A Practical Method to Prolong Expiratory Breath Holds for Abdominal Stereotactic Body Radiation Therapy
Applied Radiation Oncology — Vol. 12, Issue 3, pp. 34-42
Published: September 1, 2023
1 Department of Radiation Oncology, University of Alabama at Birmingham, Birmingham, AL
* Corresponding author: Rojymon Jacob (rjacob@uabmc.edu)
Abstract
Keywords
abdominal stereotactic body radiation therapy, expiratory breath hold, supplemental oxygen, hyperventilation
Introduction
Stereotactic body radiation therapy (SBRT) is an increasingly utilized radiation technique that enables accurate delivery of ablative radiation doses with a steep dose fall-off to surrounding tissues. However, the utilization of SBRT for moving targets can be a significant challenge. Failure to account for respiratory motion can lead to underdosing targets and overdosing normal tissues.1,2 Motion management is especially important for abdominal SBRT cases given the high dose per fraction and steep dose gradients between the tumor target and nearby gastrointestinal viscera (such as the stomach and the bowel). Various strategies have been utilized to minimize the effects of respiratory motion during abdominal/thoracic SBRT, such as abdominal compression, amplitude- and phased-based gating, and breath-hold techniques, among others.1,3-5
Voluntary breath-hold techniques are attractive for motion management during abdominal SBRT. With this strategy, the beam is intermittently enabled only when the patient is holding their breath, which is coordinated via instructions from the radiation therapists. As the tumor and target tissue are effectively stationary during beam-on, there is no need for an internal target volume margin, which minimizes the volume of irradiated normal tissue to achieve adequate tumor target coverage. This reduction in motion translates to improved on-board cone-beam CT (CBCT) image quality, allowing more accurate patient alignment prior to SBRT treatment.6-8
Both inspiratory breath-hold (IBH)5,9,10 and expiratory breath hold (EBH)11-13 techniques have been successfully utilized for SBRT treatments. While EBH is more reproducible and minimizes target motion compared with IBH,13-19 it is generally more challenging for patients to perform an EBH of sufficient duration compared with IBH.20 Physiological studies of breath holding have shown that supplemental oxygen and mild hyperventilation can significantly improve breath-hold durations,20 with each technique adding incrementally to the improvement in breath-hold duration. Several pilot studies have demonstrated the clinical effectiveness of supplemental oxygen and mild hyperventilation for deep-inspiratory breath hold (DIBH) treatment in patients with breast cancer,21-23 but the effectiveness of this technique for patients undergoing EBH is unknown.
In this article, we report the experience of the first 20 patients treated with abdominal SBRT using a supplemented EBH technique (EBHsupp) with supplemental oxygen and mild hyperventilation. We evaluated data on individual patient EBH durations and treatment times, and we compared this data with a cohort of similar patients treated with EBHs without supplementation (EBHRA, room air, no mild hyperventilation). We hypothesized that the EBHsupp technique would prolong EBHs and reduce overall treatment time compared with EBHRA.
Materials and Methods
Patient Inclusion/Exclusion Criteria
All patients receiving 3-fraction abdominal SBRT treated with an EBH technique in our department from January 2018 onward receiving between 1300 cGy and 1500 cGy per fraction were included (Institutional Review Board 120703005). Other SBRT fractionation schemes were not included to reduce heterogeneity in treatment characteristics that might influence overall treatment time (eg, reduced monitor units [MUs] per treatment for 5-fraction plans or for 3-fraction plans with lower prescription doses, increased patient practice/experience with 5-fraction treatments). Patient demographic and clinicopathologic information (age, gender, diagnosis, and comorbidities) were obtained from the medical record, and treatment details (dose per fraction, number of treatment arcs, MUs delivered per treatment) were obtained from the oncology information system (OIS) (ARIA; Varian).
Treatment Procedure Details
Prior to August 2020, patients were treated with standard EBH without oxygen supplementation or coaching/prompting of their respiratory rate (RR) prior to EBH (designated EBHRA, where RA signifies room air). After August 2020, as part of a quality improvement initiative in our department and after a successful proof-of-concept study in healthy volunteers (see Supplementary Figure 1, available in the online version of this article at www.appliedradiationoncology.com), patients were offered treatment with a supplemented oxygen, mild hyperventilation EBH technique (EBHsupp). During both CT simulation and treatment, patients received 50% supplemental oxygen (50% fraction inspired oxygen or FiO2) via Venturi mask with an appropriate adapter and were instructed to synchronize their breathing at an RR of 18 breaths/min (ie, mild hyperventilation) with audio cues from an online metronome that was beamed into the simulation or treatment room (see Figure 1 and Supplementary Figure 2, available in the online version of this article at www.appliedradiationoncology.com, an online metronome available at https://www.imusic-school.com/en/tools/online-metronome, set at 36 beats/min with stress on the first beat to give a different audio cue for inhalation vs exhalation). The oxygen content of 50% FiO2 and RR of 18 breaths/min were chosen based on the known safety of these parameters in humans and the fact that similar parameters have been utilized in cancer patients performing inspiratory breath holds during radiation treatments.21,22 While breathing to the beat of the metronome was encouraged by radiation therapists, there were no measures to forcibly maintain the patient at a strict RR of 18 breaths/min prior to EBH. Initiation of supplemental oxygen and mild hyperventilation in patients undergoing EBHsupp was initiated just prior to setup imaging and stopped right after treatment beam-off (with no prolonged time for oxygenation prior to treatment start).
