Radiation Therapy in the Treatment of Plantar Fibromatosis – Two Clinical Cases of Recurrent Disease
Images

Case Summary
Case 1
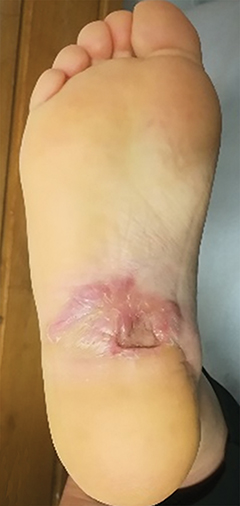
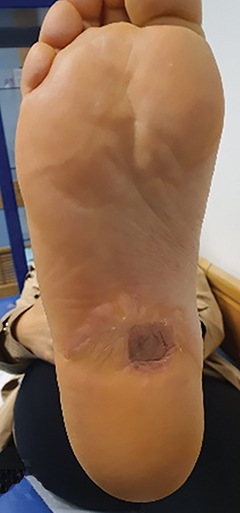
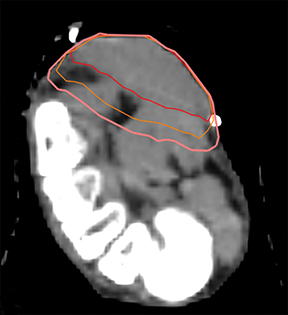
A 49-year-old woman diagnosed with right plantar fibromatosis underwent surgery in 2010. She presented with recurrence in 2018 and was treated with local betamethasone injections due to pain, without improvement. In December 2019, she underwent surgery with gross total excision and closure with skin graft. The postoperative course was complicated with dehiscence of the graft and closure by secondary intention. In September 2020, the patient presented with pain that impaired walking and had progressive growth of the lesion on the right plantar region (Figure 1A). She was referred to the radiation therapy (RT) department for evaluation.
Case 2
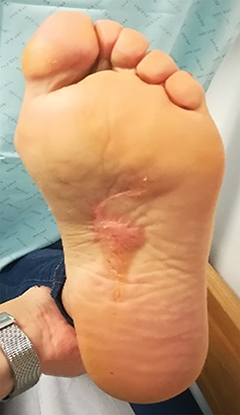
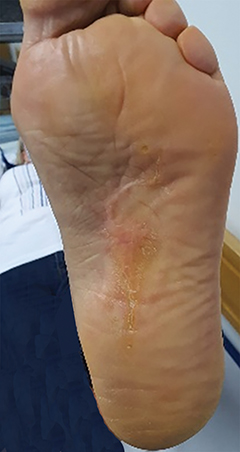
A 63-year-old woman diagnosed with left plantar fibromatosis underwent local excisions in 2017, 2018 and 2019. The last surgical excision revealed focal involvement of the lateral and deep margins by the lesion. In September 2020, the patient presented with worsening of the pain (patient rated as 10/10), without relief with physiotherapy. She was referred to the RT department for evaluation (Figure 2A).
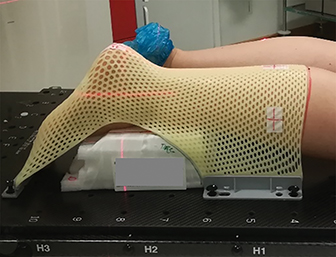
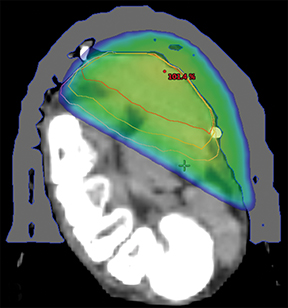
A planning computed tomography (CT) was done in both patients in a ventral decubitus position with adequate immobilization using a thermoplastic mask (Figures 3A and 3B) connected to a base plate. Under the treated foot was a foam support individually adapted to the patient’s anatomy for stability and comfort. External marks were placed on the mask for laser alignment. Both treatments were done with 6-Mv photons and a 5-mm bolus covering the plantar region, with a gross tumor volume (GTV) to clinical target volume (CTV) margin of 5 to 10 mm, and CTV to planning target volume (PTV) margin of 3 mm (Figure 4). The CTV was delineated to encompass all possible disease, both microscopic disease and disease that might have been mistaken for fibrosis and postsurgical changes. In an ideal planning, with MRI images, or in a patient without previous surgery, the planning would be done with only a margin from the GTV to the PTV. A total dose of 30 Gy was prescribed in 2 courses of 15 Gy/ 5 fractions/ 1 week separated by 6 weeks. The patients underwent treatment without relevant side effects during treatment. However, the first patient reported acute plantar pain 3 weeks after completing the total treatment, which improved after a few days without the need for pain treatment. On the first appointment after 3 months of treatment, both presented with almost complete resolution of the initial complaints of pain and walking impairment, and diminution of the visible lesions. At follow-up appointments 6 months after the end of treatment, both patients presented with maintenance of clinical response, without worsening of pain, and stable lesions with drier skin (Figures 1B and 2B).
