Chemoradiation treatment of glioblastoma multiforme: Treatment guidelines and considerations
Images


SA-CME credits are available for this article here.
Glioblastoma (GBM) is the most common primary malignant neoplasm of the brain, with an incidence of 3.19 per 100,000 persons in the US.1 Standard of care includes maximal surgical resection and radiation therapy (RT) with concomitant temozolomide (TMZ) chemotherapy. The median 3-year survival rate for a newly diagnosed patient with this aggressive cancer remains a dismal 10.1%.2 Nevertheless, recent advancements in the use of alternating electric field therapy, also known as tumor-treating fields (TTFields), and dendritic cell vaccines are beginning to challenge the status quo with initial results yielding a median overall survival of 20.9 months.3,4 Moreover, molecular characterization of primary brain tumors has had a substantial impact on the stratification of central nervous system (CNS) neoplasms. This includes a more nuanced characterization of GBM molecular markers, thus leading to the creation of an integrated diagnosis.5 In this review, we highlight the North American and European guidelines for chemoradiation of GBM created as a result of the new 2016 World Health Organization (WHO) classification system. Specifically, we focus on the factors of age, performance status, molecular markers, and disease recurrence as the main components for the clinical application of the guidelines. Furthermore, we highlight factors, such as socioeconomic and insurance status, that impact radiation treatment compliance and GBM outcomes.
Standard of Care
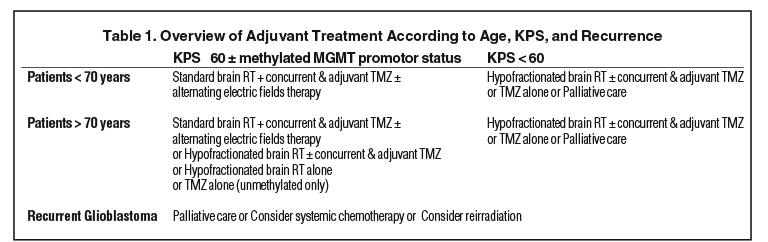
Therapy for GBM is divided into multiple strata of treatment modalities including surgery, radiation and chemotherapy. Tumor molecular markers may be used as a guiding prognostic factor to optimize a personalized treatment plan.6,7 These molecular features confer a survival advantage in GBM, as they predict a favorable treatment response. Markers screened for after a histologic diagnosis of GBM may include: O6-methylguanine DNA methyltransferase (MGMT) promoter methylation status and isocitrate dehydrogenase (IDH) mutation.7 In this article, however, we will focus on MGMT promoter methylation status. Furthermore, tumor resectability, Karnofsky Performance Score (KPS), and patient age are important components of the clinical-care decision-making process (Table 1).8
Patients Age < 70
For patients age < 70 years and a KPS ≥ 60, guidelines recommend maximal surgical resection followed by adjuvant therapy.7-11 The type of adjuvant treatment is dictated by postresection KPS and MGMT promoter status. For patients < 70, postresection KPS ≥ 60, and methylated MGMT promoter status, guidelines recommend standard brain RT, concurrent plus adjuvant temozolomide (TMZ), and TTFields.7 Recommendations remain the same for patients with the same age and KPS bracket but an unmethylated/indeterminate MGMT promoter.7 However, standard brain RT alone is an option for this second group. According to the American Society for Radiation Oncology (ASTRO) guidelines, standard brain RT entails partial-brain RT of 60 Gy in 2-Gy fractions (30 total fractions) delivered throughout 6 weeks.8 Similarly, the European Association for Neuro-Oncology (EANO) guidelines recommend focal RT of 50-60 Gy in 1.8-2.0 Gy fractions following surgical resection or biopsy in patients < 70 years of age and a KPS ≥ 70. Gross total resection has been shown to improve outcomes and is therefore recommended for clinically eligible patients.6 Both ASTRO and EANO guidelines recommend targeted delivery of radiation against whole-brain therapy to minimize toxicity to structures such as the optic nerves, optic chiasm, retinas, brainstem, pituitary, cochlea, hippocampus and other sensitive structures.6-8
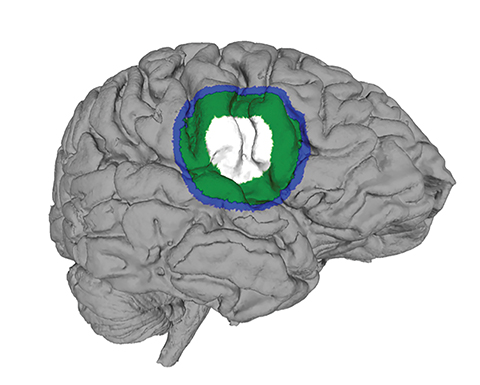
Determining the tumor volumes is an important consideration when conducting partial-brain RT in GBM patients (Figure 1). The gross tumor volume (GTV) includes the surgical bed and any area of postsurgical or postbiopsy T1 MRI enhancement. The clinical target volume (CTV) is defined as the GTV and any residual T2W or fluid-attenuated inversion recovery (FLAIR) signal abnormalities plus an additional margin of 1 to 2.5 cm. Finally, the planning target volume (PTV) of 0.3 cm to 0.5 cm is added onto the CTV to account for daily setup error.6 Radiation may be administered using 1- or 2-phase radiation target volume strategies. The 1-phase target volume approach encompasses the CTV and margin without targeting edema.8 In contrast, the 2-phase target volume method includes the CTV, margin, and edema measured using hyperintense T2 and FLAIR MRI regions as a guide. This is subsequently narrowed down to target only the gross residual tumor and resection cavity in the second phase.8
