Acoustic neuroma
Images
CASE SUMMARY
A 59-year-old ambidextrous male presented with progressive right-sided hearing loss over several years. Pertinent physical exam findings included decreased hearing acuity on the right side and intact facial and trigeminal nerves.
IMAGING FINDINGS
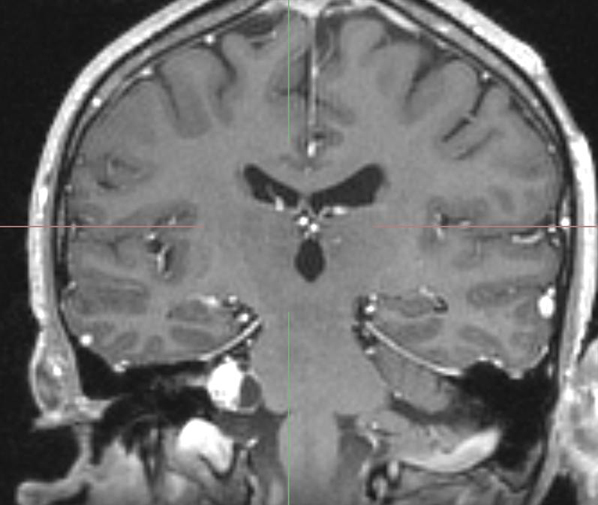
Magnetic resonance imaging (MRI) of the brain showed a 1.5- to 1.8-cm solid and cystic mass that enhances on T1 with contrast at the right cerebellopontine angle and extends into the internal auditory canal, abutting the cerebellum medially (Figure 1).
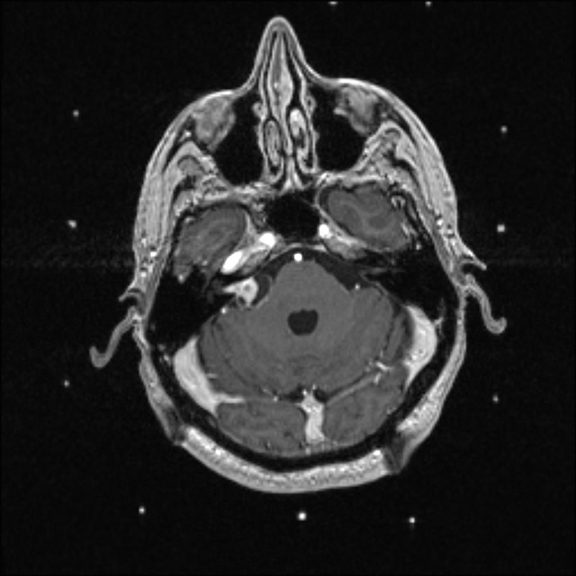
On the day of treatment a stereolocalization MRI showed a mass in the right cerebellopontine angle extending into the right internal auditory canal with heterogeneous enhancement and cystic peripheral areas. Mass measured approximately 2.5 × 2.1 × 1.2 cm in craniocaudal (CC), transverse, and anteroposterior (AP) dimensions, respectively. There was mild mass effect on adjacent brainstem. Remainder of the cranial nerves were normal in course and caliber. (Figure 2)
DIAGNOSIS
Acoustic neuroma (vestibular schwannoma)
DISCUSSION
Acoustic neuromas, or vestibular schwannomas, are benign Schwann cell neoplasms arising from the myelin sheath of the vestibular portion of the eighth cranial nerve, and rarely the cochlear portion. These tumors comprise 80% to 90% of the tumors of the cerebellopontine angle, and 8% of intracranial tumors overall. The incidence is about 1/100,000 per year1 and has been rising due to the increase in MRI usage and subsequent incidental discovery. The median age of diagnosis is 50,1 and the tumors are seen unilaterally in 90% of cases with no predisposition for either side. Patients with neurofibromatosis type 2 are frequently seen with bilateral acoustic neuromas. Risk factors and associations for the development of this tumor include exposure to loud noise, childhood exposure of the cerebellopontine angle to low-dose radiation, and parathyroid adenoma.
The classical presentation of this lesion is unilateral hearing loss in about 95% of patients.2 Higher frequencies and speech discrimination are disproportionately decreased compared to overall hearing loss. Tinnitus is also a common symptom, seen in about 63% of patients. Other symptoms can also arise if the tumor compresses nearby structures, such as the facial nerve, trigeminal nerve, brain stem, and cerebellum. Tumor progression can result in herniation of the cerebellum as well as hydrocephalus and involvement of lower cranial nerves. Interestingly, Romberg and other conventional office balance tests are typically normal even in patients presenting with dizziness.
The differential diagnosis for such a lesion is extensive and includes meningioma, facial nerve schwannoma, glioma, cholesterol cyst, cholestetoma, hemangioma, aneurysm, arachnoid cyst, lipoma, and metastatic lesion. MRI can detect lesions as small as 1 to 2 mm in diameter and should be performed with gadolinium contrast. Alternatively, computed tomography (CT) can be done with and without contrast; however, the resolution is much more poor. An enhancing lesion on MRI/CT in the region of the internal auditory canal with possible extension into the cerebellopontine angle is the typical radiological finding.