Discussion
Plantar fibromatosis (PF), also known as Morbus Ledderhose or Ledderhose disease, is a rare benign hyperproliferative disorder of the plantar fascia. It is histologically and clinically identical to Dupuytren’s disease of the hand, and the two conditions may coexist in up to 25% to 35% of cases.1,3-5 The underlying cause is unknown, but there is an association with nicotine and alcohol abuse, diabetes mellitus, anti-epileptic use and genetic factors.1,4 Additionally, a hereditary role has been suggested as familial occurrence of the disease has been reported.2 It appears to be more common among Caucasians and some reports suggest that it affects males up to twice as often. Bilateral disease can be observed in approximately 25% of cases.3-5
The pathogenesis of PF can be divided into three phases: 1) the initial proliferative phase, with proliferation of fibroblasts and development of nodules and cords; 2) the involutional phase, with differentiation of fibroblasts into myofibroblasts; and 3) the residual phase, with a predominance of collagen fibers, in which the normal type I collagen is replaced by type III collagen.3,6
The symptoms usually start in the third to fourth decade of life,1,4,5 and it presents as single or multiple nodules on the central and medial part of the plantar fascia that may cause tension with pain, and disability in walking and fitting shoes. PF contractures of the toes occur less often.1,3,4
The main conservative options for treatments are symptom-oriented and include physiotherapy, orthotic devices, and local steroid injections. Weight loss may also be beneficial.1,3-5 If symptomatic, surgical treatment is frequently recommended, and may range from local or wide excision with a recommended resection margin of 2 to 3 cm, to subtotal or radical fasciectomy with or without skin grafting.1,3,4 Recurrence rates after surgery are high, and surgery is also associated with significant morbidity such as delayed wound healing, chronic pain, and poor functional outcomes.1,2,4
There are data that report the use and outcomes of RT in the treatment of PF (Table 1), mainly from Europe. In some regions, such as North America, the use of RT for this and other benign disease are generally frowned upon, in part due to fear of litigation, and in part because patients typically have surgical consultations before any others.
One study analyzed retrospectively the results of 25 patients (36 affected feet) following 2 RT courses of 5 x 3 Gy for a total dose of 30 Gy. With a median follow-up of 38 months (range: 12 to 67), none presented with disease progression, and 28 feet had regression of pain and tenderness, with stable symptoms in 8 feet.4,5
A small Dutch retrospective study reviewed the outcomes of 9 patients (11 feet) with PF. In all, the primary treatment was surgery alone, with a total of 26 operations and a recurrence rate of 90%. In the recurrent disease treated with surgery alone, the recurrence rate was 6/9 (67%) and in the patients treated with the combination of surgery and adjuvant RT (total of 60 Gy), the rate was 1/6 (17%). Two patients in the surgery with RT group became dystrophic, with foot pain and impaired walking.1,2
A German multicenter retrospective study reported results from 24 patients (33 feet), treated with 2 RT courses of 5 x 3.0 Gy separated by 6 weeks for a total dose of 30.0 Gy (n = 20) or 2 single fractions of 4.0 Gy on consecutive days, repeated at intervals of 4 weeks to cumulative doses ranging from 24 to 32 Gy (n = 4). Only 2 patients had previously undergone surgery and presented with recurrent disease. All of the others had been prescribed orthotics or oral anti- inflammatory drugs. With a median follow-up of 22.5 months (range: 6 to 76), no clinical progression was observed in the number or size of the lesions or in subjective associated clinical symptoms; 33% showed a complete response, 54.5% a partial response and 12.1% stable disease.3