In addition to surgery and radiation, chemotherapeutic agents are the mainstay of treatment. Concurrent and adjuvant TMZ is recommended as its addition to radiation in the treatment of newly diagnosed GBM has been shown to provide a survival benefit.12,13 A study by Ballhausen et al demonstrated improved survival with daily concurrent TMZ administration (15.7 months) during radiation treatment compared to TMZ administration for 5 out of 7 days (12.6 months).14 Currently, the Radiation Therapy Oncology Group (RTOG) / NRG, National Comprehensive Cancer Network (NCCN), EANO, and the Medical Oncology Spanish Society (SEOM) guidelines recommend 75mg/m2 daily concurrent TMZ throughout radiation treatment followed by maintenance therapy of 150-200mg/m2 for 5 days every 4 weeks for 6 cycles.6
Finally, the inclusion of TTFields as part of the treatment plan is improving the overall survival of patients with GBM. This noninvasive antimitotic therapy consists of low intensity, 200 kHz frequency, and alternating electrical currents delivered via 2 transducer arrays on a shaved scalp (Figure 2). The device is worn ≥ 18 hours/day on the same days as administrations of TMZ.3 When used at a monthly compliance of > 90% TTFields have resulted in a statistically significant improvement in the median overall survival to 24.9 months.15 TTFields, however, are an option only for patients with supratentorial disease.7
Patients Age > 70
For patients > 70 years old, performance status and MGMT status are important considerations when choosing the treatment regimen with the utmost benefit in survival and quality of life. Treatment remains controversial and attempts are underway to understand the role of TMZ and hypofractionated RT in the elderly, especially in those with unmethylated MGMT promoters and those with poor performance status.16 Nonetheless, there is surmounting evidence that RT in those >70 improves survival when compared to supportive care alone with similar quality of life and cognitive evaluations between groups.17 In addition, hypofractionated RT has been shown to reduce steroid utilization and decrease early RT termination when compared with standard-length RT.18
According to EANO guidelines, treatment decisions should be based on MGMT status for patients >70 who are not eligible for radiation with concurrent or maintenance TMZ. Patients with MGMT methylation status are recommended to receive TMZ alone for 5 consecutive days every 28 days. Patients with non-MGMT/indeterminate methylation status should undergo hypofractionated RT of 40 Gy in 15 fractions using a similar T2W abnormality plus a 2-cm margin for planning tumor volumes.6 ASTRO guidelines recommend a similar radiation schedule (40 Gy in 2.66 fractions) in addition to TMZ in elderly patients with good to reasonable performance status.8 In contrast, SEOM recommends treatment with TMZ alone for patients with poor performance status and MGMT methylation.19 In addition, the Nordic randomized phase III clinical trial found similar median survival when comparing TMZ alone to hypofractionated RT (34 Gy in 10 fractions) in patients > 60 years.20 Results from Perry et al on elderly patients with GBM, however, have led to the consideration of short-course RT (40 Gy in 15 fractions) plus TMZ as standard of care.21 CTV determinations for patients > 70 receiving hypofractionated RT should be made as described above for patients < 70 years old.6
In elderly patients with GBM, a KPS > 70, and MGMT promotor methylation, a study by Palmer et al reported that 49% of physicians surveyed recommended a standard course of radiation and chemotherapy while 39% recommended a short course of radiation and chemotherapy.22 In elderly patients with KPS > 70 and non-MGMT methylation status, 51% of physicians recommended a short course of radiation alone. In patients with KPS < 50, 57% of physicians recommended supportive care. Although more studies are needed to elucidate optimal treatments in elderly patients with GBM, evidence suggests improved outcomes with use of hypofractionated RT and TMZ. A clinical trial by Perry et al showed improved median overall survival and median progression-free survival in elderly patients age > 65 who received hypofractionated RT (40 Gy in 15 fractions) and TMZ compared with those who received hypofractionated RT alone.21
