomnia and agitation.
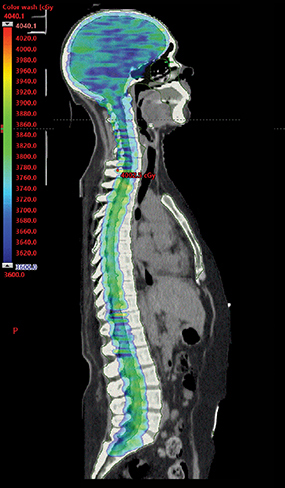
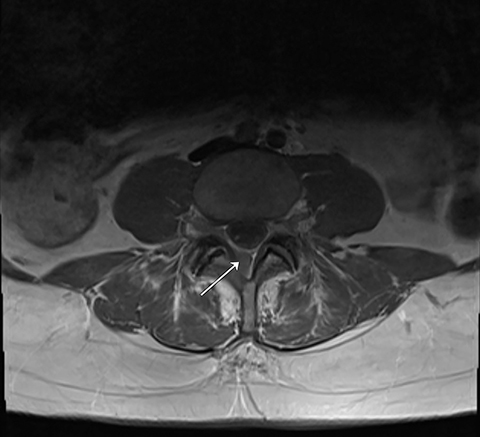
Her case was reviewed at multidisciplinary tumor board rounds and craniospinal irradiation (CSI) was recommended. Karnofsky performance status (KPS) was 80%. CSI using 3600 cGy in 20 fractions was delivered by volumetric-modulated arc therapy (VMAT) technique, completed in January 2020 and tolerated well (Figure 1). Three 360-degree arcs with separate isocenters and x coordinates aligning to the lateral midpoint of the full spinal column were planned using 6 MV photons. Linearly ramping junctions were designed as developed according to previously published institutional protocols.1 As per institutional image-guided radiation therapy guidelines, we used kV imaging for patient straightening and cone-beam CT imaging for each isocenter matched to anatomical setup prior to treatment. A 6-degrees-of-freedom couch was used. Re-imaging tolerances were 2 mm for translation and 1.5 degrees for rotation.2 The dose variance for set-up errors was expected to be less as shifts were always applied. The match was adjusted preferentially to optimize the overlap region, followed by ensuring complete planning target volume coverage of the entire brain and spine. The effects of allowable dose variance are not modelled prior to treatment on patient-specific plans at our institution. Her course was interrupted after fraction nine by a hospital admission for a right-hand burn and she was subsequently diagnosed with mania requiring dexamethasone taper and initiation of divalproex. This admission resulted in missing three consecutive days of treatment, which was resumed thereafter with no further treatment interruptions. She required six subsequent therapeutic lumbar punctures during CSI for symptom relief.
Her symptoms of headache, vision loss and nausea improved, and her neuropathic pain stabilized. She was able to completely taper off dexamethasone. There was complete resolution of papilledema and dexamethasone-related side effects. At multidisciplinary tumor board, palbociclib and letrozole after radiation therapy (RT) were recommended. Symptoms remained well controlled with a KPS of 70% at eight months post-RT without dexamethasone.
The patient presented with back pain 9 months post-RT and was found to have thoracic and lumbar epidural disease and mild hydrocephalus. Despite no evidence of radiological LMD, marked papilledema was again noted. She experienced a seizure requiring initiation of antiepileptic medications. Palliative RT to the lumbar spine was delivered with 2000 cGy in 8 fractions. Unfortunately, she continued to progress and a lumbar drain trial was unsuccessful for managing symptoms of increased intracranial pressure (ICP). Malignant cells were confirmed in CSF. She died 10 months post-RT, approximately one year after diagnosis of LMD.
