Cardiotoxicity and Radiation Therapy: A Review of Clinical Impact in Breast and Thoracic Malignancies
Images
SA-CME credits are available for this article here.
Cardiotoxicity can be an unfortunate side effect from cancer therapies including chemotherapy, hormonal therapy, and radiation therapy (RT). Subacute cardiotoxicity can occur during systemic therapies but is often considered a late effect from RT. Several different clinical conditions can result from radiation-induced cardiotoxicity (RIC): cardiomyopathy, myocarditis, pericarditis, acute coronary syndrome, congestive heart failure, and valvular disease. Cardiac injury remains multifactorial, however, with some patients receiving radiation dose to the heart and never experiencing a resultant clinical condition while others can be severely affected and even die. Data have shown that the existence of heart conditions (hypertension, diabetes, prior myocardial infarction, etc.) prior to therapy can result in a synergistic effect of cardiac injury.1 In addition, receiving systemic therapy agents during or in close chronologic proximity to RT also can have a synergistic effect.2-5 To date, no “protective” agent, except for decreased radiation dose, has been identified to minimize risk from RT.
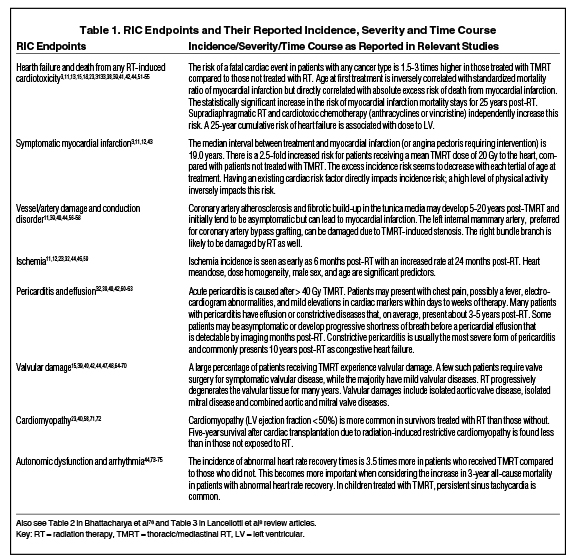
A variety of imaging modalities are available to assess cardiac function, including multigated acquisition scans, single-photon emission computed tomography, echocardiography (and derivatives thereof), cardiac magnetic resonance imaging, and invasive procedures such as cardiac catheterization. Other cardiac imaging assessments can also be performed, such as CT angiography (CTA) and assessment of coronary calcifications; however, these do no assess cardiac function. To date the “best” modality has not been determined; each modality has strengths and weaknesses, and costs vary widely, as described in detail by several publications.6,7 Table 4 in Lancellotti et al’s review article gives a thorough and concise summary of imaging techniques for RIC diagnosis.8 Additional work is needed to develop a standard method of assessing RIC. The pathophysiology of RIC is primarily associated with fibrosis and chronic inflammation. The mechanisms of action as currently understood have been described in previous publications. What remains lacking are models that can integrate the role of other medical comorbidities (hypertension, diabetes mellitus, hyperlipidemia, etc.) with cardiotoxic systemic effects.9,10 Table 1
describes several clinical conditions associated with RIC and the incidence as described in the literature.
The focus of this manuscript is to review the current literature regarding the clinical impact of RIC in the setting of breast cancers and thoracic malignancies including lung cancer, esophageal cancer and mediastinal lymphomas.
Impact by Disease Site
Breast Cancers
Three large cohort studies have shown a correlation between increased radiation dose to the heart and incidence of cardiac morbidity for women treated for breast cancer. The first study, by Darby et al, was published in 2013 and showed a linear 7.4% increased incidence of major coronary events per gray of radiation to the mean heart.11 This study was a population-based, case-control study in which the incidence of major coronary events (including myocardial infarction, coronary revascularization, or death from ischemic heart disease) was counted in 2168 women who underwent breast radiation between 1958 and 2001. The average mean dose to the heart was 4.9 Gy. The study showed the risk of cardiovascular events to begin within the first 5 years following RT completion and continue to increase up to 30 years after treatment. They found no difference in proportional increase in the rate of major coronary events per radiation dose unit in women with or without known cardiac risk factors at the time of RT. Criticisms of this study include changes across the eras of RT delivery as well as changing diagnosis and treatment of cardiac disease/events. Patients in this study did not undergo CT-based planning, and mean heart doses were estimated from 2-dimensional techniques. Concern was thus raised about the accuracy of the prediction model. The strength of this study, however, was the long-term data provided.