Finally, despite advantages seen in patients < 70 years, and calls from the medical community and several medical governing bodies worldwide, the EANO and ASTRO have yet to include the use of TTFields in this patient cohort. However, 1 randomized clinical trial demonstrated a survival benefit in patients > 70 with a good performance status (KPS ≥ 70).3
Recurrent Glioblastoma
Unfortunately, most patients experience GBM recurrence despite maximal surgical resection, radiation and chemotherapy. Typically, recurrence of GBM occurs locally, most commonly within approximately 2 cm of the surgical resection cavity.23,24 One study found a median progression-free survival of 7 months after local tumor recurrence.24 The median overall survival rate after diagnosis of recurrence is still an estimated 22-44 weeks.25 Nonetheless, maximal safe surgical resection can be done in clinically eligible patients; however, no consensus exists regarding maximal safe resection or dosage or type of chemoradiation therapy for tumor recurrence; the treatment plan remains the choice of the physician and patient.23,24 To date, Scoccianti et al provides the most comprehensive effort to create a treatment protocol for recurrent GBM combining various approaches used in the US and Europe.26 The results of their retrospective analysis suggest that radiation-only therapy as a salvage treatment has the likelihood of a relatively good outcome.26 Patients are stratified according to the CTV of the recurrent neoplasm. Moreover, to minimize neurotoxicity patients should be treated using different fractionation and differentiated total dose in 2 Gy fractions. If the CTV is < 12.5 ml, then < 65 Gy with radiosurgery should be administered; if > 12.5 ml and < 35 ml, then <50 Gy with hypofractionated stereotactic RT should be administered; and if > 35 ml and < 50 ml, then < 36 Gy with conventionally fractionated RT should be administered.26 Furthermore, newer technologies such as proton beam therapy may be a promising modality given its role in many skull base tumors and pediatric cancers; however, it has not yet established itself in the treatment of GBM. More research will be required to determine whether protons and other heavy particles offer an advantage in GBM dosimetry.27
In the US, several options are available to the patient. First, if the postresection KPS > 60, systemic chemotherapy can be considered. Recommended regimens include TMZ, bevacizumab, lomustine/carmustine, procarbazine, and/or vincristine. If there has been a long time between stopping TMZ and tumor progression, it is reasonable to restart the patient on TMZ—especially if the tumor is MGMT methylated.28 Similarly, lomustine/carmustine is a reasonable second-line therapy for a tumor that is MGMT methylated.29 Next, although bevacizumab has not demonstrated improved overall survival in recurrent GBM, it is still FDA-approved based on improved performance status.30,31 Furthermore, evidence from the EF-11 randomized phase III clinical trial indicates the equivalence of chemotherapy and TTFields in treatment of recurrent GBM. TTFields were found comparable to chemotherapy in median survival and progression-free survival with improved quality of life seen in the TTFields cohort.32
Radiation Treatment and Social Determinants of Health
In recent years, social determinants of health—the circumstances in which people are born, grow up, live, work and age, and the systems put in place to deal with illness—have increasingly become a topic of research in the treatment of GBM.33 One influential factor driving this exploration is the ever-rising cost of US healthcare, especially in neuro-oncology. As the use of TMZ, trial-combined chemotherapy (such as TMZ and bevacizumab), and TTFields has increased, so has the overall cost of the treatment course for newly diagnosed GBM. A recently published analysis evaluating the direct medical costs of GBM found that the mean total cumulative costs per patient from 3 months prediagnosis to 12 months, and to 5 years post diagnosis were $201,749 and $268,031, respectively.34 Broken down further, the average per-patient per-month post-GBM diagnosis was $7,394.34 Given these substantial costs, there is little surprise that the standard-of-care treatment course can be deemed cost-prohibitive. Rhome et al found that compliance with chemotherapy treatment was associated with male gender, white race, younger age (< 50 years), higher performance status (> 70), insurance status, higher income/education, and receipt of treatment at an academic center.35 This can have an overwhelming negative effect on overall patient survival, especially when compliance with treatment such as TTFields is closely linked with overall rates of survival.15