IMAGING FINDINGS
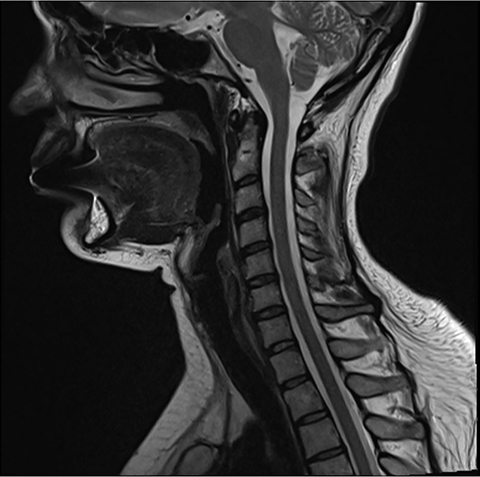
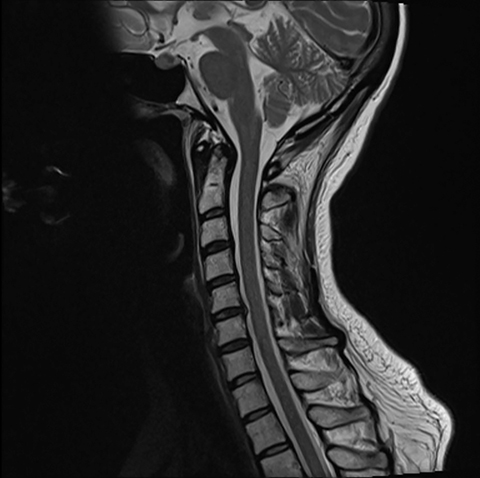
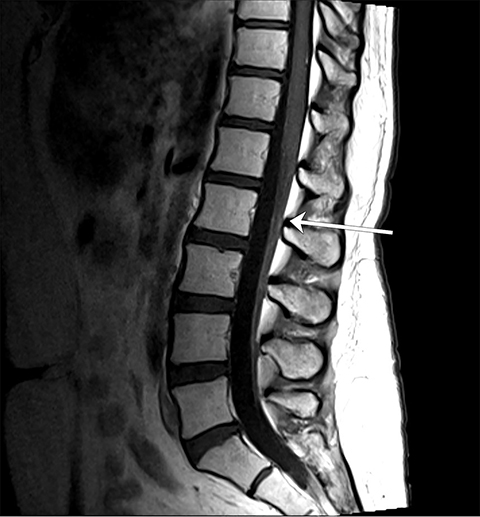
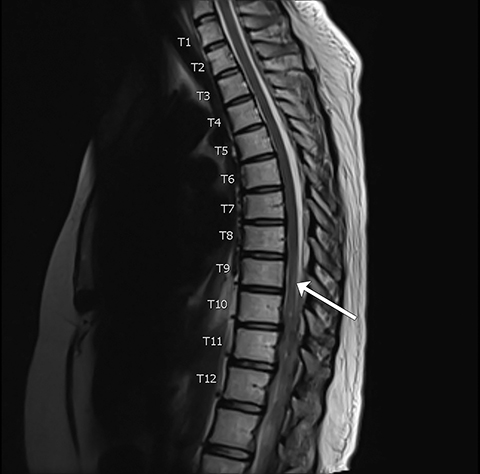
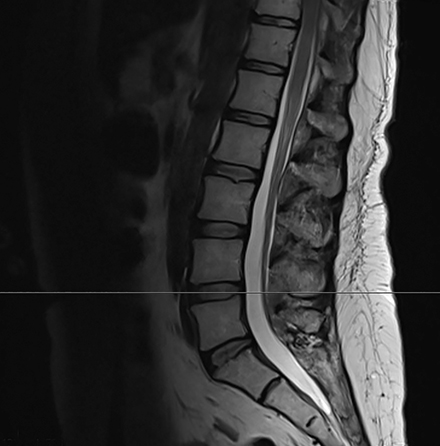
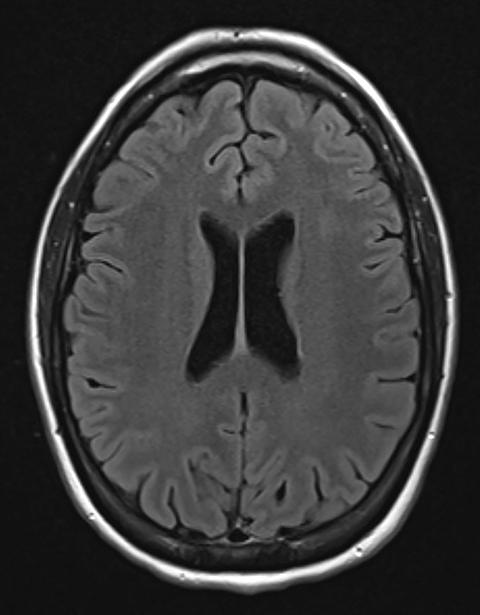
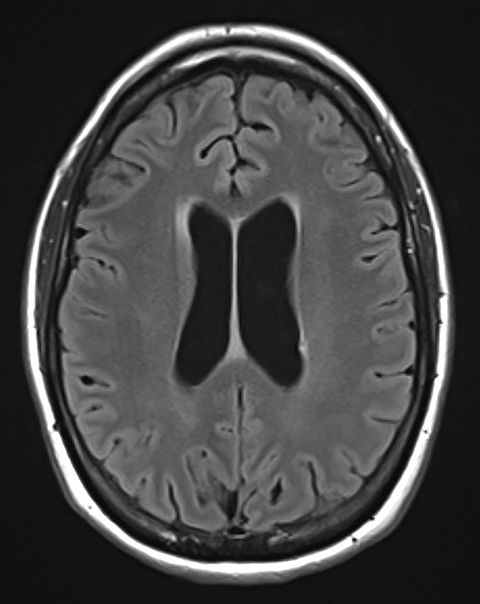
Pre-RT MRI of the brain and cervical spine showed no evidence of LMD or intracranial abnormality (Figure 2). A CT head venogram failed to demonstrate evidence of venous thrombosis. A re-staging bone scan and CT scans of the head, chest, abdomen and pelvis showed no evidence of recurrent or metastatic disease. Post-RT MRI spine images showed suspicious mild nodular enhancement of the cauda equina at the level of L2 and L3 suggestive for LMD (Figure 2). There was no evidence of intracranial LMD or disease elsewhere in the spine. At the time of disease progression, MRI spine showed diffuse thoracic and lumbar epidural enhancement with extension into the neural foramina and mild spinal canal stenosis from T6 to T10 (Figure 3). Mild progressive ventriculomegaly was also noted. There was no evidence of leptomeningeal enhancement.
DIAGNOSIS
LMD secondary to recurrent breast cancer
DISCUSSION
LMD occurs in up to 5% of breast cancer cases and portends a poor prognosis.3 Higher incidence rates and median survival in recent years may reflect improved detection on imaging and advances in systemic treatment options. LMD is defined as tumor cell infiltration of the pia mater, arachnoid, and subarachnoid space, which line the spine and brain. Common symptoms include headache, nausea, emesis, gait instability, cranial nerve deficits, seizures, motor and sensory impairment, and altered mental status. These symptoms result from increased ICP due to impaired CSF resorption or disease infiltration of local structures. MRI usage has now led to asymptomatic diagnoses of LMD.
LMD diagnosis often requires a combination of CSF cytologic analysis, imaging and neurological assessment. T1-weighted MRI with gadolinium is superior to CT for detecting LMD, although the sensitivity is only approximately 70%.4,5 MRI should be done prior to lumbar puncture and ventricular shunt placement where possible to prevent false positives from