A Just Enough Interaction Segmentation Tool Improves Consistency and Efficiency for Radiation Therapy Contouring of Meningiomas
Images



Abstract
Purpose: To develop a user-friendly segmentation tool requiring minimal expert interaction to reduce physician workload and improve reproducibility.
Methods: Sixteen treated meningiomas cases were manually contoured, then contoured using the JEI-LOGISMOS (just-enough-interaction layered optimal graph image segmentation for multiple objects and surfaces) segmentation tool by two central nervous system experts. Cases were randomly displayed for both manual and JEI-LOGISMOS analyses in several sessions to avoid bias. Segmentation accuracy indices were determined as continuous variables: mean (± standard deviations) or median (and interquartile ranges [IQR]) where appropriate. Computer-analysis accuracy was evaluated using point-wise 3-dimensional (3D) surface distance errors and volumetric linear regression. To assess reproducibility, the Dice coefficient along with 3D relative volume difference (RVD) were obtained. To evaluate the efficiency of the automated method, time required for automated contouring with JEI and manual contouring was compared using Wilcoxon signed-rank test.
Results: Our 3D LOGISMOS segmentations using JEI with both experts achieved subvoxel precision (voxel size ~1 mm) for meningioma tumor surfaces (JEI signed error: 0.86 ± 1.82 mm for expert 1, 0.24 ± 1.26 mm for expert 2) and provided accurate volume measurements in comparison to manual contouring (volume regression: R2 = 0.93, P < 0.001 for expert 1, R2 = 0.96, P < 0.001 for expert 2). The interobserver variability of automated contouring showed better reproducibility compared with manual contouring (Dice: 87.4% vs 83.6%; RVD: -1.1% vs 14.9%). Median time required for contouring cases was significantly reduced for both experts (-204 seconds per case, P = 0.01, 46.5% faster for expert 1 and -228 seconds per case, P = 0.04, 35.8% faster for expert 2).
Conclusion: Automated contouring using a JEI approach following the automated 3D LOGISMOS segmentation improves reproducibility and efficiency of contouring for meningiomas. Volumes obtained using manual tracing and JEI-LOGISMOS were highly comparable.
Meningiomas are benign neoplasms that originate from the arachnoid layer of the meninges surrounding the brain. They are the fourth most common primary brain tumor with a female predilection (2:1) associated with hormonal stimulation of these tumors.1,2 Radiographically, meningiomas appear as conspicuous contrast-enhancing masses on computed tomography (CT) and MRI with relatively smooth borders, and are attached to dural surfaces surrounding brain. They may be calcified and occasionally can invade or remodel the surrounding bone. They can present with neurologic symptoms generally caused by mass effect on adjacent brain structures rather than invasion into normal central nervous system tissue. Meningioma mass effect symptoms are usually dictated by location and can include headaches and seizures, as well as focal neurologic symptoms such as weakness, numbness, pain or cranial nerve deficits. They may also rarely result in global neurologic deterioration from hydrocephalus or extreme mass effect.1 Unlike benign meningiomas, atypical or malignant variants can invade surrounding brain tissues directly.3 Meningiomas are also frequently found incidentally on scans and, therefore, may have no symptoms.
Surgical resection is the standard treatment for meningiomas and results in cure in more than 90% of cases when complete excision is achieved.1 However, when meningiomas are around or directly involve critical neurologic structures and cranial nerves of the skull base, surgical resection can result in profound morbidity. Direct involvement of dural venous sinuses or the cavernous sinus may complicate surgical approaches.4,5 Hence, when meningiomas are poorly resectable or the patient is a poor candidate for resection, radiosurgery has been shown to achieve long-term control rates approaching surgical excision outcomes.6 Radiosurgery is a more convenient outpatient procedure that may be particularly well-suited for elderly or infirm patients. Radiation is also used after incomplete resection to prevent tumor regrowth or if meningiomas are atypical or malignant and, thus, have greater potential for recurrence.7 Therefore, radiation therapy and radiosurgery are important therapeutic modalities for these more difficult-to-manage meningiomas and are associated with control rates between 80% to 95% depending on the size and subtype of meningioma.
Accurate identification of the tumor target and anatomy through manual contouring is a critical step in planning radiosurgery or radiation therapy. Since most meningiomas are benign, the gross tumor volume (GTV) effectively defines the target. Treatments generally use minimal margins around meningioma targets and radiation is most often stereotactically delivered (single or multifraction) or treatments use maximal precision image guidance for daily fractionation. Currently, radiation oncologists manually identify and define target lesions in a treatment planning system based on MR imaging in the vast majority of cases. Manually defined tumor targets require significant physician time and effort. Furthermore, many fine points of tumor growth along dural surfaces may be overestimated or underestimated based on physician interpretation of the MR. Even the same physician contouring the same lesion on different occasions will show inconsistency in these subtle interpretations while contouring.8-10 When lesions are complex in shape and location, there is increased variability in target definition among different physicians as well as with the same physician. Additionally, larger and more complex lesions may take the physician an hour or more to contour manually and are also usually less consistently well reproduced.
