Complete response after treatment with pembrolizumab in a patient with metastatic cutaneous squamous cell carcinoma involving th
Images

CASE SUMMARY
An 84-year-old man with a history of numerous nonmelanomatous skin cancers of the face presented with blurry vision and diplopia, as well as numbness and foreign body sensation to the right face. MRI demonstrated an enhancing mass encompassing the lateral rectus muscle in the right orbit, extending dorsally until the anterior cavernous sinus (Figure 1A). This prompted a biopsy of the right orbital mass, which revealed poorly differentiated squamous cell carcinoma, suggesting recurrence of a previously treated cutaneous squamous cell carcinoma (cSCC) of unknown location. The mass was confirmed on staging positron emission tomography/computed tomography (PET/CT) with no evidence of nodal or distant metastatic involvement. The patient declined surgical intervention and instead was initiated on pembrolizumab, 200 mg every 3 weeks for 2 years, without any additional adjuvant chemotherapy or radiation therapy. For the patient’s convenience, pembrolizumab was chosen over nivolumab, which is dosed twice a week. At the patient’s next follow-up visit, 2-and-a-half weeks after the first cycle, the patient noted improvement of his diplopia and complete resolution of associated pain. An MRI obtained 4 months after initiation of pembrolizumab demonstrated a near complete response of the mass. At the patient’s most recent follow-up, 2 years after initiation of pembrolizumab therapy, the patient has maintained his near complete response on follow-up MRIs, with further resolution of his visual symptoms (Figure 1B). Throughout treatment, the patient experienced minimal toxicity, including hypothyroidism treated with replacement thyroid hormone.
IMAGING FINDINGS
Initial diagnostic MRI demonstrated an enhancing mass encompassing the right lateral rectus muscle extending posteriorly until the anterior cavernous sinus. A staging PET/CT scan demonstrated an F-18 fluorodeoxyglucose (FDG)-avid soft-tissue mass of the right orbit consistent with the mass seen on MRI as well as 2 FDG-avid cutaneous nodules in the left cheek and left posterior ear respectively, but no evidence of nodal or distant metastatic disease. The 2 cutaneous nodules, 1 of which was proven on biopsy to be basal cell carcinoma, were no longer noted on examination after completion of pembrolizumab therapy. During treatment with pembrolizumab, follow-up MRIs were obtained every 2 months for the first 6 months and then every 3 months afterward. These demonstrated continued near complete response of the cancer beginning 4 months after starting therapy.
DIAGNOSIS
Recurrent cutaneous squamous cell carcinoma of the right orbit involving the anterior cavernous sinus
DISCUSSION
PD-1 is a cell surface protein that sends a signal to dampen T-cell responses when activated. In cancer cells, PD-1 is often turned on with the goal of suppressing immune responses. Thus, preventing these molecules from interacting with their binding partners via antibody blockade can restore anti-tumor immune responses with significant clinical benefits.1 Pembrolizumab, a monoclonal antibody targeting PD-1, has been shown to be efficacious in treating melanoma, lung cancer, mucosal head and neck cancer, gastric cancer, urothelial cancer, and triple-negative breast cancer.1 In immunosuppressed populations, cSCC has a high mutational burden and has increased incidence. In other cancers, these attributes predict likely response to immunotherapy with a checkpoint inhibitor, prompting the investigation of PD1 inhibition as treatment for unresectable, locally advanced or metastatic cSCC.2