procedure-related meningeal contrast enhancement. Cytologic analysis is the gold standard for LMD diagnosis and is highly specific but poorly sensitive. High-volume CSF samples (>10 cc), immediate processing with avoidance of refrigeration, and CSF collection from a site known to have LMD can reduce false negative results. Repeat sampling of three high-volume CSF samples can increase cytology sensitivity up to 90%.4,5 Other abnormal CSF findings may include elevated opening pressure, elevated leukocyte count, elevated protein, and decreased glucose.4 Diagnostic algorithms recommend obtaining both cerebrospinal MRI and lumbar puncture. 4,6
This case highlights the diagnostic challenges of LMD. Despite presenting with signs and symptoms of increased ICP and irritation of the C8 nerve roots, no metastatic disease was visualized on gadolinium-enhanced MRI of the brain and cervical spine. We note that a full spine MRI was not included as part of the initial workup and it is unknown whether the lumbar spine disease seen on baseline pre-chemotherapy imaging was present prior to CSI. We recommend obtaining an MRI of the entire neuroaxis if LMD is suspected. In cases where no disease is visualized on MRI despite signs and symptoms suggestive of LMD, a high-volume lumbar puncture for cytologic analysis is also recommended. If there is a high degree of clinical suspicion for LMD, a total of three high-volume lumbar punctures may be pursued.4,5
Management options for LMD consist of a combination of systemic therapy, intrathecal chemotherapy, radiation therapy and best supportive care. Radiation therapy is indicated for symptomatic obstructive or bulky disease and may enhance intrathecal therapy delivery.7 Localized radiation approaches include whole-brain radiation therapy, involved-field radiation therapy and stereotactic radiation therapy. Several guidelines caution against CSI due to toxicity, namely myelosuppression.3,6 We would expect major toxicities to be limited by modern CSI delivery techniques including the use of VMAT, helical tomotherapy and proton therapy due to better dose conformity. For our VMAT CSI radiation therapy plan, mean bone marrow dose delivered was 1530 cGy and bone marrow V20 was 36%.