The development of an automated segmentation tool has the potential to both improve the consistency of contouring between and among different radiation oncologists and improve the efficiency of contouring for radiation therapy planning. While MR is used for this contouring, these MR images are generally fused to a contrasted CT for dose calculation. Primary contouring tasks occur on the MR data set. Over the past several years, algorithms, including atlas-based contouring, machine learning and shape/appearance methods became the basis for the development of many institution-specific segmentation tools.11 These tools have shown the potential to improve the workflow of head and neck, breast, lung and abdomen cancer radiation therapy planning.12-16
In this study, we present a rapid, robust and highly accurate semi-automatic tumor lesion contouring solution based on layered optimal graph image segmentation for multiple objects and surfaces (LOGISMOS) with an optional just enough interaction (JEI) postsegmentation editing of target surfaces.17 The algorithm works through identification of a lesion bounded by a spherical volume of interest in which to establish the contour. Using the imaging features or central identified tumor, the algorithm performs a graph-search optimization for surfaces to identify the surface of the lesion where the maximum change in intensity occurs. In performing this function, a change at one point may be propagated to redefine the surface in 3 dimensions (3D) and thereby avert the requirement for manual slice-by-slice editing of contours. An additional motivation for developing this tool is the daily application of imaging for treatment set-up accuracy using MR linear accelerator (MR-linac) devices. The ability to rapidly adapt contours in 3D planes simultaneously will be critical for on-line segmentation that is essential for treatment plan adaptation at several anatomic sites. While not directly applied to this meningioma model, the method may be applied to other images with or without contrast. The approach has substantial capability to address these needs for rapid adaptation of these noncontrasted images although this is not the primary goal of this report. Select optimization of the algorithm for specific applications will likely be needed. Our current report addresses the capability in the complex model of skull base, highly irregular meningiomas with contrast MR. The development of a framework for this work through a JEI-LOGISMOS method can fully leverage this new MR-linac potential.
Developed at the Iowa Institute for Biomedical Imaging, the LOGISMOS segmentation framework facilitates highly efficient multidimensional, multilayered, and multiobject optimum graph-based segmentation and surface editing on image data from various modalities (CT, MR, ultrasound, optical coherence tomography [OCT], etc.) with the current report using MR. LOGISMOS has previously demonstrated successful applications in cardiology, ophthalmology, neuroscience, pulmonology, radiation oncology and other areas.18-22
Methods
Patient Selection and Task Randomization
Sixteen patients treated for radiographically presumed benign meningioma with radiation therapy at the authors’ institution were enrolled in this IRB-approved retrospective imaging study. T1-weighted MRI data were used. Each subject’s MR image was contoured twice by each of three physicians, once using manual contouring (Monaco, Elekta) and once using JEI-LOGISMOS semi-automatic segmentation. A total of 32 contouring tasks per physician were generated for this patient cohort. Manual or semi-automated segmentation tasks were assigned to each physician in random order (manual vs semi-automated). Manual and semi-automatic contouring sessions for each case by the same radiation oncologist were separated temporally to minimize bias from the radiation oncologist’s previous contouring session. After completing sessions, significant agreement was noted in manual contours between two more experienced radiation oncologists specializing in central nervous system (CNS) radiation therapy compared with their non-CNS specializing colleague. The manually traced contours from this non-CNS specializing physician were not used for results reporting, and related findings are presented in the discussion.
Overview of LOGISMOS and JEI-LOGISMOS Algorithms
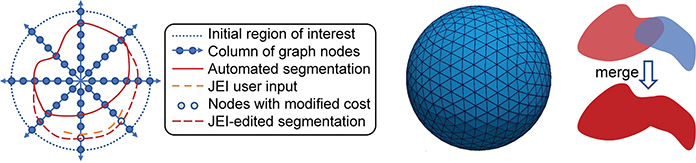
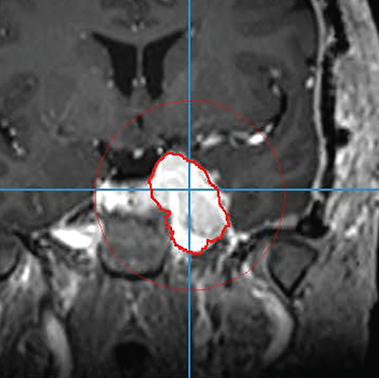
The automated LOGISMOS method is initialized by the physician interactively placing a sphere encompassing the tumor. Starting from the center of the sphere, columns of graph nodes are constructed. Each graph node is assigned a cost that represents the unlikeness of the node being on the object boundary. The LOGISMOS segmentation finds the optimal set of graph nodes, one per column, with minimum total cost, thus defining the object surface. During the search for the optimal solution, prior knowledge such as the shape and anatomy of the target constrains the segmentation so that it is the one with minimum total cost among all possible solutions that meet the constraints. If needed, errors in the automated segmentation may be corrected by the user’s interaction with the 3D LOGISMOS algorithm rather than by slice-by-slice manual retracing (Figures 1A, 1B). This process uses our JEI approach that considers the expert hints pointing to the correct boundary locations to modify the underlying segmentation cost functions as needed and searches for a new optimal solution in 3D under the modified cost. If a significant amount is felt lacking an alternative, the JEI approach is used to identify the center of the area missed and a sphere is placed over this area subsequently, followed by repeating the process above. Once this second overlapping area is identified, a merge of the contours can be accomplished (Figure 1C). The completed contour of the complex skull base meningioma is shown in Figure 2.