Unfortunately, supporting evidence in this matter is only beginning to be discovered, despite the use of surgery, chemotherapy, and radiation in the treatment of GBM for more than 20 years. For example, a recent national survey of NCCN panel members showed that neither sexual orientation nor gender identity, which are part of social determinants of health, were thought to be relevant to the focus of the NCCN guidelines.36 Moreover, 77% responded that their panels currently do not address LGBTQ issues, with no plans to address them in the future.36
Furthermore, socioeconomic status, which encompasses education, income, and occupation, has been shown to impact time to radiation treatment.37 One study by Pollom et al showed that in patients who underwent gross total tumor resection, those who received radiation within 15 to 21 days had a statistically significant improved survival with a trend in improved survival in those receiving treatment within 22 to 35 days.36 The study found that patients who had Medicaid, government insurance, were uninsured, or lived in metropolitan areas were less likely to receive radiation within 35 days compared to patients from higher income areas. Other studies have shown the impact of insurance on radiation treatment. A study by Brown et al demonstrated a significant association between insurance type and odds of receiving radiation treatment. Patients with Medicare had the highest odds of receiving radiation, Medicaid patients had lower odds, and uninsured patients had the lowest odds.38 Lastly, a study by Chandra et al showed that uninsured patients had significantly lower rates of radiation and TMZ treatment.39
Conclusion
In this review, we provide a simple overview of the current state of radiation use for treatment of GBM. Some of the most important prognostic factors and guiding principles are based on age, performance status, and tumor molecular markers. Conventionally fractionated stereotactic RT for patients < 70 years old yields the best results for progression-free survival. Hypofractionated stereotactic RT for patients > 70 years old can also be considered for improved progression-free survival. Recent studies have elucidated the benefit of newer treatment modalities such as TTFields and their significant benefit in progression-free and overall survival. Lastly, recent literature has demonstrated the impact of socioeconomic status and insurance status on radiation treatment after GBM surgical resection.
References
- Tamimi AF, Juweid M. Epidemiology and outcome of glioblastoma. In: De Vleeschouwer S, ed, Glioblastoma. Brisbane, Australia: Codon Publications; 2017. doi:10.15586/CODON.GLIOBLASTOMA.2017.CH8
- Ostrom QT, Gittleman H, Liao P, et al. CBTRUS Statistical Report: Primary brain and other central nervous system tumors diagnosed in the United States in 2010-2014. Neuro Oncol. 2017;19(suppl_5):v1-v88. doi:10.1093/neuonc/nox158
- Stupp R, Taillibert S, Kanner A, et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. JAMA. 2017;318(23):2306-2316. doi:10.1001/jama.2017.18718
- Liau LM, Ashkan K, Tran DD, et al. First results on survival from a large phase 3 clinical trial of an autologous dendritic cell vaccine in newly diagnosed glioblastoma. J Transl Med. 2018;16(1):142. doi:10.1186/s12967-018-1507-6
- Gupta A, Dwivedi T. A simplified overview of World Health Organization classification update of central nervous system tumors 2016. J Neurosci Rural Pract. 2017;8(4):629. doi:10.4103/jnrp.jnrp_168_17
- Weller M, van den Bent M, Tonn JC, et al. European Association for Neuro-Oncology (EANO) guideline on the diagnosis and treatment of adult astrocytic and oligodendroglial gliomas. Lancet Oncol. 2017;18(6):e315-e329. doi:10.1016/S1470-2045(17)30194-8
- Nabors LB, Portnow J, Ammirati M, et al. NCCN Guidelines Insights: Central Nervous System Cancers, Version 1.2017. J Natl Compr Cancer Netw. 2017;15(11).