Historically, CSI has been a technically challenging approach and modern CSI delivery techniques attempt to address several of these technical considerations. Multiple field junctions pose dosimetric challenges for which shifts were conventionally used. Setup inaccuracies could result in either increased toxicity or reduced tumor control. In VMAT CSI, auto-feathering of field junctions aims to optimize multiple isocenter placement to lengthen the dose gradient, thereby improving dose homogeneity to the planned target volume and robustness to field setup error.8 Helical tomotherapy has the advantage of delivering a homogeneous dose over an extended vertical field without requiring field junctions, thus avoiding the associated challenges. Proton CSI results in less toxicity owing to more favorable dosimetry with dramatic dose sparing of anterior structures, and has been demonstrated to reduce gastrointestinal and hematologic toxicities.9 Vertebral body-sparing proton CSI is of particular interest in the pediatric population to preserve adult height potential and to reduce the risk of second malignancy.10 While preliminary study of a hypofractionated proton CSI regimen suggests safe delivery, it requires further investigation.11 A prospective clinical trial investigating vertebral body-sparing proton therapy in the pediatric population is underway and results are eagerly awaited.12
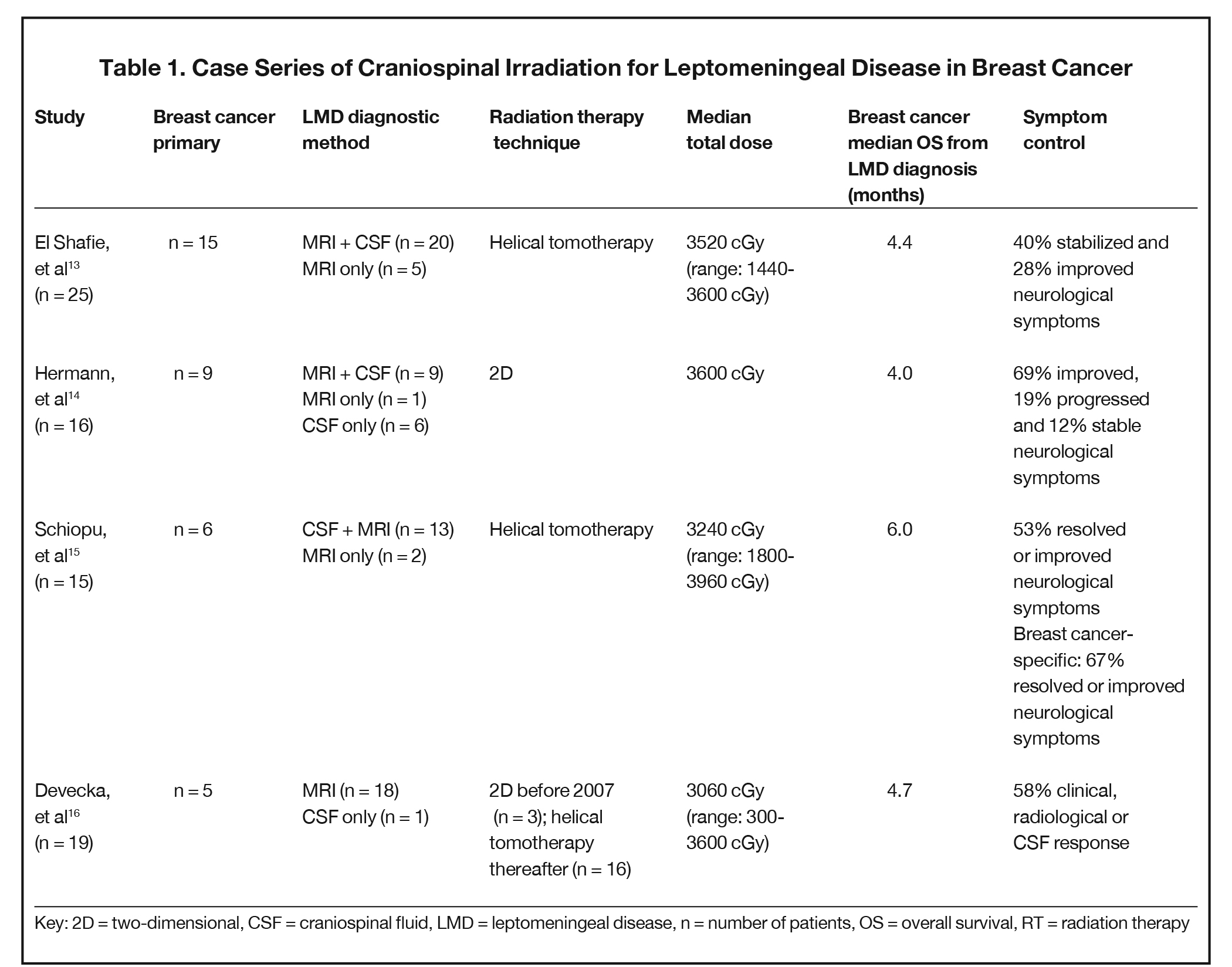
A summary of studies in breast cancer CSI for LMD indicate a role for palliation of symptoms (Table 1).13-17 Median overall survival from LMD diagnosis treated with CSI ranged from four to six months, with the majority of patients in each study reporting improvement or stability of response. Our patient responded remarkably well with several months of durable response before passing away 12 months after LMD diagnosis. In contrast to our patient treated with the VMAT technique, the majority of these studies used the helical tomotherapy technique, with