Manual Contouring
Manual contouring was performed in a Monaco Treatment Planning System v5.19.03d (Elekta). The T1-weighted contrast-enhanced MRI for each subject was loaded in the contouring session. The participating radiation oncologist used the manual contouring tools, such as a paint brush or polygon generation tool, as they would clinically, on each axial slice of the MRI image. The manual contouring was self-timed using a watch or timed by a colleague using a stopwatch; timing started when the radiation oncologist made the first mouse click on the image, and ended when the radiation oncologist declared the contouring concluded and clicked the “save” button. The manual contours were exported in DICOM format as local files for further processing and analysis.
Semi-automatic Contouring
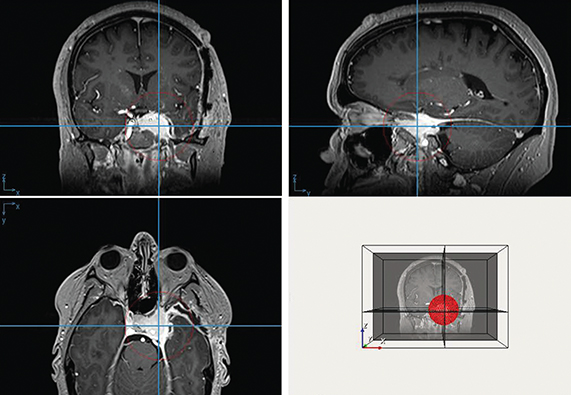
Semi-automatic contouring was performed in RadOnco Analyzer, an in-house software based on the JEI-LOGISMO) algorithm as described above. The preprocessed T1-weighted contrast-enhanced MRI for each subject was loaded in this software. The participating radiation oncologist searched the approximate volumetric center of meningioma using cross hairs, dropped a “Center” point, set the maximum extent of contour search region using a sphere visible in three planar views (axial, sagittal and coronal) (Figure 2A), and requested the software to automatically find the boundary of the meningioma (Figure 2B). JEI editing was then performed if necessary (Figure 2C). If additional regions were needed as alternatives, this was also included in the time. The timing of the automatic contouring was also recorded by stopwatch: timing started after the radiation oncologist clicked on the cross hairs to find the “Center” point and stopped when the radiation oncologist clicked the “Finish” button. The contours were automatically saved in an analyzable format by the software.
Continuous variables are described as mean (± standard deviations) or median (and interquartile ranges [IQR]) where appropriate. To evaluate computer-analysis accuracy and reproducibility, point-wise distance errors of 3D surfaces and Dice coefficient along with relative volume difference (RVD) of 3D volumes were reported and compared with manual contouring by paired t-test. To evaluate efficiency of the semi-automated method, times required for automated contouring with JEI-LOGISMOS and manual contouring were compared using a mixed effects regression analysis with random effects for experts and patients. The R coding environment was employed for statistical computing, while a P-value of 0.05 denoted statistical significance.