In 2017, van den Bogaard et al published a study looking at 910 women treated at a single institution with RT following breast-conserving therapy.12 The primary endpoint of the study was to evaluate the incidence of acute coronary events (ACE). The investigators evaluated mean heart doses as well as dose to cardiac substructures, including the right and left atria and ventricles, to determine whether dose to a particular cardiac substructure correlated with more risk. All patients underwent CT-based planning. The mean heart dose was 2.37 Gy, with a median follow-up of 7.6 years. Three percent of patients experienced an ACE. This study showed a risk of 16.5% increased incidence of ACE per gray of RT to the mean heart within the first 9 years after RT, with a c-statistic of 0.79 that ultimately validated Darby’s model. When evaluation by cardiac substructure was performed, the volume of the left ventricle receiving 5 Gy was the most important predictor of acute coronary events, with increasing doses predicting increased risk. Based on their statistical analysis, a threshold of mean value of 16.85% was associated with no ACE while a mean value of 29.4% was associated with an ACE. Of note, however, increasing doses of RT to the left ventricle were associated with increasing risks of an ACE. The authors also evaluated risk by decade of life at diagnosis (40s, 50s, 60s, 70s, 80s) and showed the highest risks for women in their eighth decade compared to the fourth decade. They also evaluated incidence by cardiac risk factor and found patients with a prior history of ischemic heart disease to have an exponentially worse risk of ACE compared to those with prior histories of hypertension or diabetes. The strengths of this analysis include the CT-based planning nature of their study with exact heart dosimetry and dosimetry to cardiac substructures, modern methods of diagnosis and treatment of ACEs, and moderately long follow-up. Weaknesses include the shorter nature of follow-up (compared to Darby et al). The incidences would likely continue to increase, with a slight modification of the risk ratio over time.
Taylor et al performed a systematic review of individual patient data published from 2010 to 2015.13 Their analysis included more than 40 000 patients, with a median follow-up of 10 years. Estimates of heart doses were used in this study rather than individual dosimetric data. They found an increased risk of cardiac mortality with an increased risk ratio of 1.3 (95% confidence interval [CI], 1.15 to 1.46) and a 0.04 excess rate ratio of cardiac mortality per gray of whole-heart dose. Their study found a history of ischemic heart disease and smoking to be confounding factors for risk of cardiac death. The fact that this study focused on cardiac death as opposed to cardiac events likely resulted in the lower correlation of mean whole-heart dose per gray.
Some feel the risk ratios presented by the Darby and van den Boggard analyses may be overestimated.14 For example, a study of > 70 000 Dutch stage I to III breast cancer patients showed that only death due to valvular heart disease was more frequent in these patients compared to the general Dutch population.15 Further work is needed to clarify the best dosimetric parameters to use regarding heart and/or cardiac substructures and subsequent treatment planning goals, although a general consensus targets achieving the lowest dose possible to the mean heart and left ventricle. The ongoing multicenter, prospective MEDIRAD EARLY HEART study seeks to identify and validate new cardiac imaging and circulating biomarkers of RIC focusing on changes arising within first 2 years of breast cancer RT.16 Patients receiving chemotherapy will be excluded. With plans to accrue 250 patients in the age group of 40-75 years, the data generated will also allow an opportunity to explore risk models correlating dose metrics of cardiac structures with the studied biomarkers while incorporating patient-specific risk factors. In a preliminary retrospective study, RT planning based on risk models that included patient age, smoking status, and existing cardiac risk factors at the time of RT was assessed.17 The risk models were developed using published multi-institutional data. In 39 patients with left-sided breast cancer treated with comprehensive postlumpectomy locoregional conformal RT planning, median total decrease achieved in mortality or recurrence was 0.4% (range = 0.06 to 2.0%) and 0.5% (range = 0.11 to 2.2%) without and with existing cardiac risk factors, respectively.
Based on available data, a clear relationship exists between whole-heart dose and risk of cardiac events following RT for breast cancer with a significant increase in risk for left-sided breast cancer patients.15,18 The clinical reality is that, as radiation oncologists, we are often unaware of the cardiac events our patients may experience. In addition, great heterogeneity in the length and frequency of patient follow-up for breast cancer contributes to this underappreciation. Patients, with a particular focus on those with left-sided disease, should be evaluated for cardiac-sparing techniques, including but not limited to deep-inspiration breath hold (DIBH), gating, prone positioning, and/or proton therapy, to achieve the lowest dose possible. Partial-breast irradiation can also be considered for suitable patients to decrease heart exposure. In addition to dose–volumetric parameters, radiation oncologists also must engage in smoking cessation counseling as well as education and discussion of the synergistic risks of other cardiac risk factors. As a result of the available data showing the confounding nature of cardiac risk factors, additional care should be taken when delivering RT for women with a history of ischemic cardiac disease.