This patient’s experience highlights the potential for PD-1 inhibition as definitive treatment of locally advanced or metastatic cutaneous squamous cell carcinoma, an entity typically treated with local resection and radiation therapy with or without concurrent chemotherapy. Our patient’s clinical course is consistent with a previous case series that reported multiple complete and partial responses in patients with locally advanced or metastatic cSCC receiving anti-PD1 therapy.1 Of note, a phase II trial elucidated the efficacy of cemiplimab, another PD1 inhibitor, in unresectable, locally advanced and metastatic cSCC.2
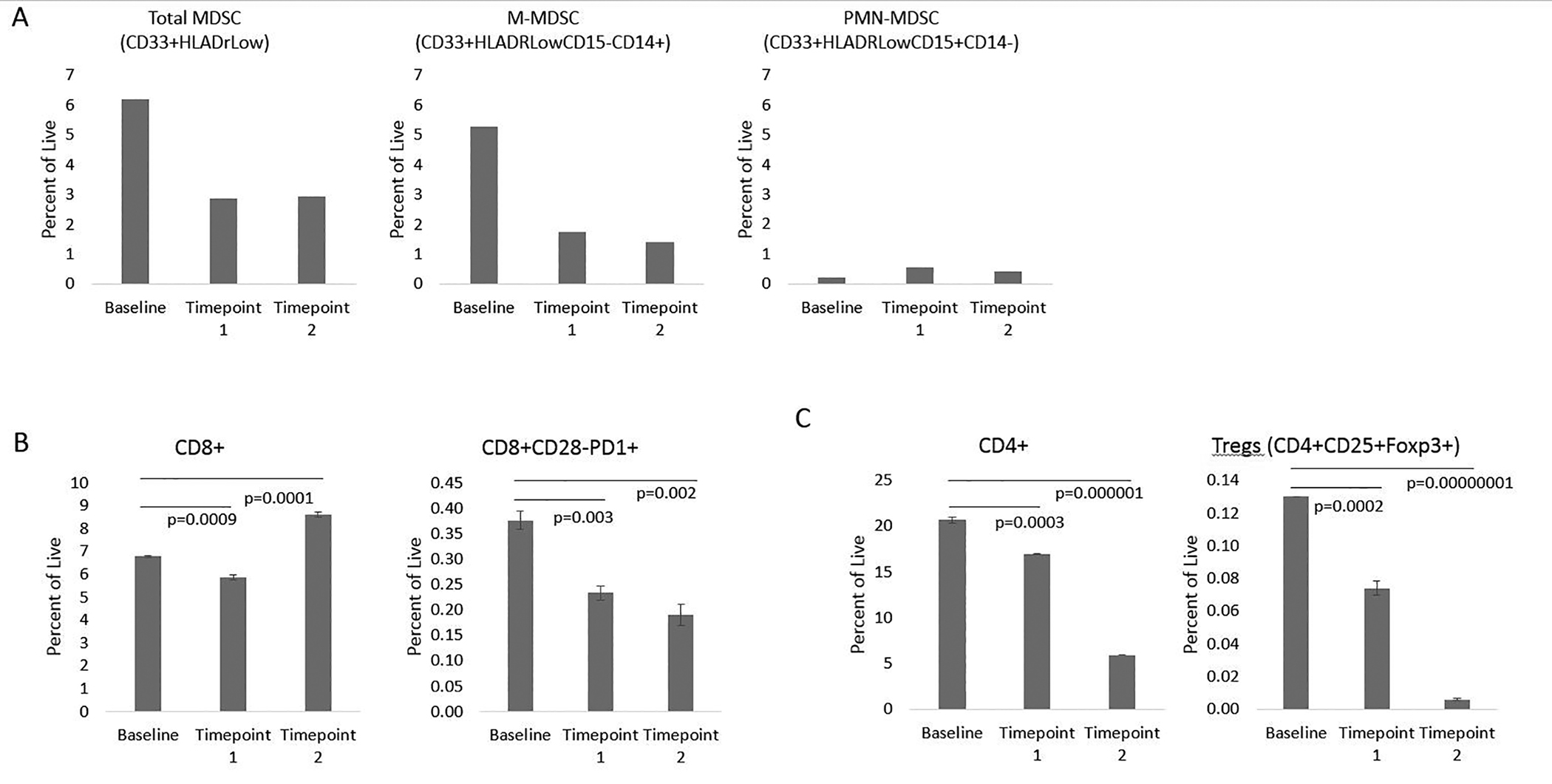
In addition, analysis of the patient’s blood was performed, as part of an IRB-approved protocol, just prior to treatment with pembrolizumab, and at 1 month and 10 months after initiation of treatment (Figure 2). A decrease in the levels of circulating myeloid-derived suppressor cells (MDSCs), particularly monocytic MDSCs was associated with response. The frequencies of circulating CD8+ T cells were slightly increased after treatment, whereas levels of circulating CD4+ T cells were significantly lower at the second time point measured. Interestingly, a significant reduction in the levels of suppressive CD8+ T cells (CD8+ CD28- PD1+) and regulatory T cells were also associated with treatment response.
The immunologic findings in this case are consistent with recent studies identifying changes in peripheral blood lymphocytes during treatment with immunotherapy. In this case, we observed a decrease in the peripheral blood levels of cell types associated with immune suppression such as MDSCs, CD8+ CD28- PD-1+ T cells, and regulatory T cells. Studies have demonstrated associations between both decreased MDSC levels and decreased regulatory T cell levels, respectively, with response to immune checkpoint inhibition in patients with melanoma.3,4 Also, CD8+ CD28+ T cells were recently shown to be critical in anti-PD-1 therapy in lung cancer patients.5 Therefore, decreased CD8+ CD28- T cell populations after treatment, which were observed in our patient’s case, may be a marker of immunotherapy sensitivity in cSCC.
CONCLUSION
This report presents a case of a patient with recurrent, poorly differentiated cutaneous squamous cell carcinoma metastatic to the right orbit, tracking posteriorly into the cavernous sinus, causing blurry vision and diplopia. The patient declined surgical intervention and instead was initiated on pembrolizumab immunotherapy. After completing his regimen, the patient had a complete response to therapy, with resolution of his visual symptoms and stable appearance of the mass on follow-up MRIs.
Analysis of the patient’s blood was performed prior to treatment, as well as at 2 time points afterward, and showed a decrease in various immune suppressive cell lines, such as circulating myeloid-derived suppressor cells, CD8+ CD28- PD-1+ T cells, and regulatory T cells. These cell lines have been associated with response to anti PD-1 therapy in melanoma and lung cancer, and as demonstrated by this case report, may be useful markers in response to immunotherapy in cutaneous squamous cell carcinoma as well.
REFERENCES
- Que SKT, Zwald FO, Schmults CD. Cutaneous squamous cell carcinoma: management of advanced and high-stage tumors. J Am Acad Dermatol. 2018;78:249-261.
- Migden MR, Rischin D, Schmults CD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. N Engl J Med. 2018;379:341-351.
- Curran MA, Montalvo W, Yagita H, et al. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci U S A. 2010;107:4275-4280.
- Weber R, Fleming V, Hu X, et al. Myeloid-derived suppressor cells hinder the anti-cancer activity of immune checkpoint inhibitors. Front Immunol. 2018;9:1310.
- Kamphorst AO, Wieland A, Nasti T, et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science. 2017;355:1423-1427.
Citation
V V, RB R, B G, CM D, J G, SA K. Complete response after treatment with pembrolizumab in a patient with metastatic cutaneous squamous cell carcinoma involving th. Appl Radiat Oncol. 2019;(4):42-44.
December 27, 2019