Results
Accuracy and Reproducibility of Automated Contouring
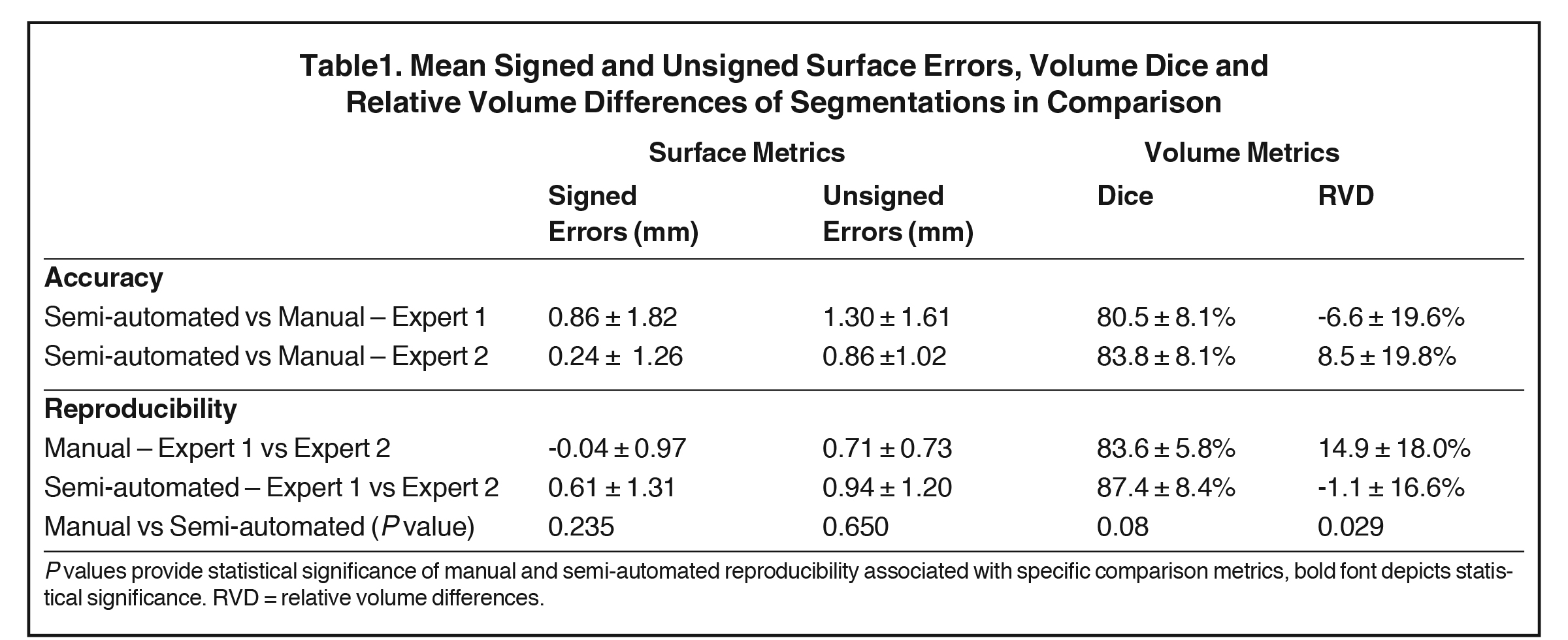
Both surface-positioning and volume interobserver comparisons of obtained segmentations (manual vs semi-automated) are given in Table 1. Segmentations of cases using semi-automated contouring achieved subvoxel precision on average (voxel size ~1 mm) for surface differences compared with manual contouring. When pairwise comparing the reproducibility of manual and semi-automated segmentations, surface-positioning differences showed no statistical differences (P > 0.2). The same pairwise comparisons of Dice coefficients of volumetric agreement showed less strong but still no statistical differences (P = 0.08). There was, however, a statistically significant improvement in relative volume reproducibility assessed as RVD, showing the superiority of our semi-automated approach (14.9 ± 18.0 % vs 1.1 ± 16.6 %, P < 0.03).
Efficiency of Automated Contouring
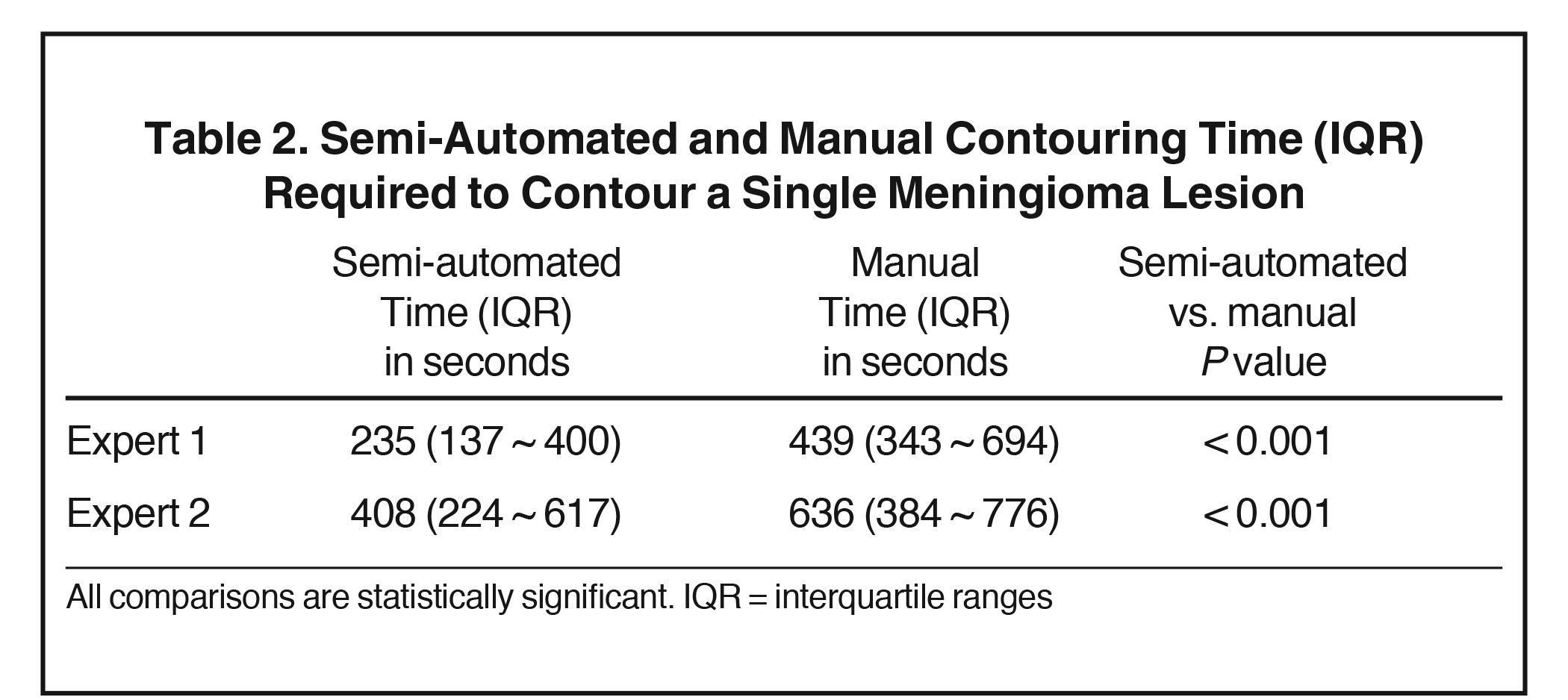
The required time to contour a single meningioma using our automated system with JEI editing and a Monaco Treatment Planning System are given in Table 2. The semi-automated contouring process was significantly faster than manual contouring for both experts (P < 0.001). To quantify the effort associated with the semi-automated method, the median numbers of JEI edits used by the two expert analysts were 1.5 and 13.6, respectively – correlating with the longer analysis times of both the manual and semi-automated segmentations (but not better segmentation results) of Expert 2 compared with Expert 1.
Discussion
Application of semi-automatic contouring tools can aid radiation oncologists in radiation therapy treatment planning by improving consistency and efficiency of contouring tumor lesions. In this study, we demonstrate the utility of the LOGISMOS-based segmentation tool with JEI postprocessing in contouring complex meningiomas in T1-weighted contrast-enhanced MRI.
Precision and Accuracy