Thoracic Malignancies (Lung and Esophageal Cancers)
Because of the overall higher mortality, evaluation of RIC in lung and esophageal cancers has proven more problematic than in breast cancer. Most patients do not live long enough to develop a cardiotoxicity. Nevertheless, recent recommendations for early screening of high-risk populations (ie, smokers) have increased the probability of diagnosing lung cancer at an earlier stage with longer life expectancy and less comorbidity.19 A 2019 statistical analysis of 11 3945 stage III non-small cell lung cancer (NSCLC) patients treated in 2004 to 2013 showed that 28% of the patients were younger than 60 years.20 Similarly, another 2019 study of 44 498 stage IV NSCLC patients treated in 2013 to 2014 showed that 31% of the patients were younger than 60 years.21 These findings highlight the importance of detecting and avoiding survival-compromising secondary complications in lung cancer RT as well as other types of thoracic RT. One study estimated the risks of RIC in lung cancer survivors to be as high as 33%.22,23 Another analysis of 127 stage III NSCLC patients treated between 1996 and 2009 showed that 2-year competing risk-adjusted RIC rates for patients with a heart mean dose of < 10 Gy, ≥ 10 to 20 Gy, and > 20 Gy were 4%, 7%, and 21%, respectively.22,24 Stam et al performed a study in 469 locally advanced NSCLC patients that showed a significant inverse correlation between increasing heart dose and survival.25 A retrospective single- institutional multivariate analysis of 251 patients with locally advanced NSCLC from Washington University, St. Louis, Missouri, for which cardiac structures were recontoured, increasing heart V50 (Vx: the percentage volume receiving ≥ x Gy), was independently associated with survival (2-year overall survival increased from < 25% for V50 ≥ 25%, to 45.9% for V50 < 25%, p < 0.0001).26
In the more recently published RTOG 0617 study, RT dose to the heart was found to be prognostic for likelihood of death. On both univariate and multivariate analysis, V5 and V30 of heart were associated with increased risk of death.27 In a secondary analysis reported subsequently, the incidence of grade 3+ cardiovascular events were lower with intensity-modulated RT (IMRT) vs 3-dimensional conformal RT (3D-CRT) (11% vs 21%, p = 0.131).28 It was postulated that heart dose might best explain inferior outcomes in the 74 Gy arm. While there were recommended constraints for heart, this was not a compliance criterion. Expectedly, to limit lung doses, an incidental increase in cardiac dose may have been seen in both groups.29 An important realization from the RTOG 0617 study was the significance of heart doses in a patient population with a median follow-up of < 24 months. This may become even more relevant in the modern era of consolidation immunotherapy, which is associated with a small risk of cardiac-related deaths.30 Dose to heart (sub)structures has also been linked with noncancer death in early stage NSCLC patients treated with stereotactic body RT (SBRT).31 In an analysis of 803 patients, at a median follow-up of 34.8 months, multivariate analysis identified maximum dose on the left atrium (median 6.5 Gy EQD2 [equivalent dose in 2 Gy fractions], range = 0.009 to 197, hazard ratio [HR] = 1.005, p = 0.035), and the dose to 90% of the superior vena cava (median 0.59 Gy EQD2, range = 0.003-70, HR = 1.025, p
= 0.008) were significantly associated with noncancer death.
As in lung cancer, the risks of esophageal cancer RIC have previously been underreported because of poor overall prognosis. Beukema et al conducted a retrospective analysis of patients receiving definitive concurrent chemoradiation.32 Grade 3 or higher cardiac events such as ischemia, effusions and heart failure were noted with a median follow-up of 26.1 to 57 months with an incidence ranging from 5.8 to 11.1%. Molenaar et al performed a Surveillance Epidemiology and End Results (SEER) analysis of patients receiving RT for esophageal cancer from 1973 to 2013.33 They analyzed 6514 patients, of whom 53% received RT and 44% did not. Nine percent of 5-year survivors experienced cardiac death: 336 who received RT compared to 254 who did not, with mean times to death of 25.3 and 32.2 years, respectively. On multivariate analysis, risks were highest in patients diagnosed prior to 1995 and in those with squamous cell carcinoma. Increased cardiac death in 1995 was likely partially the result of older RT techniques.
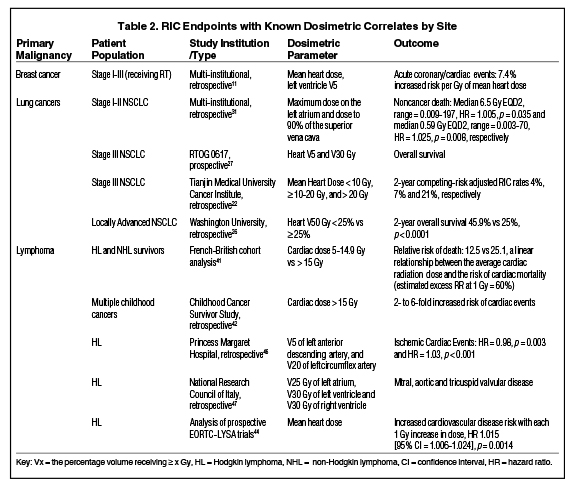
In both lung and esophageal cancers, RT techniques have progressed so that the majority of these patients are now treated with IMRT rather than 3D-CRT.34-36 IMRT has the ability to spare high doses of RT to smaller heart volumes at the cost of spreading lower doses over larger volumes. It remains unclear which is most important in avoiding RIC with data to support negative impact of both dosimetric parameters (Table 2
). Not having data to guide the decision, both lowering mean dose to whole heart and limiting high dose values to small volumes should be emphasized during treatment planning. The risk of RIC in both lung and esophageal cancers is heavily confounded by age as well as risk factors. The risk factors inherent in disease development are also risk factors for cardiac disease; as such, these patient populations are at even higher risk for RIC. In addition, many patients have been diagnosed with cardiac disease prior to their cancer diagnosis, highlighting an even greater need for heart avoidance. Because of the anatomic proximity of these cancers to the heart, however, radiomodulatory techniques such as DIBH or gating may not be as helpful in reducing heart dose; thus, other techniques, such as proton therapy, may be needed.
