Combination of volumetric-modulated arc therapy (VMAT) and partially wide tangents (PWT) for improved organ sparing in a left-si
Images




Abstract
Objective: Postmastectomy radiation therapy (PMRT) to the chest wall and regional nodal irradiation (RNI) in women with node-positive breast cancer is known to reduce locoregional recurrence and distant metastases, as well as improve overall survival. Standard 3-dimensional (3D) conformal techniques may not be able to provide a clinically optimal plan for treatment, especially when coverage to the internal mammary nodes is required. Although advanced delivery techniques such as volumetric-modulated arc therapy (VMAT) can be used to better spare the heart and lungs, there is an increase in low dose exposure to normal tissue compared with 3D conformal planning. In a situation where 3D conformal planning provides an acceptable dose distribution for the majority of critical organs, it may not be justifiable to use VMAT to reduce dose to a few organs, while increasing low-dose exposure to the entire patient. In such cases, a combination of VMAT and 3D can be suitable to generate a clinically acceptable dose distribution without an unnecessary increase in low dose exposure. In this report we describe a left- and a right-sided case, which have benefited from this approach.
Methods and Materials: The left-sided case was of a 59-year-old woman with stage IIIC cancer who had undergone a bilateral mastectomy requiring PMRT to the chest wall along with RNI. The right-sided case was of a 51-year-old woman with stage IIIC cancer who had undergone a lumpectomy requiring whole-breast radiation (WBI) with RNI. Computed tomography (CT) scans were acquired from the chin to the upper abdomen during free breathing at 3-mm slice spacing with the patient positioned on an angle board and head turned to the contralateral side. Contouring of the target volume was done as per Radiation Therapy Oncology Group (RTOG) guidelines. Three plans were done for each case, the first using 3D conformal planning with the partially wide tangents (PWT) technique, the second using volumetric-modulated arc therapy (VMAT) alone, and finally using a combination of 3D and VMAT. A dosimetric comparison of the plans was performed in each case.
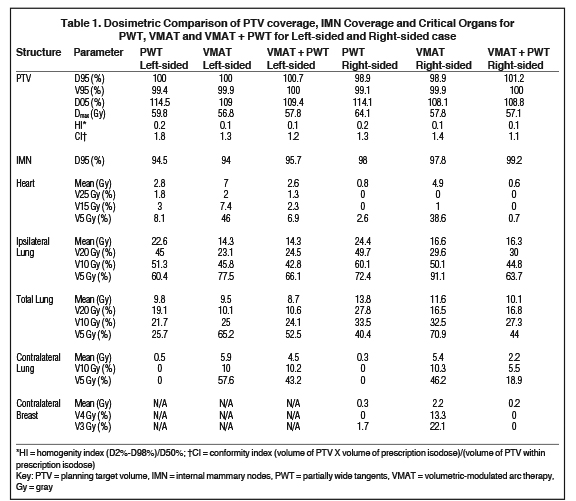
Results: For both the left- and the right-sided cases, the PTV D95, V95 as well as the IMN D95 were comparable among all the plans; however, the hotspot in terms of the PTV D05 was found to be lower for the VMAT and the combined plan. The homogeneity index (HI) and the conformity index (CI) were best with the combined plan. For both the left- and the right-sided cases, the heart was well spared with the PWT technique. The MHD was the highest with VMAT as were the volumes covered by lower doses, namely 15 Gy and 5 Gy. However, combining VMAT and PWT did not compromise the V15 Gy and V5 Gy compared with PWT. The ipsilateral lung V20 Gy was the highest (≥ 45%) with PWT technique yielding a clinically unacceptable plan in both cases. The combination of VMAT and PWT not only maintained the ipsilateral lung V20 Gy ≤ 30%, but also kept the V5 Gy comparable to or lower than the corresponding PWT plan. The trend in the result for the total lung was similar to that of the ipsilateral lung. The contralateral breast was best spared with PWT; however, the combination plan did not compromise dose to the contralateral breast compared with the former technique.