- Sulman EP, Ismaila N, Armstrong TS, et al. Radiation therapy for glioblastoma: American Society of Clinical Oncology clinical practice guideline endorsement of the American Society for Radiation Oncology guideline. J Clin Oncol. 2017. doi:10.1200/JCO.2016.70.7562
- Ghose A, Lim G, Husain S. Treatment for glioblastoma multiforme: current guidelines and Canadian practice. Curr Oncol. 2010. doi:10.3747/co.v17i6.574
- Marko NF, Weil RJ, Schroeder JL, Lang FF, Suki D, Sawaya RE. Extent of resection of glioblastoma revisited: personalized survival modeling facilitates more accurate survival prediction and supports a maximum-safe-resection approach to surgery. J Clin Oncol. 2014;32(8):774-782. doi:10.1200/JCO.2013.51.8886
- Lacroix M, Toms SA. Maximum safe resection of glioblastoma multiforme. J Clin Oncol. 2014;32(8):727-728. doi:10.1200/jco.2013. 53.2788
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. Supplemental information. N Engl J Med. 2005;352(10):987-996. doi:10.1056/NEJMoa043330
- Stupp R, Hegi ME, Mason WP, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10(5):459-466. doi:10.1016/S1470-2045(09)70025-7
- Ballhausen H, Belka C, Niyazi M, Schupp G, Nachbichler SB. Temozolomide during radiotherapy of glioblastoma multiformeTemozolomid zur Strahlentherapie von Glioblastoma multiforme. Strahlentherapie und Onkol. 2017;193(11):890-896. doi:10.1007/s00066-017-1110-4
- Toms SA, Kim CY, Nicholas G, Ram Z. Increased compliance with tumor treating fields therapy is prognostic for improved survival in the treatment of glioblastoma: a subgroup analysis of the EF-14 phase III trial. J Neurooncol. 2019;141(2):467-473. doi:10.1007/s11060-018-03057-z
- Guedes de Castro D, Matiello J, Roa W, et al. Survival outcomes with short-course radiation therapy in elderly patients with glioblastoma: data from a randomized phase 3 trial. Int J Radiat Oncol Biol Phys. 2017;98(4):931-938. doi:10.1016/j.ijrobp.2017.03.037
- Keime-Guibert F, Chinot O, Taillandier L, et al. Radiotherapy for glioblastoma in the elderly. N Engl J Med. 2007;356(15):1527-1535. doi:10.1056/NEJMoa065901
- Roa W, Brasher PMA, Bauman G, et al. Abbreviated course of radiation therapy in older patients with glioblastoma multiforme: a prospective randomized clinical trial. J Clin Oncol. 2004;22(9):1583-1588. doi:10.1200/JCO.2004.06.082
- Martínez-Garcia M, Álvarez-Linera J, Carrato C, et al. SEOM clinical guidelines for diagnosis and treatment of glioblastoma (2017). Clin Transl Oncol. 2018;20(1):22-28. doi:10.1007/s12094-017-1763-6
- Malmström A, Grønberg BH, Marosi C, et al. Temozolomide versus standard 6-week radiotherapy versus hypofractionated radiotherapy in patients older than 60 years with glioblastoma: the Nordic randomised, phase 3 trial. Lancet Oncol. 2012;13(9):916-926. doi:10.1016/S1470-2045(12)70265-6
- Perry JR, Laperriere N, O’Callaghan CJ, et al. Short-course radiation plus temozolomide in elderly patients with glioblastoma. N Engl J Med. 2017;376(11):1027-1037. doi:10.1056/NEJMoa1611977
- Palmer JD, Bhamidipati D, Mehta M, et al. Treatment recommendations for elderly patients with newly diagnosed glioblastoma lack worldwide consensus. J Neurooncol. 2018;140(2):421-426. doi:10.1007/s11060-018-2969-3
- Rapp M, Baernreuther J, Turowski B, Steiger H-J, Sabel M, Kamp MA. Recurrence pattern analysis of primary glioblastoma. World Neurosurg. 2017;103:733-740. doi:10.1016/j.wneu.2017.04.053
- Straube C, Elpula G, Gempt J, et al. Re-irradiation after gross total resection of recurrent glioblastoma. Strahlentherapie und Onkol. 2017;193(11):897-909. doi:10.1007/s00066-017-1161-6
- Clarke JL, Ennis MM, Yung WKA, et al. Is surgery at progression a prognostic marker for improved 6-month progression-free survival or overall survival for patients with recurrent glioblastoma? Neuro Oncol. 2011;13(10):1118-1124. doi:10.1093/neuonc/nor110.