Lymphoma
RT continues to play an integral role in the management of Hodgkin lymphoma (HL) and is still used in select cases of non-Hodgkin lymphoma (NHL). Both diagnoses involve treatment with cardiotoxic systemic agents that further enhance cardiac risks of RT.37 Cardiac-related death is the third most common cause of death among lymphoma survivors, who have a 5.3 to 7.3 times increased risk of cardiac mortality compared to the general population.38 Among HL survivors, the risk of fatal myocardial infarction has been found to be 2.5 times higher than in the general population.3 Most of the cardiotoxicity data is derived from childhood survivors of HL, a highly curable disease, with toxicities including valvular heart disease (21% to 41%), coronary heart disease (17% to 23%), heart failure (8% to 17%), conduction disorders (12%) and pericardial abnormalities (10%).39
Data regarding relative contribution of doxorubicin-based chemotherapy and RT in causing RIC are heterogeneous. In a single-institutional analysis of 615 HL patients from Princess Margaret Hospital, Toronto, Canada, it was shown that while the rate of cardiac morbidity was highest among patients treated with both doxorubicin and mediastinal RT (HR = 2.77, p < 0.0001), mediastinal RT without chemotherapy also significantly increased this risk (HR = 1.82, p < 0.038).5 In a report from the German-Austrian Pediatric Hodgkin’s Disease Study Group, a longitudinal follow-up analysis of 1132 HL survivors who received treatment before 18 years of age in consecutive trials between 1978 and 1995, cumulative incidence of RIC after 25 years dropped with reduced radiation dose (21% with 36 Gy RT vs 3% with no RT, p < 0.001).40 Valvular defects were diagnosed most frequently, followed by coronary artery diseases, cardiomyopathies, conduction disorders, and pericardial abnormalities. A similar linear dose-response relationship was noted in another French-British cohort analysis of 4122 children, including HL and NHL survivors treated in 8 cancer treatment centers in France and the United Kingdom.41 Cumulative anthracycline dose and average radiation dose to the heart increased the risk of death from cardiac diseases (anthracycline RR [relative risk] = 4.4, cardiac dose 5 to 14.9 Gy RR = 12.5, cardiac dose > 15 Gy RR = 25.1) with a linear relationship between the average cardiac radiation dose and the risk of cardiac mortality (estimated excess RR at 1 Gy = 60%). A Childhood Cancer Survivor Study from 26 institutions evaluated 14 358 5-year survivors of cancer diagnosed under age 21 and noted a 2 to 5 times increased risk of congestive heart failure, pericardial disease, and valvular abnormalities compared with untreated sibling survivors.42 Cardiac radiation exposure > 15 Gy also resulted in a 2- to 6-fold increased risk of the above cardiac events. A Dutch case-control study of HL patients diagnosed before age 51 years who had a 5-year follow-up showed a higher mean left ventricular dose (MLVD) (16.7 Gy vs 13.8 Gy, p = 0.003).43 The risk of heart failure was also correlated with MLVD (MLVD 1 to 15 Gy, 16 to 20 Gy, 21 to 25 Gy, and ≥ 26 Gy: RR of heart failure 1.27, 1.65, 3.84, and 4.39, respectively, Ptrend < 0.001). Further, this risk was increased with anthracycline use (MLVD 0 to 15 Gy, 16 to 20 Gy, and ≥ 21 Gy: Cumulative risk of heart failure was 4.4%, 6.2%, and 13.3%, respectively, without anthracycline and 11.2%, 15.9%, and 32.9%, respectively, with anthracycline). The largest analysis of prospective data comes from EORTC-LYSA trials for patients with HL.44 Dose to the heart and carotids was reconstructed to a demonstrated increased risk of cardiovascular disease with an increased mean heart dose (per 1 Gy increase in dose, HR = 1.015 [95% CI, 1.006-1.024], p = .0014). Dose to carotid arteries did not correlate with a similar risk.