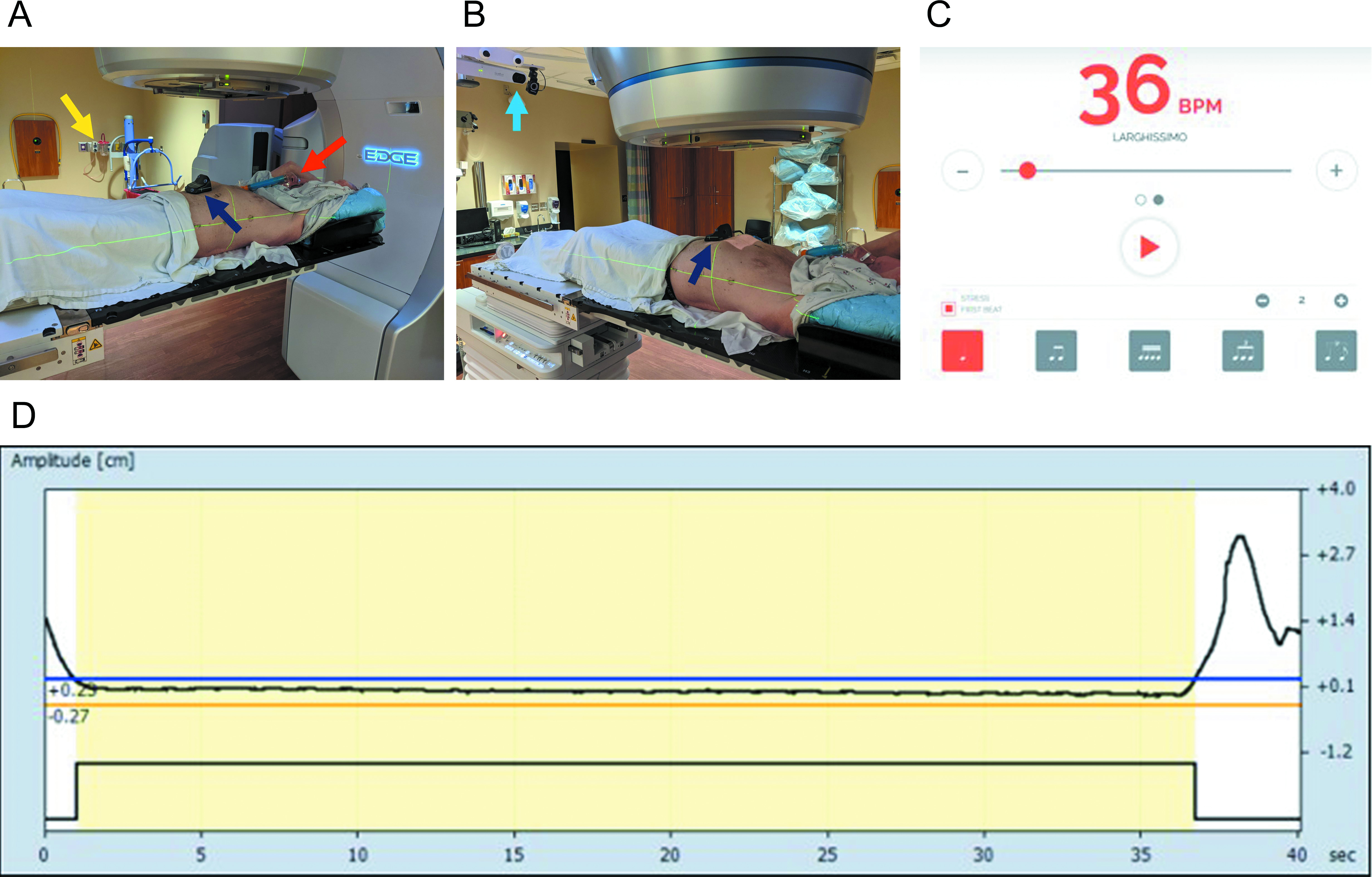
For both EBHRA and EBHsupp, patients were screened at the time of CT simulation, per standard departmental protocol, to verify their ability to perform repeated EBHs of more than 20 seconds’ duration. This was assessed by radiation therapy staff with the patient on the CT simulation table and in the treatment position with full immobilization gear and tracking of abdominal excursion via Varian’s Real-Time Position Management system (RPM). Completion of 2 consecutive, 20+ second EBHs within a 5-mm-amplitude window (as tracked by RPM) was required for the patient to move forward with EBH CT simulation and treatment. Patients not able to complete EBH simulation were treated with phased-based respiratory gating and are not described in this article. To be eligible for EBH-based SBRT treatments at our institution, all patients (both for EBHRA and EBHsupp) were required to have either implanted fiducials or radio-opaque transarterial chemoembolization (TACE) material within or directly adjacent to the target to allow for intrafraction kV x-ray real-time monitoring of motion in addition to RPM amplitude gating.
CT Simulation and Target Delineation
Following EBH CT simulation, contouring and planning were performed on the EBH CT simulation scans. Gross tumor volumes (GTVs) were defined on the EBH CT simulation scan with assistance from fused diagnostic images (eg, triple-phase MRI). For post-TACE targets without residual enhancing tumors, a clinical target volume (CTV) encompassing the TACE volume was contoured in lieu of a GTV. Otherwise, CTV was a 3- to 4-mm isotropic expansion from the GTV with cropping at natural boundaries (eg, edge of the liver). The planning target volume was generated via an isotropic 5-mm expansion of the CTV. Tracking structures (fiducials or TACE) were contoured with the bone window. Two planning organ-at-risk (PRV) volumes were generated for these tracking structures via 3-mm and 5-mm isotropic expansions for PRV3 and PRV5 structures, respectively, to allow for intrafraction kV assessment of tracking structure displacement during treatments. Volumetric-modulated arc therapy (VMAT) plans consisting of 2 or 3 coplanar arcs were generated for all patients.