Conclusions: Combination of VMAT and 3D was not only able to preserve the mean dose to the heart, but was able to also reduce the ipsilateral lung V20 Gy without increasing low dose to all of the organs compared to using VMAT alone.
Postmastectomy radiation therapy (PMRT) to the chest wall and regional nodal irradiation (RNI) in women with node-positive breast cancer is known to reduce locoregional recurrence and distant metastases, while improving overall survival.1,2 The addition of RNI to whole-breast irradiation (WBI) after breast-conserving surgery (BCS) has also been shown to reduce the rate of breast cancer recurrence.3,4 While irradiating the chest wall and internal mammary nodes (IMNs) with 3-dimensional (3D) conformal planning, the partially wide tangents (PWT) technique has been shown to provide the most appropriate balance between target coverage and normal tissue sparing.5,6 Regardless, the requirement to irradiate the IMNs increases exposure to the heart and lungs. When standard 3D conformal planning techniques are unable to adequately reduce dose to the heart and lung, advanced treatment planning and delivery techniques such as intensity-modulated radiation therapy (IMRT) or volumetric-modulated arc therapy (VMAT) have helped improve sparing.7-9 However, since the beams/arcs irradiate the target from many projections/angles, there is an increase in low dose exposure to normal tissue compared with 3D conformal planning. This caveat has limited the widespread application of VMAT for breast cancer treatment.
Our clinic was recently presented with a left- and a right-sided case, where in using PWT, the mean heart dose (MHD) was 2.8 Gy and 0.8 Gy, respectively; however, the ipsilateral lung V20 Gy was ≥ 45% in both cases. Hence, even though the MHD was acceptable for treatment, the ipsilateral lung V20 Gy needed to be reduced to ≤ 30% for a clinically viable plan.10,11 If VMAT alone were used, it would increase the MHD and the spread of low dose to other critical organs. In this technical report, we describe how a combination of 3D conformal planning using PWT over the region of the heart and VMAT superior to the heart can help maintain the advantage of low mean heart dose attained with 3D while reducing dose to the ipsilateral lung, meeting the desirable dose constraint. Through a dosimetric comparison of PWT, VMAT and a combination of VMAT and PWT, we show that the combination of the 2 techniques is not only able to preserve the MHD, but will also reduce the ipsilateral lung V20 Gy without increasing low dose to all the organs as compared to using VMAT alone. In this manner, the advantages of both techniques can be preserved.
Methods and Materials
The left-sided case was of a 59-year-old woman with stage IIIC cancer who had undergone a bilateral mastectomy requiring PMRT to the chest wall along with RNI. The right-sided case was of a 51-year-old woman with stage IIIC cancer who had undergone a lumpectomy requiring WBI with RNI. Computed tomography (CT) scans were acquired from the chin to the upper abdomen during free breathing at 3-mm slice spacing with the patient positioned on an angle board and head turned to the contralateral side.
Target delineation
Contouring of the clinical target volume (CTV) chest wall/breast tissue, axillary level I, II, III, supraclavicular and IMNs was done per Radiation Therapy Oncology Group (RTOG) guidelines.12 The planning target volume (PTV) for the left-sided case was CTV + 5 mm and included the skin as this was post-mastectomy radiation. A 5-mm bolus was used over the chest wall area for the left-sided case to adequately cover the skin. For the right-sided case, the first 5 mm of the skin was excluded from the PTV, since the breast tissue was intact for this case. Critical organs contoured were the heart, ipsilateral lung and contralateral lung. The contralateral breast was also contoured for the right-sided case. Energy used for planning was 6 MV for the left-sided case, and a mix of 6 MV and 16 MV for the right-sided case due to a larger separation. Dose prescribed was 50 Gy in 25 fractions. Treatment planning for all plans was done on the Eclipse V 13.6 (Varian, Palo Alto, California).