A major limitation of such survivorship studies is lack of details on true 3D cardiac dose and treatment with conventional large-field treatments including mantle/mini-mantle, total body radiation or use of cobalt-60 machines. With the evolution of more modern treatment planning principles of reduced treatment dose targeting smaller involved-site and involved-nodal regions combined with increasing use of modern treatment technologies such as IMRT and proton therapy, dose to cardiac substructures may become more relevant than whole cardiac dose.45 In a random sample of 125 HL patients treated with mediastinal RT, 44 cardiac events were documented, of which 70% were ischemic.46 In a sub- analysis of ischemic cardiac events, V5 of the left anterior descending artery (HR = 0.98, p = .003), and V20 of the left circumflex artery (HR = 1.03, p < .001) were found to be significant predictors. In a modern cohort analysis, 56 patients undergoing cytotoxic chemotherapy and involved-field 3D-CRT for HL were retrospectively analyzed.47 V25 Gy of left atrium, V30 Gy of left ventricle and V30 Gy of right ventricle correlated with mitral, aortic and tricuspid valvular disease, respectively, yielding 32.1% of patients developing valvular regurgitation and/or stenosis after a median follow-up of 70.5 months. In a more recent prospective analysis, 179 consecutive asymptomatic patients with HL were evaluated with coronary CTA.48 With a median follow-up of 11.6 years, 26% survivors demonstrated CTA abnormalities, with 15% of patients demonstrating changes within 5 years and 6.7% demonstrating severe stenoses requiring surgical procedures. Radiation dose to the coronary artery origins was noted to be prognostic.
Ongoing efforts will require continued monitoring of 3D dose-distribution to cardiac substructures in the era of modern radiation planning and delivery principles to refine the dosimetric constraints. Equally important will be efforts toward cardiac rehabilitation.
Treatment and Management of Radiation-Induced Cardiotoxicity
To date, no treatment is available to reverse or treat the effects of RIC. The focus of treatment paradigms has been on optimizing medical management of other cardiac risk factors, such as hypertension and diabetes, and preventing disease through education toward a smoke-free and heart-healthy lifestyle. Smoking cessation and counseling have played critical roles in reducing risk.
Over the last several years, the field of cardio-oncology has emerged as a multidisciplinary field of cardiologists, medical oncologists, and sometimes radiation oncologists specializing in both temporary and long-term effects of oncology-related cardiotoxicity with a goal of improving the quality of life of cancer survivors.49 A pilot project in lymphoma patients undergoing stem cell transplant demonstrated improved exercise levels and physical functioning with guided cardiac-rehabilitation exercises.50 Similar efforts should be initiated for patients receiving cardiac exposure from RT. The indications for patient referral for this field vary by institution/locale. In some cases, any patient at potential risk for cardiotoxicity may be referred for consultation and subsequent follow-up. In others, patients may be referred only when they begin to show signs of cardiotoxicity (eg, a patient who develops a decreased ejection fraction while on trastuzumab). As the number of cancer survivors continues to increase, the role of cardio-oncology becomes more important, with a call for a greater number of providers.
Conclusion
RIC is a known late effect of breast and thoracic RT in childhood cancer survivors. Population-based and institutional analyses in recent years have provided some dosimetric correlates to better predict the risk of RIC in relationship to cardiac radiation exposure. However, assessments are limited by lack of 3D anatomical data, use of conventional treatment planning and delivery technology, and relative lack of dosimetric significance of dose to various cardiac substructures. Furthermore, true assessment of RIC is limited by lack of follow-up, cancer-related mortality, pre-existing conditions and age-related changes. In the absence of an approved treatment for RIC, reducing the clinical impact of RIC focuses on minimizing dose to the heart through advanced RT delivery techniques, smaller RT volumes and/or decreased doses of RT. Treatment paradigms also focus on preventing cardiac risk factors. With the evolution of more modern treatment planning principles of reduced treatment dose targeting smaller involved-site and involved-nodal regions combined with increasing use of modern treatment technologies such as IMRT and proton therapy, dose to cardiac substructures may become more relevant than whole cardiac dose. All radiation oncologists should be aware of RIC, with a call to action to support advanced delivery techniques. Although these techniques may sometimes come at an increased short-term cost, reducing RIC will lead to long-term gains for patients, for the scientific understanding of cardiac toxicity, and for the medical establishment.
References
- Bloom MW, Hamo CE, Cardinale D, et al. Cancer therapy-related cardiac dysfunction and heart failure: part 1: definitions, pathophysiology, risk factors, and imaging. Circ Heart Fail. 2016;9(1):e002661.
- Aleman BM, van den Belt-Dusebout AW, De Bruin ML, et al. Late cardiotoxicity after treatment for Hodgkin lymphoma. Blood. 2007;109(5):1878-1886.
- Swerdlow AJ, Higgins CD, Smith P, et al. Myocardial infarction mortality risk after treatment for Hodgkin disease: a collaborative British cohort study. J Nat Cancer Inst. 2007;99(3):206-214.
- Moja L, Tagliabue L, Balduzzi S, et al. Trastuzumab containing regimens for early breast cancer. Cochrane Database Syst Rev. 2012;(4):Cd006243.
- Myrehaug S, Pintilie M, Tsang R, et al. Cardiac morbidity following modern treatment for Hodgkin lymphoma: supra-additive cardiotoxicity of doxorubicin and radiation therapy. Leuk Lymphoma. 2008;49(8):1486-1493.
- Awadalla M, Hassan MZO, Alvi RM, Neilan TG. Advanced imaging modalities to detect cardiotoxicity. Curr Probl Cancer. 2018;42(4):386-396.
- Akhter N, Murtagh G, Yancy C. Strategies for early detection of cardiotoxicities from anticancer therapy in adults: evolving imaging techniques and emerging serum biomarkers. Future Oncol. 2015;11(14):2093-2013.