The gold standard to measure tumor contouring accuracy would be comparison with and confirmation by pathology. This, however, is not feasible in most cases, particularly in patients with CNS tumors such as meningiomas. In similar studies evaluating autosegmentation solutions, the consensus of manually segmented volumes by expert physicians is defined as the ground truths and is used for validation.11 To evaluate the accuracy and efficiency of our LOGISMOS-JEI semi-automatic segmentation tool, the consensus of the manual contours of experienced radiation oncologists (Expert 1 and 2) is built for each segmentation task and used as ground truth for each case to validate the respective autosegmentations. Our study compared the accuracy and reproducibility of semi-automated segmentation vs manual segmentation of a cohort with variably complex skull-based and convexity meningiomas. Even though the morphologies of the 16 analyzed lesions were complex and diverse, our segmentation tool remained robust, achieving surface positioning errors and Dice agreements indistinguishable between manual tracing and semi-automated analysis. Hence semi-automated JEI contours produced were clinically identical to that of standard-of-care manually produced contours. At the same time, the reproducibility of the semi-automated approach outperformed that of manual tracing when comparing relative volume differences. This demonstrates an ability of the semi-automated segmentation tool to improve inter-observer variability in RVD in a case when tumor volume delineation is performed by different radiation oncologists. This feature of the semi-automated approach will lead to more consistent treatment planning and potentially improved patient outcomes.
We believe we were able to achieve such accuracy and precision due to our base LOGISMOS algorithm coupled with the JEI algorithm postprocessing. LOGISMOS guarantees a volumetrically optimal solution with respect to the employed cost function in 3D (or nD in general) will be produced. The associated JEI steps provide an intuitive and efficient mechanism that allows the user to interact with the LOGISMOS algorithm and thus affect the segmentation result in a volumetric fashion rather than in a slice-by-slice fashion, thus yielding the adjudicated surfaces with minimal interaction efforts.