evidence suggesting comparable effectiveness with the technical advantages previously discussed.18 The challenge of LMD diagnosis as seen in our case was also illustrated by a subset of patients who only demonstrated evidence of LMD on CSF cytology. However, it was unclear whether all patients received both imaging and CSF studies. We suspect that the number of patients with radiologically occult LMD is higher given that CSF cytology requires an additional invasive procedure that may not be pursued without a high degree of clinical suspicion.
With careful patient selection, evidence suggests that multimodality treatment with CSI has the potential for durable response.19 Criteria for consideration of palliative CSI, which have been associated with favorable responses, include KPS of 70 or greater, absence of extra-CNS disease, neurologic response, and age less than or equal to 55 years at LMD diagnosis.13,16 In our case of symptomatic LMD requiring multiple therapeutic lumbar punctures, treatment with CSI attained a good response facilitating dexamethasone tapering and no further therapeutic lumbar punctures.
CONCLUSION
LMD is an uncommon complication of breast cancer associated with a poor prognosis. We highlight the challenges of diagnosis and the importance of obtaining an MRI of the entire neuroaxis, cytologic analysis, and correlation with neurological assessment. In a carefully selected population, palliative CSI can provide a significant symptomatic benefit with minimal toxicity and should be considered in addition to systemic therapy and best supportive care.
REFERENCES
- Clements N, Bojechko C. VMAT CSI: Getting the junction right. In: AAPM 59th Annual Meeting & Exhibition. 2017 Accessed May 24, 2021. https://www.aapm.org/meetings/2017AM/PRAbs.asp?mid=127&aid=37642
- Strojnik A, Méndez I, Peterlin P. Reducing the dosimetric impact of positional errors in field junctions for craniospinal irradiation using VMAT. Rep Pract Oncol Radiother. 2016;21(3):232-239.