- Lancellotti VT, Nkomo, Badano LP, et al. Expert consensus for multi-modality imaging evaluation of cardiovascular complications of radiotherapy in adults: a report from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. Eur Heart J Cardiovasc Imaging. 2013;14(8):721-740.
- Sylvester CB, Abe JI, Patel ZS, Grande-Allen JK. Radiation-induced cardiovascular disease: mechanisms and importance of linear energy transfer. Front Cardiovasc Med. 2018;5:5.
- Taunk NK, Haffty BG, Kostis JB, Goyal S. Radiation-induced heart disease: pathologic abnormalities and putative mechanisms. Front Oncol. 2015;5:39.
- Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. New Eng J Med. 2013;368(11):987-998.
- van den Bogaard VA, Ta BD, van der Schaaf A, et al. Validation and modification of a prediction model for acute cardiac events in patients with breast cancer treated with radiotherapy based on three-dimensional dose distributions to cardiac substructures. J Clin Oncol. 2017;35(11):1171-1178.
- Taylor C, Correa C, Duane FK, et al. Estimating the risks of breast cancer radiotherapy: evidence from modern radiation doses to the lungs and heart and from previous randomized trials. J Clin Oncol. 2017;35(15):1641-1649.
- Schneider U, Ernst M Hartmann M. The dose-response relationship for cardiovascular disease is not necessarily linear. Radiat Oncol. 2017;12(1):74.
- Boekel NB, Schaapveld M, Gietema JA, et al. Cardiovascular disease risk in a large, population-based cohort of breast cancer survivors. Int J Radiat Oncol Biol Phys. 2016. 94(5):1061-1072.
- Walker VA, Crijns, Langendijk J, et al. Early detection of cardiovascular changes after radiotherapy for breast cancer: protocol for a european multicenter prospective cohort study (MEDIRAD EARLY HEART Study). JMIR Res Protoc. 2018;7(10):e178.
- Stick LB, Vogelius IR, Modiri A, et al. Inverse radiotherapy planning based on bioeffect modelling for locally advanced left-sided breast cancer. Radiother Oncol. 2019;136:9-14.
- Boero IJ, Paravati AJ, Triplett DP, et al. Modern radiation therapy and cardiac outcomes in breast cancer. Int J Radiat Oncol Biol Phys. 2016;94(4):700-708.
- Final Research Plan for Lung Cancer: Screening. 2018. https://www.uspreventiveservicestaskforce.org/Page/Document/final-research-plan/lung-cancer-screening1. Accessed February 15, 2020.
- Vyfhuis MAL, Bentzen SM, Molitoris, JK, et al. Patterns of care and survival in stage III NSCLC among black and latino patients compared with white patients. Clin Lung Cancer. 2019;20(4):248-257.e4.
- Foster CC, Sher DJ, Rusthoven CG, et al. Overall survival according to immunotherapy and radiation treatment for metastatic non-small-cell lung cancer: a National Cancer Database analysis. Radiat Oncol. 2019;14(1):18.
- Ming X, Feng Y, Yang C, et al. Radiation-induced heart disease in lung cancer radiotherapy: a dosimetric update. Medicine (Baltimore). 2016. 95(41):e5051.
- Hardy D, C.C. Liu, et al. Cardiac toxicity in association with chemotherapy and radiation therapy in a large cohort of older patients with non-small-cell lung cancer. Ann Ocol. 2010;21(9):1825-1833.
- Wang K, Eblan MJ, Deal, MA, et al. Cardiac toxicity after radiotherapy for stage III non-small-cell lung cancer: pooled analysis of dose-escalation trials delivering 70 to 90 Gy. J Clin Oncol. 2017. 35(13):1387-1394.
- Stam B, van der Bijl E, van Diessen J, et al. Heart dose associated with overall survival in locally advanced NSCLC patients treated with hypofractionated chemoradiotherapy. Radiother Oncol. 2017;125(1):62-65.
- Speirs CK, DeWees TA, Rehman S, et al. Heart dose is an independent dosimetric predictor of overall survival in locally advanced non-small cell lung cancer. J Thorac Oncol. 2017;12(2):293-301.
- Bradley JD, Paulus R, Komaki R, et al. Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): a randomised, two-by-two factorial phase 3 study. Lancet Oncol. 2015;16(2):187-199.
- Chun SG, Hu C, Choy H, et al. Impact of intensity-modulated radiation therapy technique for locally advanced non-small-cell lung cancer: a secondary analysis of the NRG Oncology RTOG 0617 randomized clinical trial. J Clin Oncol. 2017;35(1):56-62.
- Thor M, Deasy JO, Hu C, et al. The role of heart-related dose-volume metrics on overall survival in the RTOG 0617 clinical trial. Int J Radiat Oncol Biol Phys. 2018;102(3):S96.
- Antonia SJ, A. Villegas, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. New Engl J Med. 2018. 379(24):2342-2350.
- Stam B, Peulen H, Guckenberger M, et al. Dose to heart substructures is associated with non-cancer death after SBRT in stage I-II NSCLC patients. Radiother Oncol. 2017;123(3):370-375.