Improvements in automatic segmentation to delineate meningiomas for radiation therapy planning have not been widely reported. There have been different groups that investigated automatic segmentation algorithms for meningioma detection and recognition. In Hsieh et al a meningioma automated segmentation tool was developed to diagnose brain tumors using MRI images. This tool used an algorithm integrating fuzzy-c-mean and region-growing technique.23 Similarly in Laukamp et al the authors adapted a deep learning model used for glioblastoma tumor detection in MRI to detect meningiomas.24,25 Their results yielded similar segmentation accuracy statistics as our segmentation tool in this study.
Efficiency
Manual contouring for radiation treatment planning is a time-consuming process that can bottleneck therapy delivery.11 The development of a semi-automated tool that can produce accurate segmentations can help radiation oncologists drastically reduce the time spent contouring. The two experts who performed the contouring had different levels of contouring experience as evidenced by their different average contouring time. Using the automated segmentation tool, both individuals reliably reduced the average amount of time spent contouring while improving reproducibility, demonstrating the tool’s consistent ability to decrease the time to segment a tumor. The number of specific types of contour-editing instances (surface point vs a region needing an alternative JEI segmentation for a region that was felt to be missed) using the JEI approach were not specifically tracked as there was a clear decrease in the total time spent on each segmentation task compared with manual segmentation. All times included the entire process of editing. In future studies, it is would be interesting to track JEI contour editing occurrences and evaluate the use of JEI editing in different tumor pathologies and different imaging modalities.
When compared to manual contouring, consistent but varying degrees of reduction in segmentation time were reported by other organ-specific automated segmentation tools.14,15,26-30 As mentioned, while automatic segmentation tools developed for meningioma detection by other groups have been reported, the efficiency of these tools was not specifically studied. In a separate but similar experiment, Oguz et al investigated the efficiency of the LOGISMOS algorithm in automated segmentation of rat brains and found drastically decreased segmentation time compared to neural network-based methods or atlas-based methods.20
Advantages
The algorithm in the segmentation tool presented in this study is packaged in in-house software called RadOnco Analyzer. This software is specifically designed and optimized for radiation oncologists to improve radiation therapy planning. It features an intuitive user interface that allowed users to easily navigate the contouring process despite varied levels of experience. Another advantage of our algorithm is it takes full advantage of the increased use of MR-linacs in radiation treatment delivery. MR produces images with high soft-tissue contrast that particularly lends itself to tumor lesion identification.11 Because we are studying meningiomas in the skull base, convexity, soft-tissue contrast and anatomy variability are less affected by daily deformities caused by a patient’s position during treatment. Lastly, the incorporation of JEI volumetric editing with our LOGISMOS auto-segmentation tool presents a distinct advantage over other segmentation tools for real time adaptation although this specific premise is not tested in the current study. While many commercial and institution-specific segmentation tools perform efficiently and accurately, most also require significant additional time to edit the contours separately and manually after segmentations are completed by the algorithm.14,31 With JEI-LOGISMOS, fast and intuitive editing occurs at the same time with each segmentation and further complements the time saved by automatic segmentation.
Impact on Performance of Less Experienced Analysts
As mentioned earlier, one of the recruited analysts (Expert 3) was a non-CNS specializing radiation oncologist (also less experienced) and the analyses were not included in the above summarizing results due to the observed tracing differences of manual analysis when compared to more experienced CNS specialist colleagues. We have, however, compared the level of agreement of Expert 3 when using fully manual and semi-automated analysis with the analyses provided by Experts 1 and 2. Table 3 shows that our semi-automated approach applied by the less expert radiation oncologist had statistically significantly improved agreement with more expert contours compared with manual efforts. These metrics were improved using the semi-automated method with analyses using Dice or RVD metrics for comparison (P < 0.001 and P < 0.02, respectively). This is an important secondary outcome of our study, demonstrating that our JEI-LOGISMOS semi-automated analysis tool is likely to have a highly positive impact on accuracy as well as reproducibility of volumetric analyses performed by less expert colleagues.
Limitations
While our analysis achieved statistical significance, we believe we can continue to improve the validity of our study by increasing the sample size of our cohort of meningioma patients as well as adding the experience of other physicians. Potential bias in case selection and potential relative benefit for skull-base lesions vs other lesions could also be considered. In addition, the issue of applying in-house software in a noncommercial non-FDA approved tool will require additional levels of validation for broad applicability and to achieve full clinical relevance. The application of specific tools to specific disease sites and imaging modalities including multiparametric imaging is also a challenge for radiation therapy planning. Treatment planning software currently does not comprehensively address the needs for improved automated and semi-automated routines for contouring and the best method to integrate such tools is poorly defined.