Partially Wide Tangents (PWT) planning
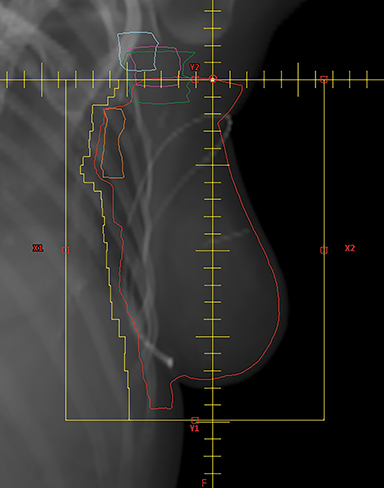
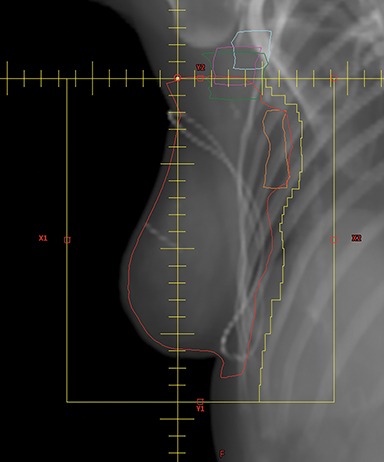
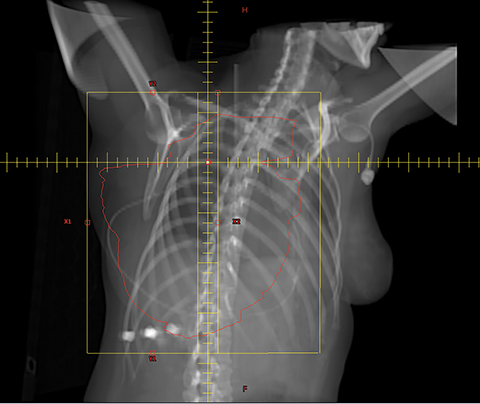
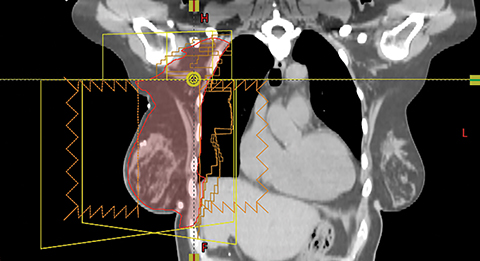
A mono-isocentric technique was used where the isocenter was placed by the clavicular head, which served as the junction/matchline of the supraclavicular field with the PWT fields. The collimator angle in this technique was deliberately kept at 0 degrees to avoid divergence of the supraclavicular field into the tangents. The angle board was adjusted to make the chest wall parallel to the posterior edge of the tangential field to minimize the amount of lung covered by these fields (Figures 1A and B). Gantry angles were adjusted for the tangential photon fields to match the divergence of the posterior field edges of the beam. The PWT fields covered the IMNs in the first 3 intercostal spaces along with some inferior portion of the axillary level I and II lymph nodes and the PTV in the beam’s eye view (BEV). The extent of the blocking was determined from the BEV of each tangential field to ensure adequate coverage of the PTV with a 7-mm margin for penumbra. Field-in-field compensation was used for planning.13
Volumetric-modulated Arc Therapy (VMAT) Planning
Planning with VMAT was per the technique described by Popescu et al.9 The projection angle at which the PTV separation is the largest in the BEV was chosen. Due to the large treatment volumes involved and the limited MLC leaf travel within an individual field, which is 15 cm on a Varian linac, treatment planning required the use of at least 2 complementary arcs to cover the extent of the PTV. These arcs overlapped at the isocenter by 2 cm and the collimator angle was kept at 0° as shown in Figure 2A. The treatment isocenter was set in the same manner as in a mono-isocentric 3-field breast technique. Since isocenter location is typically lateral to the patient’s midline, full arcs are likely to collide with the patient. In our previous study, the use of 2 complementary partial arcs within a 190-degree arc range was sufficient.14 Increasing the arc range to 220 and 240° degrees, respectively, did not improve the PTV coverage and homogeneity, and only increased the volume of the ipsilateral lung and total lung covered by doses in the range 5 Gy to 25 Gy. Increasing the number of arcs also did not improve PTV coverage, homogeneity or critical organ dose. In this study, 2 partial complementary arcs within a 200-degree range were used (Figure 2B). The optimization algorithm used was the progressive resolution optimizer (PRO) and the dose calculation algorithm was the analytical anisotropic algorithm (AAA). The VMAT plan was optimized such that priority was given to cover 95% of the IMNs with at least 90% of the prescription dose (ie, 45 Gy) or more while achieving PTV D95, V95 ≥ 95% and PTV D05 ≤ 110%, followed by mean heart dose (MHD), ipsilateral lung V20 Gy, dose to the contralateral lung and breast (for the right-sided case).