Treatment Delivery
On the days of treatment, the general treatment workflow for both EBHRA and EBHsupp treatments was as follows (see Figure 2): initial alignment to bony structures with orthogonal EBH kV images, EBH CBCT for final target/fiducial alignment, and finally delivery of the treatment with 2-3 coplanar arcs. For EBHsupp patients, supplemental oxygen and metronome-cued breathing started right before setup imaging and stopped right after treatment beam-off, with no prolonged oxygenation prior to therapy.
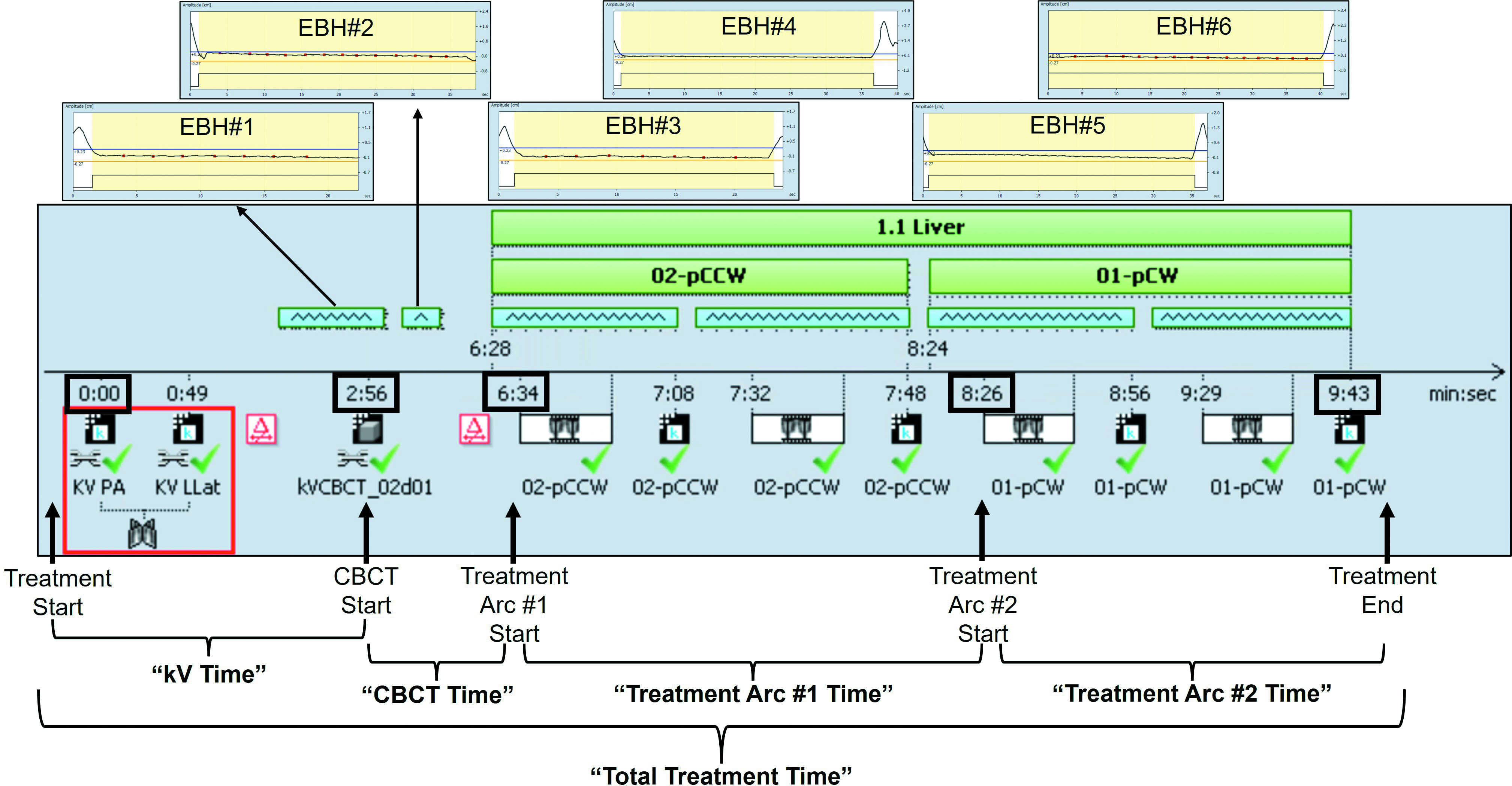
The RPM amplitude gating (5 mm) and intrafraction fiducial/TACE tracking with triggered kV images obtained every 10/20 degrees of gantry rotation were used to confirm breath-hold position during treatment (see Figure 1). Patients were instructed by therapy staff to breathe to the beat of the metronome in between EBH. A minimum of 6 breaths was recommended between each attempted EBH, but the timing of EBH attempts was left to the discretion of the radiation therapy staff. Additional images (eg, repeat kV or CBCT) were acquired as clinically necessary. All data regarding EBH duration and treatment time were automatically logged into the OIS.