- Beukema JC, van Luijk P, Widder J, Langendijk J, Muijs CT. Is cardiac toxicity a relevant issue in the radiation treatment of esophageal cancer? Radiother and Oncol. 2015;114(1):85-90.
- Molenaar RJ, Radivoyevitch T, Hulshof M, Van Laarhoven HWM. Cardiac death rates after irradiation for esophageal cancer: an epidemiologic study among esophageal cancer survivors. J Clin Oncol. 2017;35(15_suppl):4049.
- Peng J, Pond G, Donovan E, Ellis PM, Swaminath A. A comparison of radiation techniques in patients treated with concurrent chemoradiation for stage III non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 2020.
- Koshy M, Malik R, Spiotto M, et al. Association between intensity modulated radiotherapy and survival in patients with stage III non-small cell lung cancer treated with chemoradiotherapy. Lung Cancer. 2017;108:222-227.
- Haque W, Verma V, Butler EB, Teh BS. Utilization of neoadjuvant intensity-modulated radiation therapy and proton beam therapy for esophageal cancer in the United States. J Gastrointest Oncol. 2018;9(2):282-294.
- Aleman BM, van den Belt-Dusebout AW, Klokman WJ, et al. Long-term cause-specific mortality of patients treated for Hodgkin’s disease. J Clin Oncol. 2003;21(18):3431-3429.
- Boyne DJ, Mickle AT, Brenner DR, et al. Long-term risk of cardiovascular mortality in lymphoma survivors: a systematic review and meta-analysis. Cancer Med. 2018;7(9):4801-4813.
- Ratosa I, Pantar MI. Cardiotoxicity of mediastinal radiotherapy. Rep Pract Oncol Radiother. 2019;24(6):629-643.
- Schellong G, Riepenhausen M, Bruch C, et al. Late valvular and other cardiac diseases after different doses of mediastinal radiotherapy for Hodgkin disease in children and adolescents: report from the longitudinal GPOH follow-up project of the German-Austrian DAL-HD studies. Pediatr Blood Cancer. 2010;55(6):1145-52.
- Tukenova M, Guibout C, Oberlin O, et al. Role of cancer treatment in long-term overall and cardiovascular mortality after childhood cancer. J Clin Oncol. 2010;28(8);1308-1315.
- Mulrooney DA, Yeazel MW, Kawashima T, et al. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: retrospective analysis of the Childhood Cancer Survivor Study cohort. Brit Med J. 2009;339:b4606.
- van Nimwegen FA, Schaapveld M, Cutter DJ, et al. Radiation dose-response relationship for risk of coronary heart disease in survivors of hodgkin lymphoma. J Clin Oncol. 2015;34(3):235-243.
- Maraldo MV, Giusti F, Vogelius IR, et al. Cardiovascular disease after treatment for Hodgkin’s lymphoma: an analysis of nine collaborative EORTC-LYSA trials. Lancet Haematol. 2015;2(11):e492-502.
- Hoppe BS, Bates JE, Mendenhall NP, et al. The meaningless meaning of mean heart dose in mediastinal lymphoma in the modern radiation therapy era. Pract Radiat Oncol. 2019.
- Hahn E, Jiang H, Ng A, et al. Late cardiac toxicity after mediastinal radiation therapy for Hodgkin lymphoma: contributions of coronary artery and whole heart dose-volume variables to risk prediction. Int J Radiat Oncol Biol Phys. 2017;98(5):1116-1123.
- Cella L, Liuzzi R, Conson M, et al. Dosimetric predictors of asymptomatic heart valvular dysfunction following mediastinal irradiation for Hodgkin’s lymphoma. Radiother Oncol. 2011;101(2):316-321.
- Girinsky T, M’Kacher R, Lessard N, et al. Prospective coronary heart disease screening in asymptomatic Hodgkin lymphoma patients using coronary computed tomography angiography: results and risk factor analysis. Int J Radiat Oncol Biol Phys. 2014;89(1):59-66.
- Koutsoukis A, Ntalianis A, Repasos E, et al. Cardio-oncology: a focus on cardiotoxicity. Euro Cardiol. 2018;13(1):64-69.
- Rothe D, Cox-Kennett N, Buijs DM, et al. Cardiac rehabilitation in patients with lymphoma undergoing autologous hematopoietic stem cell transplantation: a cardio-oncology pilot project. Can J Cardiol. 2018;34(10 Suppl 2):S263-s269.
- Raghunathan D, Khilji MI, Hassan SA, Yusuf SW. Radiation-induced cardiovascular disease. Curr Atheroscler Rep. 2017;19(5):22.
- Hudson MM, Ness KK, Gurney JG, et al. Clinical ascertainment of health outcomes among adults treated for childhood cancer. J Am Med Assoc. 2013;309(22):2371-2381.
- Fidler MM, Reulen RC, Henson K, et al. Population-based long-term cardiac-specific mortality among 34 489 five-year survivors of childhood cancer in Great Britain. Circulation. 2017;135(10):951-963.