Volumetric-modulated Arc Therapy (VMAT) and Partially Wide Tangents (PWT) Planning
For both the left- and the right-sided cases, the PTV by the heart was adequately covered while maintaining acceptable MHD (desired to keep ≤ 3 Gy). However, the ipsilateral lung V20 Gy was unacceptably high at ≥ 45%. It is desirable to keep it ≤ 30% to reduce the likelihood of toxicity.10,11 VMAT planning was hence used to improve the ipsilateral lung dose, over the region superior to the heart. The PTV superior to the junction was planned with VMAT and inferior to the junction with PWT. Matching of the superior and inferior plans was accomplished with the single isocenter half-beam block technique15,16 using an asymmetric jaw. Figure 3 demonstrates the positioning of the isocenter and the matchline. In the left-sided case, the gantry start and stop angles were 160 and 320 degrees, respectively, for the VMAT plan; for the right-sided case, they were 210 and 50 degrees, respectively. To avoid hotspots at the junction while planning with VMAT, the PTV for optimization purposes (PTV-OPT) was defined 6 mm superior from the isocenter/junction. To avoid areas of underdosing/cold spots at the junction, in this study, the slices of the PTV in between the level of the isocenter and the inferior-most slice of the PTV-OPT were constrained in the optimizer to receive at least 45 Gy, limiting the hotspot to be ≤ 105%. This dose was determined iteratively such that the PTV D95 of the combined VMAT and PWT plans was ≥ 95% and the PTV D05 ≤ 110%. The collimator angle was kept at 0 degrees for the PWT and the VMAT plan to avoid beams from both plans from diverging into each other. Blocking in the PWT plan was constructed such that the PTV was covered without compromising heart sparing. The VMAT plan was optimized with the priority given to cover 95% of the IMNs with at least 90% of the prescription dose or more while achieving PTV D95, V95 ≥ 95%, and PTV D05 ≤ 110% in the combined VMAT and PWT plans. The next priority was to minimize the ipsilateral lung V20 Gy, followed by dose to the contralateral lung and breast.
VMAT plans went through patient-specific quality assurance (QA) using portal dosimetry and 2-dimensional diode array. Absolute dose comparison was performed between the calculated and measured plans.
Results
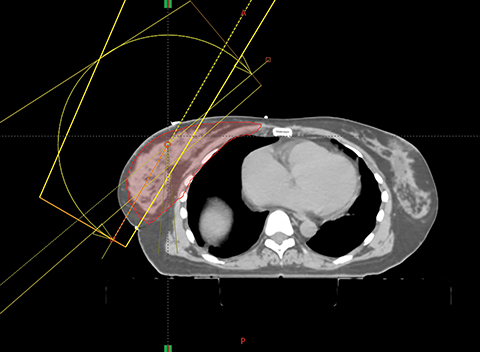
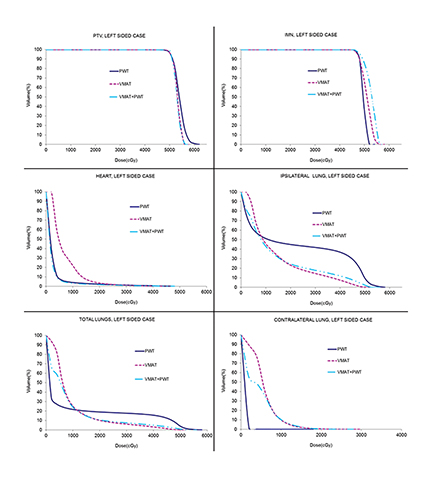
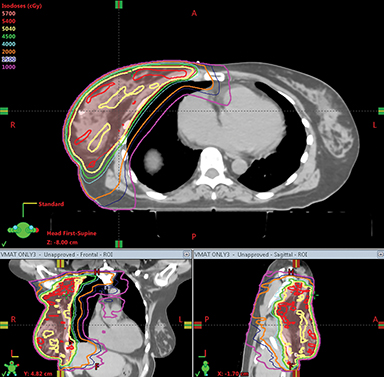
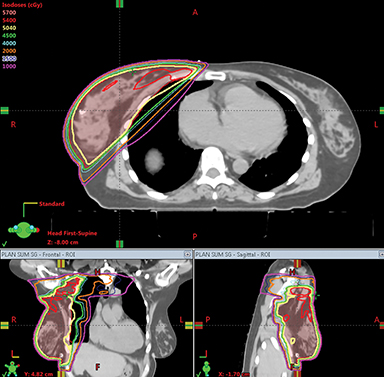