Data Extraction
Individual patient EBH duration data for every breath hold for all treatments were extracted from the OIS in text file form, including beam on/off times during EBH (Figure 2). Individual patient EBH durations were penalized for beam holds (the time the beam was off during breath hold due to either excursion outside RPM amplitude window or fiducial excursion outside PRV5; see Figure 2 and Supplementary Figure S3, available in the online version of this article at www.appliedradiationoncology.com, for examples) so that only EBH time while the beam was on was counted. Total treatment time, time for completion of EBH orthogonal kV images, time for completion of EBH CBCT, and time for completion of each treatment arc were also extracted from the OIS and defined as illustrated in Figure 2. Briefly, the “kV Time” was defined as the time from Treatment Start (start of first kV acquisition) until CBCT Start (start of CBCT acquisition). The “CBCT Time” was defined as the time from CBCT Start until the time of Treatment Arc #1 Start (start of delivery of first treatment arc). The “Treatment Arc #1 Time” was defined as the time from Treatment Arc #1 Start until the time of Treatment Arc #2 Start (start of delivery of second treatment arc). The “Treatment Arc #2 Time” was defined as the time from Treatment Arc #2 Start until Treatment End (completion of second treatment arc). The “Total Treatment Time” was defined as the time from Treatment Start until Treatment End.
Statistical Analysis
Patient demographic, clinical, and treatment characteristics were compared between the EBHRA and EBHsupp groups via Student t-test (continuous variables) and the chi-square test (categorical variables). For categorical variables with more than 2 categories, variables were dichotomized prior to performing the chi-square test. Specifically, the “Diagnosis” variable was dichotomized as hepatocellular carcinoma or nonhepatocellular carcinoma and the “Dose per Fraction” variable was dichotomized as 1500 or more cGy or less than 1500 cGy. Individual patient max EBH, mean EBH, median EBH, and the number of EBHs required to complete treatment were compared between the EBHRA and EBHsupp groups via unpaired Student t-test. Individual patient EBH percentiles (10th, 25th, 50th, 75th, and 90th) were also determined and compared between groups via the Student t-test. Total treatment time and time for completion of individual treatment components (eg, time for completion of CBCT) were determined and compared between groups via Mann-Whitney U test given the non-normal distribution of the data. For all statistical tests, a P value < 0.05 was considered statistically significant.
Results
We identified a total of 46 patients meeting inclusion criteria who received 3-fraction SBRT with EBH treatment in our department after January 2018. Prior to the initiation of the EBHsupp technique in August 2020, 26 patients were treated with 28 treatment plans via standard EBHRA (2 patients had 2 liver tumors that were treated with separate treatment plans), accounting for a total of 83 EBHRA treatments (28 treatment plans × 3 fractions = 84 treatments minus 1 patient who received a liver transplant after the completion of only 2 fractions). At the time of this analysis, 20 patients were treated with the EBHsupp technique with 24 treatment plans (4 patients had 2 liver tumors who were treated with separate treatment plans) for a total of 72 EBHsupp treatments (24 treatment plans × 3 fractions = 72 treatments).