The proposed approach is not free of real-world technical limitations resulting in logistical difficulty to incorporate a tool like this in an established clinical workflow. Incorporation in any established clinical workflow involves many steps and the described tool replaces just one of them. As a result, the JEI-LOGISMOS semi-automated segmentation must be properly interfaced with the adjacent modules of the workflow. Such a step, however, requires cooperation by manufacturers of the respective modules and workflows potentially impacting regulatory approvals of the entire workflow pipeline.
Future Work
Application of our semi-automated algorithms for treatment planning tasks with physician supervision is an important next step furthering the implementation of these tools. We believe the MR-linac environment is ideal for this application since the need for increased efficiency and consistency is compounded by the daily requirements of treatment modifications. Expansion of the patient cohort and testing group of physicians will also be important. While the LOGISMOS framework has been successfully used to segment structures known to have poor contrast uptake including knee cartilage and the basal ganglia, it would be important in the future to evaluate our algorithm in a noncontrast MRI environment.
Conclusion
Automated contouring using a JEI approach following the automated LOGISMOS segmentation markedly improves reproducibility and efficiency of contouring for meningiomas. Evidence also suggests that it may positively improve segmentation performance of less-expert analysts. Our study presents a user-friendly and versatile tool with a robust base algorithm allowing radiation oncologists to efficiently plan radiation treatment while improving accuracy.
References
- Marosi C, Hassler M, Roessler K, et al. Meningioma. Crit Rev Oncol Hematol. 2008;67(2):153-171. doi:10.1016/j.critrevonc.2008.01.010
- Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
- Garzon-Muvdi T, Yang W, Lim M, Brem H, Huang J. Atypical and anaplastic meningioma: outcomes in a population based study. J Neurooncol. 2017; 133(2):321-330. doi:10.1007/s11060-017-2436-6
- Fariselli L, Biroli A, Signorelli A, Broggi M, Marchetti M, Biroli F. The cavernous sinus meningiomas’ dilemma: Surgery or stereotactic radiosurgery? Rep Pract Oncol Radiother. 2016;21(4):379-385. doi:10.1016/j.rpor.2015.05.002
- Walsh MT, Couldwell WT. Management options for cavernous sinus meningiomas. J Neurooncol. 2009; 92(3):307-316. doi:10.1007/s11060-009-9824-5
- Lee JYK, Niranjan A, McInerney J, Kondziolka D, Flickinger JC, Lunsford LD. Stereotactic radiosurgery providing long-term tumor control of cavernous sinus meningiomas. J Neurosurg. 2002;97(1):65-72. doi:10.3171/jns.2002.97.1.0065
- Klinger DR, Flores BC, Lewis JJ, Barnett SL. The treatment of cavernous sinus meningiomas: evolution of a modern approach. Neurosurg Focus. 2013;35(6):E8. doi:10.3171/2013.9.FOCUS13345
- Vinod SK, Jameson MG, Min M, Holloway LC. Uncertainties in volume delineation in radiation oncology: a systematic review and recommendations for future studies. Radiother Oncol. 2016;121(2):169-179. doi:10.1016/j.radonc.2016.09.009
- Weltens C, Menten J, Feron M, et al. Interobserver variations in gross tumor volume delineation of brain tumors on computed tomography and impact of magnetic resonance imaging. Radiother Oncol. 2001;60(1):49-59. doi:10.1016/S0167-8140(01)00371-1
- Growcott S, Dembrey T, Patel R, Eaton D, Cameron A. Inter-observer variability in target volume delineations of benign and metastatic brain tumours for stereotactic radiosurgery: results of a national quality assurance programme. Clin Oncol. 2020;32(1):13-25. doi:10.1016/j.clon.2019.06.015
- Sharp G, Fritscher KD, Pekar V, et al. Vision 20/20: perspectives on automated image segmentation for radiotherapy. Med Phys. 2014;41(5):050902. doi:10.1118/1.4871620
- Guo Z, Guo N, Gong K, Zhong S, Li Q. Gross tumor volume segmentation for head and neck cancer radiotherapy using deep dense multi-modality network. Phys Med Biol. 2019;64(20):205015. doi:10.1088/1361-6560/ab440d
- Tong Y, Udupa JK, Wu X, et al. Hierarchical model-based object localization for auto-contouring in head and neck radiation therapy planning. Proc SPIE. 2018;10578. doi:10.1117/12.2294042