- Boerma M, Sridharan V, Mao X-W, et al. Effects of ionizing radiation on the heart. Mutat Res. 2016;770(Pt B):319-327.
- van Nimwegen FA, Ntentas G, Darby SC, et al. Risk of heart failure in survivors of Hodgkin lymphoma: effects of cardiac exposure to radiation and anthracyclines. Blood. 2017;129(16):2257-2265.
- Heidenreich A, Schnittger I, Strauss HW, et al. Screening for coronary artery disease after mediastinal irradiation for Hodgkin’s disease. J Clin Oncol. 2007;25(1):43-49.
- Brown ML, Schaff HV, Sundt TM. Conduit choice for coronary artery bypass grafting after mediastinal radiation. J Thorac Cardiovasc Surg. 2008;136(5):1167-1171.
- Totterman KJ, E. Pesonen, Siltanen P. Radiation-related chronic heart disease. Chest. 1983;83(6):875-878.
- Marks LB, Yu X, Zhou S-M, et al. The incidence and functional consequences of RT-associated cardiac perfusion defects. Int J Radiat Oncol Biol Phys. 2005;63(1):214-223.
- Mousavi N and A. Nohria, Radiation-induced cardiovascular disease. Curr Treat Options Cardiovasc Med. 2013;15(5):507-517.
- Veinot J and W.D. Edwards, Pathology of radiation-induced heart disease: a surgical and autopsy study of 27 cases. Hum Pathol. 1996.27(8):766-73.
- Yusuf SW, Sami S, Daher IN, et al. Radiation-induced heart disease: a clinical update. Cardiol Res Pract. 2011;2011:317659.
- Ling LH, Oh JK, Schaff HV, et al. Constrictive pericarditis in the modern era: evolving clinical spectrum and impact on outcome after pericardiectomy. Circulation. 1999;100(13):1380-1386.
- Brosius FC 3rd, Waller BF, Roberts WC. Radiation heart disease. Analysis of 16 young (aged 15 to 33 years) necropsy patients who received over 3,500 rads to the heart. Am J Med. 1981;70(3):519-530.
- Bijl JM, Roos MM, Leeuwen-Segarceanu EM, et al. Assessment of valvular disorders in survivors of hodgkin’s lymphoma treated by mediastinal radiotherapy +/- chemotherapy. Am J Cardiol. 2016;117(4):691-696.
- Heidenreich A, Hancock SL, Lee BK, Mariscal CS, Schnittger I. Asymptomatic cardiac disease following mediastinal irradiation. J Am Coll Cardiol. 2003;42(4):743-749.
- Cella L, Liuzz R, Conson M, et al. Multivariate normal tissue complication probability modeling of heart valve dysfunction in Hodgkin lymphoma survivors. Int J Radiat Oncol Biol Phys. 2013;87(2):304-310.
- Paven E, Cimadevilla C, Urena M, et al. Management of radiation-induced valvular heart disease due to Hodgkin’s lymphoma in the modern era. EuroIntervention, 2018;13(15):e1771-e1773.
- Hull MC, Morris CG, Pepine CJ, Mendenhall NP. Valvular dysfunction and carotid, subclavian, and coronary artery disease in survivors of hodgkin lymphoma treated with radiation therapy. J Am Med Assoc. 2003;290(21):2831-2837.
- Cutter DJ, Schaapvel M, Darby SC, et al. Risk for valvular heart disease after treatment for hodgkin lymphoma. J Nat Cancer Inst. 2015;107(4).
- Mulrooney DA, Armstrong GT, Huang S, et al. Cardiac outcomes in adult survivors of childhood cancer exposed to cardiotoxic therapy: a cross-sectional study. Ann Internal Med. 2016;164(2):93-101.
- Al-Kindi SG, Oliveira GH. Heart transplantation outcomes in radiation-induced restrictive cardiomyopathy. J Cardiac Failure. 2016;22(6):475-478.
- Groarke JD, Tanguturi VK, Hainer J, et al. Abnormal exercise response in long-term survivors of Hodgkin lymphoma treated with thoracic irradiation: evidence of cardiac autonomic dysfunction and impact on outcomes. J Am Coll Cardiol. 2015;65(6):573-583.
- Adams MJ, Lipshultz SE, Schwartz C, Fajardo LF, Coen V, Constine LS. Radiation-associated cardiovascular disease: manifestations and management. Semin Radiat Oncol. 2003;13(3):346-356.
- Wu S, Tam M, Vega RM, Perez CA, Gerber NK. Effect of Breast irradiation on cardiac disease in women enrolled in BCIRG-001 at 10-year follow-up. Int J Radiat Oncol Biol Phys. 2017;99(3):541-548.
- Bhattacharya S, Asaithamby A. Ionizing radiation and heart risks. Sem Cell Develop Biol. 2016;58:14-25.
Citation
EM N, A M, P M. Cardiotoxicity and Radiation Therapy: A Review of Clinical Impact in Breast and Thoracic Malignancies. Appl Radiat Oncol. 2020;(1):16-23.
April 4, 2020