Patient demographic, clinicopathologic, and treatment parameters are shown in Table 1. Overall, there were no significant differences in any parameter between patients in the EBHRA and EBHsupp groups. The mean patient age was approximately 62 years old in both groups (62.4 vs 63.2, P = .788), and most patients were male (76.9% vs 90.0%, P = .246) and carried a diagnosis of hepatocellular carcinoma (73% vs 70%, P = .883). In all cases, patients received EBH SBRT to the liver. Patient comorbidity burden was similar between the groups as judged by the Charlson comorbidity index (CCI, 7.31 vs 7.60, P = .590). Treatment parameters, including dose per fraction and MU delivered per treatment, were similar between the groups, with nearly all patients receiving treatment with 2 co-planar arc VMAT plans (2 patients in the EBHsupp group were treated with 3 co-planar arc VMAT plans).
| VARIABLE | EBHRA(n = 26) | EBHsupp(n = 20) | P VALUE |
|---|---|---|---|
| Age (mean {SD}) | 62.4 {11.1} | 63.2 {10.0} | 0.7881 |
| Gender | |||
| Female | 6 (23.1) | 2 (10.0) | 0.246 |
| Male | 20 (76.9) | 18 (90.0) | |
| Diagnosis | |||
| Hepatocellular carcinoma | 19 (73.1) | 15 (70.0) | 0.883 |
| Intrahepatic cholangiocarcinoma | 1 (3.8) | 1 (5.0) | |
| Liver metastasis | 6 (23.1) | 4 (20.0) | |
| CCI (mean {SD}) | 7.31 {1.52} | 7.60 {2.14} | 0.59 |
| Current smoker (%) | 9 (34.6) | 5 (25.0) | 0.482 |
| Dose per fraction (cGy) | 0.415 | ||
| 1300 | 3 (11.5) | 5 (25.0) | |
| 1400 | 1 (3.8) | 0 (.0) | |
| 1500 | 22 (84.6) | 15 (75.0) | |
| Number of treatment arcs | 0.099 | ||
| 2 | 26 (100.0) | 18 (90.0) | |
| 3 | 0 (.0) | 2 (10.0) | |
| MU per treatment (mean {SD}) | 4162 {967} | 4058 {950} | 0.695 |
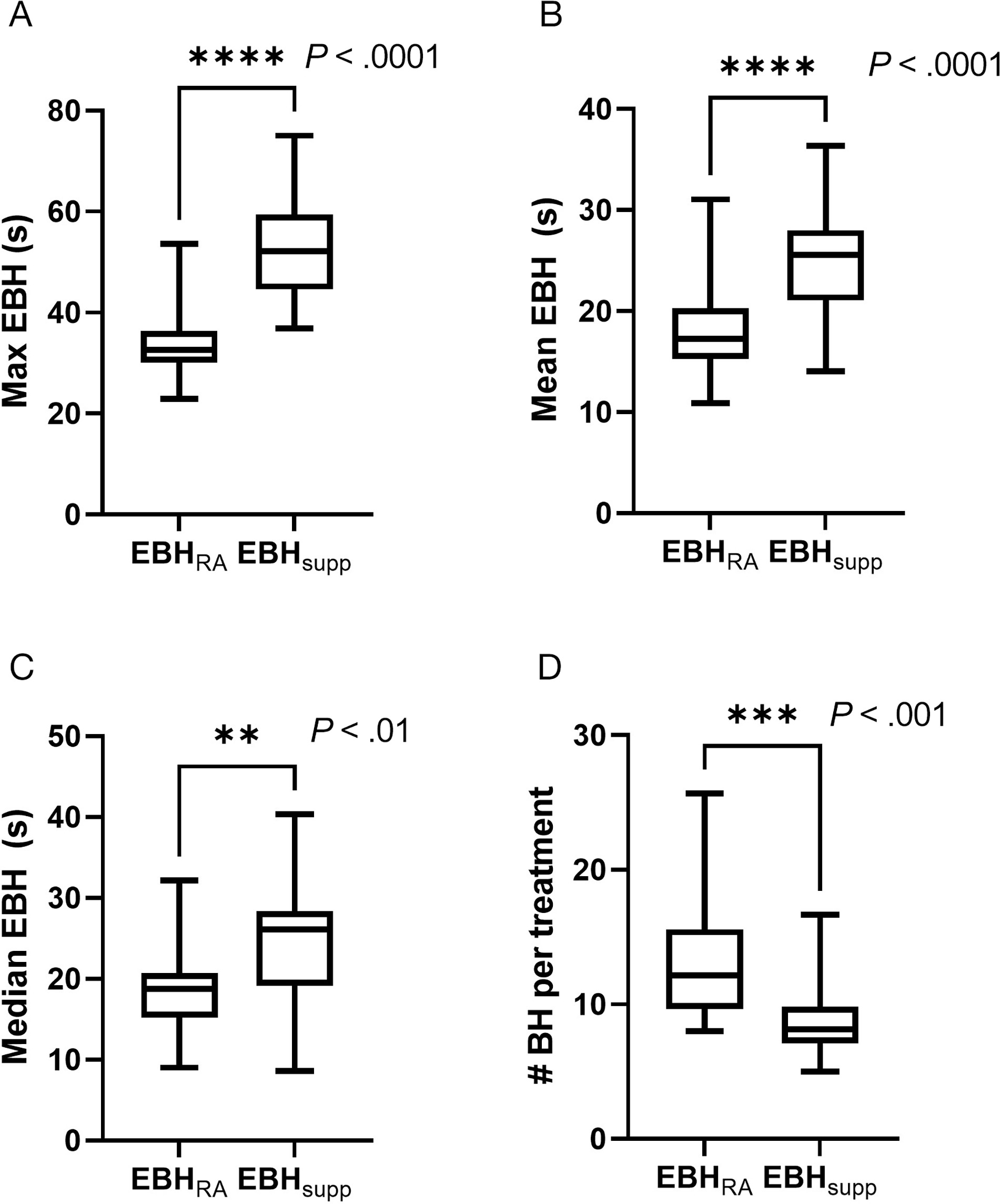
A total of 1735 individual EBHs were extracted from the OIS (Supplementary Figure S3, available in the online version of this article at www.appliedradiationoncology.com) and showed a significant increase in EBH duration for patients treated with the EBHsupp technique (Figure 3, Table 2). Maximum EBH for patients was significantly increased for patients treated with EBHsupp, with a difference in maximum EBH of ~18 seconds (34.5 s vs 52.8 s, P < .001). Mean (18.2 s vs 25.1 s, P < .001) and median (18.7 s vs 24.9 s, P = .002) EBH were also significantly increased for patients treated with EBHsupp compared with EBHRA. There was a corresponding decrease in the number of EBHs required to complete each treatment (12.7 vs 8.9, P < .001). Table 2 shows a percentile breakdown of patient EBH between the 2 groups with a statistically increased EBH observed with EBHsupp treatment for all individual patient EBH percentiles except for the lowest (10th) percentile (10th percentile: 6.4 s vs 7.8 s, P = .219).