Dosimetric comparison of the various plans for the left and the right-sided cases are shown in Table 1. Dose volume histograms are shown in Figures 4A and B, while the comparison of the dose distribution for VMAT and VMAT + PWT are shown in Figure 5.
Planning Target Volume (PTV) Coverage
For both the left- and the right-sided cases, the PTV D95, V95 as well as the IMN D95 were comparable among all the plans, namely, PWT vs VMAT, vs VMAT + PWT. The hotspot in terms of the PTV D05 was the least with VMAT or the combined plan. The homogeneity index (HI) and the conformity index (CI) were best with the VMAT + PWT plan.
Heart
For both the left- and the right-sided cases, the heart was well spared with the PWT technique. If VMAT alone were to be used to cover the PTV, the MHD was increased by almost 4 Gy. By combining VMAT and PWT, the advantage of low MHD achieved with PWT was not compromised. The V25 Gy was similar among the 3 techniques. However, volumes covered by lower doses, namely 15 Gy and 5 Gy, were highest with VMAT alone as expected. Combining VMAT and PWT did not compromise the V15 Gy and V5 Gy compared with PWT.
Ipsilateral Lung
For both cases, the PWT technique yielded a clinically unacceptable plan with the ipsilateral lung V20 Gy at ≥ 45%. VMAT alone reduced this by ≥ 20%, but the cost was an increase in V5 Gy by almost 20%. The combination of VMAT and PWT not only maintained the ipsilateral lung V20 Gy ≤ 30% at values similar to that of the VMAT plan, but also kept the V5 Gy comparable to or lower than the corresponding PWT plan.
Contralateral Lung
The contralateral lung was best spared with PWT and worst with VMAT in both cases. Doses to the contralateral lung with the combined plan were in between these 2 techniques.
Total Lung
The trend in the result for the total lung was similar to that of the ipsilateral lung. In both cases, the total lung mean dose and V20 Gy were the highest with PWT. Although using VMAT helped lower these dosimetric parameters, the cost was an increase in V5 Gy. Combining the 2 techniques helped maintain the dosimetric advantage of VMAT for the mean total lung dose and V20 Gy, while reducing the V10 Gy and V5 Gy, compared to using VMAT alone.
Contralateral Breast
For the right-sided case, the contralateral breast was spared well with PWT and was the worst with VMAT as expected due to low dose bath. The combined plan did not compromise dose to the contralateral breast.
Intensity-modulated radiation therapy (IMRT) Quality Assurance (QA)
Absolute dose comparison was performed between the calculated and measured plans over all dose regions and > 99% of the points agreed to within 3% and 3 mm.