- Scoccianti S, Francolini G, Carta GA, et al. Re-irradiation as salvage treatment in recurrent glioblastoma: A comprehensive literature review to provide practical answers to frequently asked questions. Crit Rev Oncol Hematol. 2018;126:80-91. doi:10.1016/j.critrevonc.2018.03.024.
- Mizumoto M, Okumura T, Ishikawa E, et al. Reirradiation for recurrent malignant brain tumor with radiotherapy or proton beam therapy. Strahlentherapie und Onkol. 2013. doi:10.1007/s00066-013-0390-6.
- Perry JR, Rizek P, Cashman R, Morrison M, Morrison T. Temozolomide rechallenge in recurrent malignant glioma by using a continuous temozolomide schedule. Cancer. 2008;113(8):2152-2157. doi:10.1002/cncr.23813
- Reithmeier T, Graf E, Piroth T, Trippel M, Pinsker MO, Nikkhah G. BCNU for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors. BMC Cancer. 2010;10(1):30. doi:10.1186/1471-2407-10-30
- Wick W, Weller M, van den Bent M, Stupp R. Bevacizumab and recurrent malignant gliomas: a European perspective. J Clin Oncol. 2010;28(12):e188-e189. doi:10.1200/JCO.2009. 26.9027
- Gilbert MR, Dignam JJ, Armstrong TS, et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N Engl J Med. 2014;370(8):699-708. doi:10.1056/NEJMoa1308573
- Stupp R, Wong ET, Kanner AA, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: A randomised phase III trial of a novel treatment modality. Eur J Cancer. 2012;48(14):2192-2202. doi:10.1016/j.ejca.2012.04.011
- World Health Organization. Commission on Social Determinants of Health Final Report. https://apps.who.int/iris/bitstream/handle/10665/43943/9789241563703_eng.pdf;jsessionid=D2EBD07109299FE8553B7C7B50CB2B48?sequence=1. Accessed February 27, 2019.
- Jiang S, Hill K, Patel D, et al. Direct medical costs of treatment in newly-diagnosed high-grade glioma among commercially insured US patients. J Med Econ. 2017;20(12):1237-1243. doi:10.1080/13696998.2017.1364258
- Rhome R, Fisher R, Hormigo A, Parikh RR. Disparities in receipt of modern concurrent chemoradiotherapy in glioblastoma. J Neurooncol. 2016;128(2):241-250. doi:10.1007/s11060-016-2101-5
- Hudson J, Schabath MB, Sanchez J, et al. Sexual and gender minority issues across NCCN guidelines: results from a national survey. J Natl Compr Cancer Netw. 2017;15(11)1379-1382.
- Pollom EL, Fujimoto DK, Han SS, Harris JP, Tharin SA, Soltys SG. Newly diagnosed glioblastoma: Adverse socioeconomic factors correlate with delay in radiotherapy initiation and worse overall survival. J Radiat Res. 2018;59:i11-i18. doi:10.1093/jrr/rrx103
- Brown DA, Himes BT, Kerezoudis P, et al. Insurance correlates with improved access to care and outcome among glioblastoma patients. Neuro Oncol. 2018;20(10):1374-1382. doi:10.1093/neuonc/noy102
- Chandra A, Rick JW, Dalle Ore C, et al. Disparities in health care determine prognosis in newly diagnosed glioblastoma. Neurosurg Focus. 2018;44(6):E16. doi:10.3171/2018.3. FOCUS1852
Citation
KM RP, OG V, SA T. Chemoradiation treatment of glioblastoma multiforme: Treatment guidelines and considerations. Appl Radiat Oncol. 2018;(3):24-28.
September 4, 2019