| STATISTIC | EBHRA (s) | EBHsupp (s) | P VALUE |
|---|---|---|---|
| 10th percentile | 6.4 | 7.8 | 0.219 |
| 25th percentile | 11.3 | 14.7 | 0.031 |
| 50th percentile | 18.7 | 24.9 | 0.002 |
| 75th percentile | 24.6 | 34.7 | <.001 |
| 90th percentile | 29.3 | 43.1 | <.001 |
| Max | 34.5 | 52.8 | <.001 |
| Mean | 18.2 | 25.1 | <.001 |
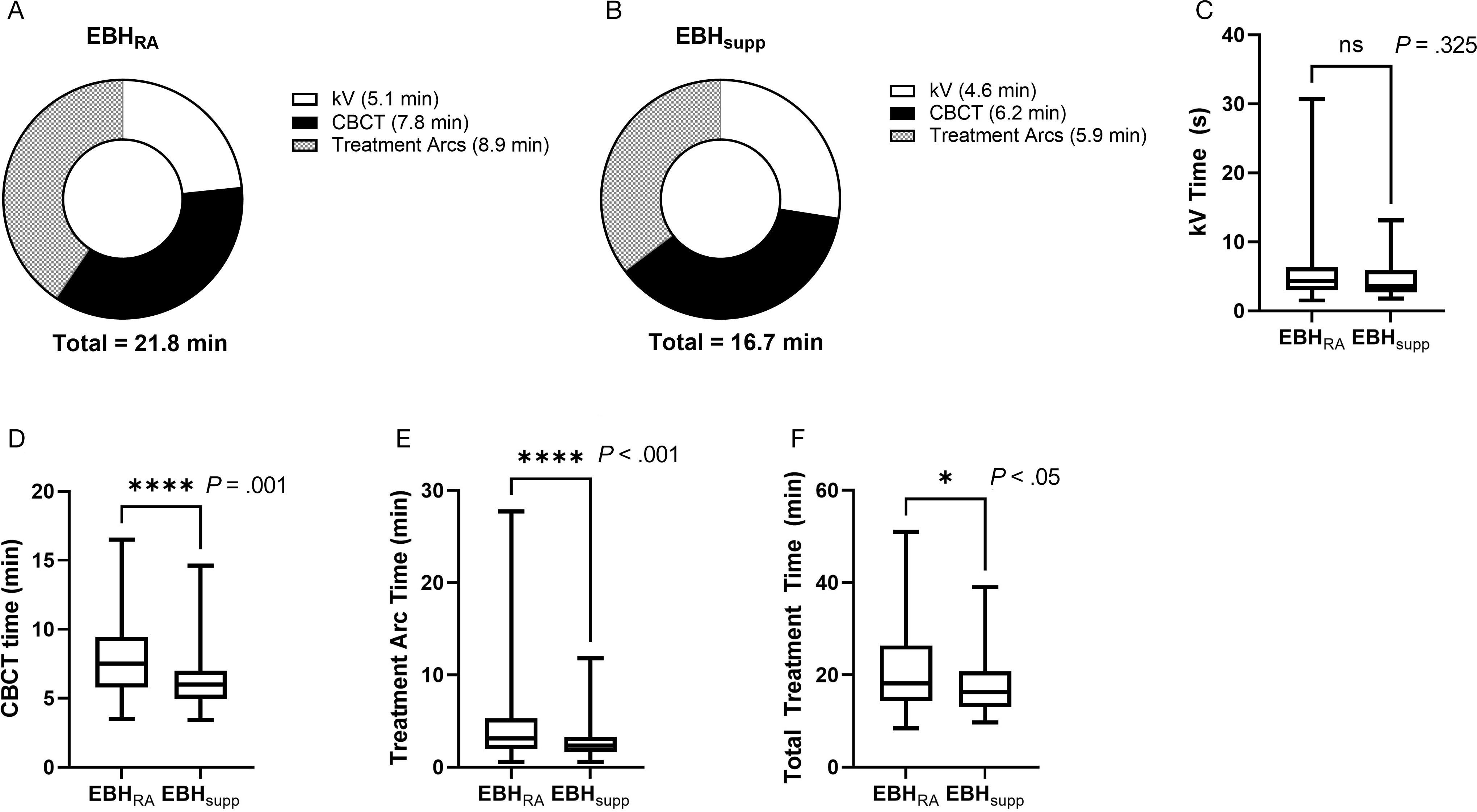
Examination of treatment times between the 2 groups (Figure 4) showed a significant reduction in the time required for completion of treatment in patients treated with EBHsupp compared with EBHRA, with a mean reduction of ~5 minutes (21.8 min vs 16.7 min, P = .025) and a median reduction of ~2 minutes (18.1 min vs 16.2 min). While the time required for completion of kV radiographs (“kV Time”) was not different between the 2 groups (P = .325), the time required for completion of CBCT (“CBCT Time,” P < .001) and each treatment arc (“Treatment Arc Time,” P < .001) was significantly reduced in patients treated with the EBHsupp technique.
Discussion
Expiratory breath hold is an effective method for motion management in patients undergoing abdominal SBRT treatments but is underutilized due to patients’ difficulty in performing repeated EBH of sufficient duration.1 In this study, we found that patients undergoing abdominal SBRT with supplemental oxygen and mild hyperventilation exhibited prolonged EBH durations compared with patients treated with nonsupplemented EBH. Patients in the EBHsupp group performed EBH of longer duration by all reported metrics (maximum EBH, mean EBH, and median EBH) and required less total EBH to complete treatments. These results agree with previously published experiences utilizing similar techniques in patients undergoing DIBH for breast cancer treatments20-22 as well as a recent randomized study of volunteers undergoing EBH that showed an improvement in median EBH duration from 24 seconds to 49 seconds with supplemental oxygen and mild hyperventilation.24
The reduction in treatment times observed in the EBHsupp group compared with the EBHRA group is likely related to improve EBH using the supplemented technique. This is supported by significant improvements in time to complete tasks that required prolonged EBH (eg, CBCT, treatment arcs) and a lack of significant improvement in tasks that did not require prolonged EBH (eg, kV acquisition/alignment). There were no significant differences in patient clinical/pathological characteristics between the groups (eg, age, diagnosis, and comorbidity index) nor in treatment-related parameters that might be expected to influence treatment time (eg, dose per fraction, MU delivered, and number of treatment arcs). During the study period, there were no other changes in departmental protocols or treatment-planning techniques as an alternate explanation for the reduction in treatment time observed. While the EBHsupp technique reduced treatment times in our study, there was an initial time investment (~10-15 min) at the time of CT simulation for added patient training for breathing with a metronome and the use of a Venturi mask.