Discussion
With published results from the National Cancer Institute of Canada [NCIC] Clinical Trials Group MA.20 and European Organization for Research and Treatment of Cancer (EORTC) trials,3,4 more breast cancer patients are likely to receive RNI that includes the IMNs. Including the IMNs in the treatment volume increases exposure to normal tissue. Treatment planning techniques are needed that can adequately cover the target volume while alleviating unnecessary exposure to the critical organs. In situations where the dosimetric results are not acceptable for both the heart and the ipsilateral lung, VMAT alone would suffice.9 In this work, we have presented a left- and right-sided case in which the PWT plan met the dose constraints for the heart but not for the ipsilateral lung. Using PWT to cover the PTV in the region over the heart, and VMAT to cover the PTV superior to the heart, has helped meet the constraint for the ipsilateral lung V20 Gy without compromising heart dose or increasing the low dose as much as in the case of using VMAT alone. Studies in patients who received locoregional RT that included IMNs while maintaining the ipsilateral lung V20 Gy < 30%, showed ~6% grade 1 and 2 radiation pneumonitis (RP) and no incidence of grade 3 and 4 RP.10,11,17 In patients for whom the V20 Gy was around 35%, the grade 1 and 2 complication rates had risen to 23% and 11.5%, respectively. These studies emphasize the importance of adhering to the constraint of the ipsilateral lung V20 Gy < 30%. In both cases reported here, we were not able to keep the ipsilateral lung V20 Gy < 30% using the PWT technique alone. However, the combination of VMAT and PWT met this constraint for both cases, while limiting the low dose.
Exposure of the heart during radiation therapy is unavoidable, especially for left-sided cases, which increases the likelihood of developing ischemic heart disease in the long term. The rates of major coronary events increase linearly with the mean dose to the heart.18 Using VMAT alone to treat the PTV unnecessarily increased exposure to the heart, thus increasing the mean heart dose and low dose to unspecified normal tissue, both of which were lower with PWT. The influence of low dose on heart disease, specifically the volume of the heart covered by 1 to 2 Gy with radiation therapy, has been investigated in the literature.19 No correlation has been found between low dose and cardiac function or perfusion defects, or worsening of these defects within a short-term follow-up (1 year after exposure). The study concluded that with average MHD < 5 Gy (1.1 to 6.1 Gy), no clinically significant defects were found after radiation therapy. Due to the absence of long-term follow-up data with respect to low dose to the heart, for patients treated with VMAT, it is optimal to keep volumes of the heart covered within all dose ranges as low as possible while maintaining adequate coverage to the target. This caveat of increased low dose prevented the use of VMAT alone for both cases, making the combination of VMAT and PWT the preferred choice.
Dose to the contralateral lung with this combination approach, however, was increased compared to PWT. Although it was found to be less compared with VMAT, long-term follow-up again unavailable, the impact of low dose to the contralateral lung on the incidence of RP is currently unclear, although rates of grade 3 or higher RP have not been observed, even when the V5 Gy to the ipsilateral lung was 100%.20
Using this combination technique also increases low dose exposure to other normal tissue; however, the exposure is less compared with using VMAT alone to cover the entire PTV. The primary aim of this combination was to reduce the ipsilateral lung V20 Gy, as studies have shown that its value of > 30% is associated with increased rates of short-term radiation pneumonitis and change in short-term pulmonary function. For the right-sided case, increased exposure from the VMAT portion of the combination plan to the contralateral breast was not a concern because the PTV by that region was covered using PWT. Although the left-sided case had undergone a bilateral mastectomy, again, increased exposure from the VMAT portion of the plan to this region was not a concern for the same reason.
Both patients were treated with free breathing as they were not capable of deep inspiration breath hold. In addition to the standard PWT fields, there were 2 partial arcs to treat, each of which took 67 seconds. Hence, the treatment time required was a little over 2 minutes more than standard treatment.
Conclusion
In this technical report, we have presented a unique situation of a left- and right-sided case whereby the MHD was acceptable for treatment; however, the ipsilateral lung V20 Gy had exceeded the required dosimetric tolerance. A combination of VMAT and PWT was not only able to meet constraints for the ipsilateral lung, but did so without compromising doses to other critical organs such as the heart. This was achievable with the PWT technique and without increasing unnecessary exposure to all other organs compared to VMAT alone.
References
- Clarke M, Collins R, Darby S, et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;366:2087-2106.
- EBCTCG (Early Breast Cancer Trialists’ Collaborative Group). Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet. 2014;383(9935):2127-2135.