- Mitchell RA, Wai P, Colgan R, Kirby AM, Donovan EM. Improving the efficiency of breast radiotherapy treatment planning using a semi‐automated approach. J Appl Clin Med Phys. 2016;18(1):18-24. doi:10.1002/acm2.12006
- Liang F, Qian P, Su K-H, et al. Abdominal, multi-organ, auto-contouring method for online adaptive magnetic resonance guided radiotherapy: an intelligent, multi-level fusion approach. Artif Intell Med. 2018;90:34-41. doi:10.1016/j.artmed.2018.07.001
- Li D, Liu L, Chen J, et al. Augmenting atlas-based liver segmentation for radiotherapy treatment planning by incorporating image features proximal to the atlas contours. Phys Med Biol. 2016;62(1):272-288. doi:10.1088/1361-6560/62/1/272
- Yin Y, Zhang X, Williams R, Wu X, Anderson DD, Sonka M. LOGISMOS – Layered optimal graph image segmentation of multiple objects and surfaces: cartilage segmentation in the knee joint. IEEE Trans Med Imaging. 2010;29(12):2023-2037. doi:10.1109/TMI.2010.2058861
- Kashyap S, Oguz I, Zhang H, Sonka M. Automated segmentation of knee MRI using hierarchical classifiers and just enough interaction based learning:
- data from Osteoarthritis Initiative. Med Image Comput Comput-Assist Interv. 2016;9901:344-351. doi:10.1007/978-3-319-46723-8_40
- Chen Z, Pazdernik M, Zhang H, et al. Quantitative 3D analysis of coronary wall morphology in heart transplant patients: OCT-assessed cardiac allograft vasculopathy progression. Med Image Anal. 2018;50:95-105. doi:10.1016/j.media.2018.09.003
- Oguz I, Zhang H, Rumple A, Sonka M. RATS: Rapid automatic tissue segmentation in rodent brain MRI. J Neurosci Methods. 2014;221:175-182. doi:10.1016/j.jneumeth.2013.09.021
- Sonka M, Abràmoff MD. Quantitative analysis of retinal OCT. Med Image Anal. 2016;33:165-169. doi:10.1016/j.media.2016.06.001
- Zhang H, Lee K, Chen Z, Kashyap S, Sonka M. Chapter 11 – LOGISMOS-JEI: Segmentation using optimal graph search and just-enough interaction. In: Zhou SK, Rueckert D, Fichtinger G, eds. Handbook of Medical Image Computing and Computer Assisted Intervention. Academic Press; 2020:249-272. doi:10.1016/B978-0-12-816176-0.00016-8
- Hsieh TM, Liu Y-M, Liao C-C, Xiao F, Chiang I-J, Wong J-M. Automatic segmentation of meningioma from non-contrasted brain MRI integrating fuzzy clustering and region growing. BMC Med Inform Decis Mak. 2011;11(1):54. doi:10.1186/1472-6947-11-54
- Laukamp KR, Pennig L, Thiele F, et al. Automated meningioma segmentation in multiparametric MRI: comparable effectiveness of a deep learning model and manual segmentation. Clin Neuroradiol. Published online February 14, 2020. doi:10.1007/s00062-020-00884-4
- Laukamp KR, Thiele F, Shakirin G, et al. Fully automated detection and segmentation of meningiomas using deep learning on routine multiparametric MRI. Eur Radiol. 2019;29(1):124-132. doi:10.1007/s00330-018-5595-8
- Kosmin M, Ledsam J, Romera-Paredes B, et al. Rapid advances in auto-segmentation of organs at risk and target volumes in head and neck cancer. Radiother Oncol. 2019;135:130-140. doi:10.1016/j.radonc.2019.03.004
- Simmat I, Georg P, Georg D, Birkfellner W, Goldner G, Stock M. Assessment of accuracy and efficiency of atlas-based autosegmentation for prostate radiotherapy in a variety of clinical conditions. Strahlenther Onkol. 2012;188(9):807-815. doi:10.1007/s00066-012-0117-0
- Lustberg T, Soest J van, Gooding M, et al. Clinical evaluation of atlas and deep learning based automatic contouring for lung cancer. Radiother Oncol. 2018; 126(2):312-317. doi:10.1016/j.radonc.2017.11.012
- Men K, Zhang T, Chen X, et al. Fully automatic and robust segmentation of the clinical target volume for radiotherapy of breast cancer using big data and deep learning. Phys Med. 2018;50:13-19. doi:10.1016/j.ejmp.2018.05.006
- Haas B, Coradi T, Scholz M, et al. Automatic segmentation of thoracic and pelvic CT images for radiotherapy planning using implicit anatomic knowledge and organ-specific segmentation strategies. Phys Med Biol. 2008;53(6):1751-1771. doi:10.1088/0031-9155/53/6/017
- Meillan N, Bibault J-E, Vautier J, et al. Automatic intracranial segmentation: is the clinician still needed? Technol Cancer Res Treat. 2018;17. doi:10.1177/1533034617748839
Citation
W L, Z C, H Z, D W, BJ S, K P, M S, M S, JM B. A Just Enough Interaction Segmentation Tool Improves Consistency and Efficiency for Radiation Therapy Contouring of Meningiomas. Appl Radiat Oncol. 2020;(4):31-38.
December 24, 2020