The reported EBHsupp technique was safe in our study population. We had no patients who had any issues or symptoms related to the breath-hold component of their treatments (eg, no lightheadedness, syncope, tingling, tetany, or other concerns), which is in agreement with other studies that have shown the safety of breath holds (both inspiratory and expiratory) with mild hyperventilation and supplemental oxygen.20-25 The mild hyperventilation used in this study was chosen based on the RRs previously utilized and found to be safe in breast cancer patients undergoing DIBH treatment with prolonged hyperventilation.22,23 We specifically avoided more rapid hyperventilation given the theoretical increased risk of tetany that can occur with more aggressive hyperventilation and associated hypocapnia.26 Similarly, the level of supplemental oxygen of 50% FiO2 was selected based on safety as supplemental oxygen levels above 60% are associated with an increased risk of absorptive atelectasis.25,27-30
One safety issue that should be mentioned for all breath-hold treatments (not just supplemented ones such as the EBHsupp method) is the well-established increase in blood pressure during prolonged breath holds.25,31,32 However, the risk and severity of blood pressure rise during breath holds do not appear to be worsened by supplemented techniques, including a similar technique with mild hyperventilation and supplemental oxygen.25 Further, a recent randomized study of EBHs in volunteers did not find a significant change in blood pressure during EBH, possibly due to the relatively modest prolongation of EBHs with supplemented techniques (< 1 min) compared with DIBH (up to 5 min prolonged breath holds reported).24 In general, patient cardiopulmonary comorbidities should be considered by the treating radiation oncologist prior to proceeding with breath-hold treatment with discussion of the technique with the patient’s other involved physicians (eg, cardiologist and pulmonologist) for those with significant cardiopulmonary comorbidities.
There are several limitations to this study. First, this was not a randomized trial and. therefore, there is a possibility of biases (eg, selection bias) that could have partially influenced the results between the EBHRA and EBHsupp groups, though patient groups were well balanced overall with respect to both clinical parameters and treatment parameters. Second, all the patients described here received liver SBRT and, therefore, the results may not be generalizable to all patients receiving abdominal SBRT, though we have additionally treated several patients with primary pancreatic cancer with pancreas SBRT with the EBHsupp method with similar experience to those treated with liver SBRT. Lastly, this was a “real-world” study of EBHsupp implementation in a busy radiation oncology clinic, and we did not attempt to capture nor control all aspects of respiratory physiology that govern breath-hold capacity. While we encouraged patients to breathe at a rate that would normally correspond to mild hyperventilation, we did not forcibly control patient RR or tidal volumes and did not measure partial pressures of carbon dioxide; therefore, whether hyperventilation/hypocapnia was achieved for each patient is unknown. Future mechanistic studies of patients undergoing repeated, supplemented EBH with real-time measurement of these parameters will be helpful in further optimizing supplementation/hyperventilation protocols and ensuring uniformity of technique among individual patients.
Conclusions
Patients receiving supplemental oxygen and mild hyperventilation exhibited prolonged EBH time and reduced overall treatment time during abdominal SBRT. This intervention is simple, inexpensive, safe, and may improve individual patient breath-hold times, reduce treatment time, and increase the number of patients eligible for EBH-based abdominal SBRT.
References
- Brandner E, Chetty I, Giaddui T, Xiao Y, Huq M. Motion management strategies and technical issues associated with stereotactic body radiotherapy of thoracic and upper abdominal tumors: a review from NRG oncology. Med Phys. 2017;44(6):2595-2612. doi:doi:10.1002/mp.12227.
- Caillet V, Booth J, Keall P. IGRT and motion management during lung SBRT delivery. Phys Med. 2017;44:113-122. doi:doi:10.1016/j.ejmp.2017.06.006.
- Campbell W, Jones B, Schefter T, Goodman K, Miften M. An evaluation of motion mitigation techniques for pancreatic SBRT. Radiother Oncol. 2017;124(1):168-173. doi:doi:10.1016/j.radonc.2017.05.013.
- Dieterich S, Green O, Booth J. SBRT targets that move with respiration. Phys Med. 2018;56:19-24. doi:doi:10.1016/j.ejmp.2018.10.021.
- Boda-Heggemann J, Knopf A, Simeonova-Chergou A. Deep inspiration breath hold-based radiation therapy: a clinical review. Int J Radiat Oncol Biol Phys. 2016;94(3):478-492. doi:doi:10.1016/j.ijrobp.2015.11.049.
- Shen S, Duan J, Fiveash J. Validation of target volume and position in respiratory gated CT planning and treatment. Med Phys. 2003;30(12):3196-3205. doi:doi:10.1118/1.1626121.
- Shimizu S, Shirato H, Kagei K. Impact of respiratory movement on the computed tomographic images of small lung tumors in three-dimensional (3D) radiotherapy. Int J Radiat Oncol Biol Phys. 2000;46(5):1127-1133. doi:doi:10.1016/s0360-3016(99)00352-1.
- Mageras G, Pevsner A, Yorke E. Measurement of lung tumor motion using respiration-correlated CT. Int J Radiat Oncol Biol Phys. 2004;60(3):933-941. doi:doi:10.1016/j.ijrobp.2004.06.021.
- Hanley J, Debois M, Mah D. Deep inspiration breath-hold technique for lung tumors: the potential value of target immobilization and reduced lung density in dose escalation. Int J Radiat Oncol Biol Phys. 1999;45(3):603-611. doi:doi:10.1016/s0360-3016(99)00154-6.
- Murphy M, Martin D, Whyte R. The effectiveness of breath-holding to stabilize lung and pancreas tumors during radiosurgery. Int J Radiat Oncol Biol Phys. 2002;53(2):475-482. doi:doi:10.1016/s0360-3016(01)02822-x.
- Lin Y, Ozawa S, Miura H. Split-VMAT technique to control the expiratory breath-hold time in liver stereotactic body radiation therapy. Phys Med. 2017;40:17-23. doi:doi:10.1016/j.ejmp.2017.06.022.
- Yang W, Fraass B, Reznik R. Adequacy of inhale/exhale breathhold CT based ITV margins and image-guided registration for free-breathing pancreas and liver SBRT. Radiat Oncol. 2014;9:11. doi:doi:10.1186/1748-717X-9-11.