- Whelan TJ, Olivotto IA, Parulekar WR, et al. Regional nodal irradiation in early-stage breast cancer. N Engl J Med. 2015;373(4):307-316.
- Poortmans PM, Collette S, Kirkove C, et al. Internal mammary and medial supraclavicular irradiation in breast cancer. N Engl J Med. 2015;373(4):
- 317-327.
- Marks LB, Hebert ME, Bentel, G et al. To treat or not to treat the internal mammary nodes: a possible compromise. Int J Radiat Oncol Biol Phys. 1994;29(4):903-909.
- Pierce LJ, Butler JB, Martel MK, et al. Postmastectomy radiotherapy of the chest wall: Dosimetric comparison of common techniques. Int J Radiat Oncol Biol Phys. 2002;52(5):1220-1230.
- Beckham WA, Popescu CC, Patenaude VV, et al. Is multibeam IMRT better than standard treatment for patients with left-sided breast cancer? Int J Radiat Oncol Biol Phys. 2007;69(3):918-924.
- Popescu CC, Olivotto IA, Patenaude VV, et al. Inverse-planned, dynamic, multi-beam, intensity-modulated radiation therapy (IMRT): A promising technique when target volume is the left breast and internal mammary lymph nodes. Med Dosim. 2006;31:283-291.
- Popescu CC, Olivotto IA, Beckham WA, et al. Volumetric modulated arc therapy improves dosimetry and reduces treatment time compared to conventional intensity-modulated radiotherapy for locoregional radiotherapy of left-sided breast cancer and internal mammary nodes. Int J Radiat Oncol Biol Phys. 2010;76(1):287-295.
- Goldman UB, Anderson M, Wennberg B et al. Radiation pneumonitis and pulmonary function with lung dose-volume constraints in breast cancer irradiation. Journal of Radiotherapy in Practice 2014;13:211-217.
- Lind PA, Wennberg B, Gagliardi G, et al. Pulmonary complications following different radiotherapy techniques for breast cancer, and the association to irradiated lung volume and dose. Breast Cancer Res Treat. 2001;68:199-210.
- Radiation Therapy Oncology Group. RTOG Breast Cancer Contouring Atlas. https://www.rtog.org/CoreLab/ContouringAtlases/BreastCancerAtlas.aspx Accessed September 13, 2017.
- Kestin LL, Sharpe MB, Frazier RC, et al. Intensity modulation to improve dose uniformity with tangential breast radiotherapy: Initial clinical experience. Int J Radiat Oncol Biol Phys. 2000;48(5):1559-1568.
- Dumane VA, Hunt MA, Green S, et al. Dosimetric comparison of volumetric modulated arc therapy, static field intensity modulated radiation therapy, and 3D conformal planning for treatment of a right-sided reconstructed chest wall and regional nodal case. J Radiother. 2014;1-12.
- Li JG, Liu C, Kim S, et al. Matching IMRT fields with static photon field in treatment of head-and-neck cancer. Med Dosim. 2005;30(3):135-138.
- Amdur RJ, Liu C, Li J, et al. Matching intensity-modulated radiation therapy to an anterior low neck field. Int J Radiat Oncol Biol Phys. 2007;69(2):S46-S48.
- Rothwell RI, Kelly SA, Joslin CA, et al. Radiation pneumonitis in patients treated for breast cancer. Radiother Oncol. 1985;4(1):9-14.
- Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. New Engl J Med. 2013;368(11):987-998.
- Chung E, Corbett JR, Moran JM, et al. Is there a dose-response relationship for heart disease with low-dose radiation therapy? Int J Radiat Oncol Biol Phys. 2013;85(4):959-964.
- Ho AY, Ballangrud AM, Li G, et al. Pneumonitis rates following comprehensive nodal irradiation in breast cancer patients: results of a phase I feasibility trial of intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys. 2013;87(2):S48-S49.
Citation
VA D, YC L, S G. Combination of volumetric-modulated arc therapy (VMAT) and partially wide tangents (PWT) for improved organ sparing in a left-si. Appl Radiat Oncol. 2018;(2):31-38.
June 19, 2018