- Figura NB, Rizk VT, Armaghani AJ, et al. Breast leptomeningeal disease: a review of current practices and updates on management. Breast Cancer Res Treat. 2019;177(2):277-294.
- Franzoi MA, Hortobagyi GN. Leptomeningeal carcinomatosis in patients with breast cancer. Crit Rev Oncol Hematol. 2019;135:85-94.
- Wang N, Bertalan MS, Brastianos PK. Leptomeningeal metastasis from systemic cancer: review and update on management. Cancer. 2018;124(1):
- 21-35.
- Le Rhun E, Weller M, Brandsma D, et al. EANO–ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up of patients with leptomeningeal metastasis from solid tumours. Ann Oncol. 2017;28:iv84-99.
- Feyer P, Sautter-Bihl M-L, Budach W, et al. DEGRO Practical guidelines for palliative radiotherapy of breast cancer patients: brain metastases and leptomeningeal carcinomatosis. Strahlenther Onkol. 2010;186(2):63-69.
- Athiyaman H, Mayilvaganan A, Singh D. A simple planning technique of craniospinal irradiation in the eclipse treatment planning system. J Med Phys Assoc Med Phys India. 2014;39(4):251-258.
- Brown AP, Barney CL, Grosshans DR, et al. Proton beam craniospinal irradiation reduces acute toxicity for adults with medulloblastoma. Int J Radiat Oncol Biol Phys. 2013 Jun 1;86(2):277-284.
- MacEwan I, Chou B, Moretz J, Loredo L, Bush D, Slater JD. Effects of vertebral-body-sparing proton craniospinal irradiation on the spine of young pediatric patients with medulloblastoma. Adv Radiat Oncol. 2017;2(2):220-227.
- Yang TJ, Wijetunga NA, Yamada J, et al. Clinical trial of proton craniospinal irradiation for leptomeningeal metastases. Neuro-Oncol. 2021;23(1):134-143.
- MacDonald S. Craniospinal irradiation using proton beam scanning with selective vertebral body/bone sparing to improve marrow reserve and decrease growth decrement for children. 2021. Report No.: NCT03281889. Accessed May 18, 2021. https://clinicaltrials.gov/ct2/show/NCT03281889
- El Shafie RA, Böhm K, Weber D, et al. Outcome and prognostic factors following palliative craniospinal irradiation for leptomeningeal carcinomatosis. Cancer Manag Res. 2019;11:789-801.
- Hermann B, Hültenschmidt B, Sautter-Bihl ML. Radiotherapy of the neuroaxis for palliative treatment of leptomeningeal carcinomatosis. Strahlenther Onkol Organ Dtsch Rontgengesellschaft Al. 2001;177(4):195-199.
- Schiopu SR, Habl G, Haefner M, et al. Helical tomotherapy in patients with leptomeningeal metastases. Cancer Manag Res. 2019;11:401-419.
- Devecka M, Duma MN, Wilkens JJ, et al. Craniospinal irradiation (CSI) in patients with leptomeningeal metastases: risk-benefit-profile and development of a prognostic score for decision making in the palliative setting. BMC Cancer. 2020;20(1):501.
- Morikawa A, Jordan L, Rozner R, et al. Characteristics and outcomes of patients with breast cancer with leptomeningeal metastasis. Clin Breast Cancer. 2017;17(1):23-28.
- Myers PA, Mavroidis P, Papanikolaou N, Stathakis S. Comparing conformal, arc radiotherapy and helical tomotherapy in craniospinal irradiation planning. J Appl Clin Med Phys. 2014 Sep 8;15(5):12-28.
- Meissner M, Addeo A. intrathecal methotrexate and craniospinal radiotherapy can be an effective treatment of carcinomatous meningitis in patients with breast cancer: case reports. Case Rep Oncol. 2016;9(3):586-592.
>
Citation
JK K, M Y, JW H, D G, N L. Craniospinal Irradiation for Leptomeningeal Disease in Recurrent Breast Cancer. Appl Radiat Oncol. 2021;(2):42-47.
July 27, 2021