- Oliver P, Yewondwossen M, Summers C. Influence of intra- and interfraction motion on planning target volume margin in liver stereotactic body radiation therapy using breath hold. Adv Radiat Oncol. 2021;6(1). doi:doi:10.1016/j.adro.2020.10.023.
- Seppenwoolde Y, Shirato H, Kitamura K. Precise and real-time measurement of 3D tumor motion in lung due to breathing and heartbeat, measured during radiotherapy. Int J Radiat Oncol Biol Phys. 2002;53(4):822-834. doi:doi:10.1016/s0360-3016(02)02803-1.
- George R, Chung T, Vedam S. Audio-visual biofeedback for respiratory-gated radiotherapy: impact of audio instruction and audio-visual biofeedback on respiratory-gated radiotherapy. Int J Radiat Oncol Biol Phys. 2006;65(3):924-933. doi:doi:10.1016/j.ijrobp.2006.02.035.
- Vedam S, Keall P, Kini V, Mohan R. Determining parameters for respiration-gated radiotherapy. Med Phys. 2001;28(10):2139-2146. doi:doi:10.1118/1.1406524.
- Goossens S, Senny F, Lee J, Janssens G, Geets X. Assessment of tumor motion reproducibility with audio-visual coaching through successive 4D CT sessions. J Appl Clin Med Phys. 2014;15(1):4332. doi:doi:10.1120/jacmp.v15i1.4332.
- Lens E, Gurney-Champion O, Tekelenburg D. Abdominal organ motion during inhalation and exhalation breath-holds: pancreatic motion at different lung volumes compared. Radiother Oncol. 2016;121(2):268-275. doi:doi:10.1016/j.radonc.2016.09.012.
- Holland A, Goldfarb J, Edelman R. Diaphragmatic and cardiac motion during suspended breathing: preliminary experience and implications for breath-hold MR imaging. Radiology;209(2):483-489. doi:doi:10.1148/radiology.209.2.9807578.
- Parkes M. Breath-holding and its breakpoint. Exp Physiol. 2006;91:1-15. doi:doi:10.1113/expphysiol.2005.031625.
- Parkes M, Green S, Kilby W. The feasibility, safety and optimization of multiple prolonged breath-holds for radiotherapy. Radiother Oncol. 2019;141:296-303. doi:doi:10.1016/j.radonc.2019.06.014.
- Parkes M, Green S, Stevens A. Safely prolonging single breath-holds to >5 min in patients with cancer; feasibility and applications for radiotherapy. Br J Radiol. 2016;89(1063). doi:doi:10.1259/bjr.20160194.
- Parkes M, Green S, Cashmore J, Ghafoor Q, Clutton-Brock T. Shortening the preparation time of the single prolonged breath-hold for radiotherapy sessions. Br J Radiol. 2022;95(1130). doi:doi:10.1259/bjr.20210408.
- Towell V, Gysen K, Cross S, Kk Low G. Efficacy of preoxygenation administration in volunteers, in extending the end-expiration breath-hold duration for application to abdominal radiotherapy. Tech Innov Patient Support Radiat Oncol. 2023;26. doi:doi:10.1016/j.tipsro.2023.100208.
- Parkes M, Green S, Stevens A, Clutton-Brock T. Assessing and ensuring patient safety during breath-holding for radiotherapy. Br J Radiol. 2014;87(1043). doi:doi:10.1259/bjr.20140454.
- Macefield G, Burke D. Paraesthesiae and tetany induced by voluntary hyperventilation. Increased excitability of human cutaneous and motor axons. Brain. 1991;114 (Pt 1B):527-540. doi:doi:10.1093/brain/114.1.527.
- O’Brien J. Absorption atelectasis: incidence and clinical implications. AANA J. 2013;81(3):205-208.
- Kim H, Min J, Lee J. The effect of oxygen concentration on atelectasis formation during induction of general anesthesia in children: a prospective randomized controlled trial. Paediatr Anaesth. 2021;31(12):1276-1281. doi:doi:10.1111/pan.14304.
- Rothen H, Sporre B, Engberg G. Prevention of atelectasis during general anaesthesia. Lancet. 1995;345(8962):1387-1391. doi:doi:10.1016/s0140-6736(95)92595-3.
- Edmark L, Kostova-Aherdan K, Enlund M, Hedenstierna G. Optimal oxygen concentration during induction of general anesthesia. Anesthesiology. 2003;98(1):28-33. doi:doi:10.1097/00000542-200301000-00008.
- Pingitore A, Gemignani A, Menicucci D. Cardiovascular response to acute hypoxemia induced by prolonged breath holding in air. Am J Physiol Heart Circ Physiol. 2008;294(1):H449-55. doi:doi:10.1152/ajpheart.00607.2007.
- Heusser K, Dzamonja G, Tank J. Cardiovascular regulation during apnea in elite divers. Hypertension. 2009;53(4):719-724. doi:doi:10.1161/HYPERTENSIONAHA.108.127530.
Disclosures
The authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere. A portion of this data (from the first 10 treated patients with the described technique) was presented in poster form at the ASTRO 2021 Annual Meeting. Full citation as follows: Schneider CS, Shen S, Fiveash JB, Jacob, R. A practical method to prolong expiratory breath holds for abdominal stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys. 2021;111(3)Suppl:e543.
Citation
. A Practical Method to Prolong Expiratory Breath Holds for Abdominal Stereotactic Body Radiation Therapy. Applied Radiation Oncology. 2023;12(3):34-42. doi:10.37549/ARO-D-23-00012.