Treatment options include observation, surgery, radiosurgery, fractionated radiotherapy, and proton therapy. Observation alone can be considered when the patient is older than 60, has significant comorbities, has a small tumor size, lacks symptoms, or due to patient preference. About 43% of patients will progress, 51% will remain stable, and 6% will regress with conservative management.3 Surgery has about a 15% to 20% 10-year recurrence rate for partially or incompletely resected tumors.4,5 Preservation of remaining hearing is between 37.5% and 57% and can vary greatly depending on the technique.6,7 The chance of mortality from surgery for acoustic neuromas is about 1% and depends heavily on the size of the tumor and surrounding structures.8 Surgical intervention would be indicated if the lesion is causing any mass effect symptoms. Radiosurgery is typically used for tumors ≤ 3 cm and has an excellent local control rate of > 95% in most institutions.9 Radiosurgery is typically defined as radiotherapy treatment utilizing a single fraction. Fractionated radiotherapy typically involves 5 or 6 weeks of therapy. There are similar outcomes between fractionated radiotherapy and radiosurgery, but fractionated radiotherapy requires a more intense time commitment for the patient.9 Hearing preservation is achieved in about 55% to 71% of cases with very low facial and trigeminal nerve toxicities.9 Proton therapy is the newest treatment technique used for acoustic neuromas, but the outcomes have not yet been on par with that of other more conventional therapies.9,10 In addition, the cost of proton therapy is also much greater than more established therapies.
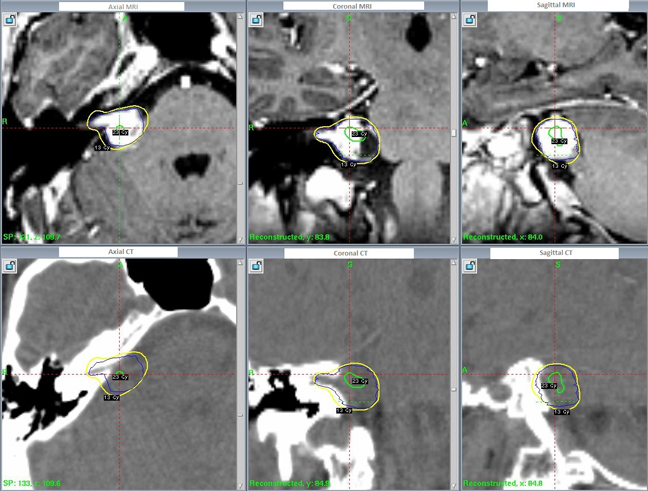
This particular patient opted for radiosurgery. The radiotheraputic technique used was GammaKnife stereotactic radiosurgery and the patient was treated with 13Gy to the 51% isodose line in a single fraction. The treatment planning was performed using a high-resolution, thin-slice MR after the fitting of a head frame (Figure 2). Subsequent follow-up at 6 months displayed cessation of tumor progression and the patient’s hearing loss was stabilized. He will continue to be followed with yearly MRIs.
CONCLUSION
Acoustic neuromas are relatively common benign intracranial lesions, which present typically with hearing loss and, less commonly, tinnitus. MRI findings will show an enhancing lesion arising from the internal auditory canal and possibly extending into the cerebellopontine angle. The incidence is rising due to increasing utilization of MRI. The current standard of care at most institutions is radiosurgery due to the very high local control rate, and relatively low rates of morbidity when compared to other treatments.
REFERENCES
- Propp JM, McCarthy BJ, Davis FG. Preston-Martin S. Descriptive epidemiology of vestibular schwannomas. Neuro Oncol. 2006;8:1-11.
- Matthies C, Samii M. Management of 1000 vestibular schwannomas (acoustic neuromas): Clinical presentation. Neurosurgery. 1997;40:1-9; discussion 9-10.
- Smouha EE, Yoo M, Mohr K, Davis RP. Conservative management of acoustic neuroma: A meta-analysis and proposed treatment algorithm. Laryngoscope. 2005;115:450-454.
- Cerullo L, Grutsch J, Osterdock R. Recurrence of vestibular (acoustic) schwannomas in surgical patients where preservation of facial and cochlear nerve is the priority. Br J Neurosurg. 1998;12: 547-552.
- Ohta S, Yokoyama T, Nishizawa S, Uemura K. Regrowth of the residual tumour after acoustic neurinoma surgery. Br J Neurosurg. 1998;12: 419-422.
- Regis J, Pellet W, Delsanti C, et al. Functional outcome after gamma knife surgery or microsurgery for vestibular schwannomas. J Neurosurg. 2002;97:1091-1100.
- Shahinian HK, Ra Y. 527 fully endoscopic resections of vestibular schwannomas. Minim Invasive Neurosurg. 2011;54:61-67.
- Samii M, Matthies C. Management of 1000 vestibular schwannomas (acoustic neuromas): Surgical management and results with an emphasis on complications and how to avoid them. Neurosurgery. 1997;40:11-21; discussion 21-23.
- Murphy ES, Suh JH. Radiotherapy for vestibular schwannomas: A critical review. Int J Radiat Oncol Biol Phys. 2011;79:985-997.
- Weber DC, Chan AW, Bussiere MR, et al. Proton beam radiosurgery for vestibular schwannoma: Tumor control and cranial nerve toxicity. Neurosurgery. 2003;53:577-86; discussion 586-588.