More recently, a single-institution, prospective, nonrandomized cohort study reported results of 158 patients (270 feet) with symptomatic progressive disease. Of these, 91 patients (136 feet) underwent RT and 67 patients (134 feet) did not. The PTV was defined as palpable disease with a 2 cm margin. The dose delivered was 2 courses of 15 Gy / 5 fractions over 1 week, with a 12-week break between courses, for a total dose of 30 Gy / 10 fractions. With a mean follow-up of 68 months (range: 24 to 160), 92% of the irradiated group had stable clinical gross disease or reduction of the lesion size (complete or partial response) (vs 62% in the nonirradiated [NI] group). Eight percent developed progressive disease (vs 38% in the NI group) and 5% underwent salvage surgery (vs 21% in the NI group). Improvement of symptoms was seen in 79% following RT (vs 19% in the NI group). Acute side effects occurred in 26.5%, with slight erythema and/or dry desquamation (21.3%), a diffuse erythema with areas of moist desquamation (5.0%), and late effects with dryness or fibrosis (16.2%).4
The suggested radiobiological mechanism of RT is based mostly on the inhibition of the proliferation of the fibroblasts and myofibroblasts, known to be the cause of the symptoms and progression of the disease. Thus, the optimal time for the use of RT is in actively progressing disease.3,4,6
The RT target volume should include all of the detectable or palpable gross lesions with a minimum margin of 3 to 5 mm5 to 10 mm,6,7 and the treatment can be delivered using orthovoltage photons or electrons.1,5,6,7 Individual shielding with protection of the surrounding normal soft tissues is advised.5,6
Different dose schemes are described, such as: a single dose of 2 to 3 Gy,5 and 2 single fractions of 4.0 Gy on consecutive days, repeated at intervals of 4 weeks to cumulative doses from 24 to 32 Gy.3 Also described is the use of a total dose of 15 to 21 Gy,5,6 or 60 Gy,2 in 5 fractions per week. However, the scheme with a total dose of 30 Gy in 2 separated courses of 15 Gy in 5 fractions/ week is the most reported. The interval between courses varies among authors, with most describing a 6-3,6 to 12-week break.1,4
The described acute side effects of RT are generally mild, including mainly radiation dermatitis, hyperpigmentation and edema.3 Dryness and fibrosis are reported as late side effects.1,3,4,6 There is a theoretical risk of radiation-induced skin cancer, which decreases with the age of the patient at the time of treatment; however, this has not been formally demonstrated.1,4,6 The risk of irradiated bone fracture and foot dystrophia appears to be associated with higher RT doses.2
Conclusions
The underlying cause of PF remains unknown. Although there is no standardized treatment approach, surgical management is often recomended when conservative local therapies fail. However, surgery is rarely effective and often leads to poor functional outcomes and wound complications. In the reported data available, RT seems to be a well-tolerated and effective treatment modality, with good local control and symptomatic benefit. Our results, while anecdotal, coincide with the RT data reviewed. RT should be a first consideration for failure of initial conservative management.
References
- Taylor R, Hatfield P, McKeown S, et al. A review of the use of radiotherapy in the UK for the treatment of benign clinical conditions and benign tumours. The Royal College of Radiologists. Clin Oncol. 2015;1:115.
- De Bree E, Zoetmulder F, Keus R, et al. Incidence and treatment of recurrent plantar fibromatosis by surgery and postoperative radiotherapy. Am J Surg. 2004;187(1):33-38.
- Heyd R, Dorn A, Herkströter M, et al. Radiation therapy for early stages of morbus Ledderhose. Strahlenther Onkol 2010;186(1):24-29.
- Seegenschmiedt MH, Wielpuetz M, Hanslian M, et al. Long-term outcome of radiotherapy for primary and recurrent Ledderhose disease (chapter 50). In: Eaton C, Seegen
- schmiedt MH, Bayat A, Gabbiani G, Werker P, Wach W, eds. Dupuytren’s disease and related hyperproliferative disorders. Springer-Verlag; 2012; 409-27.
- Seegenschmiedt MH, Micke O, Niewald M, et al. DEGRO guidelines for radiotherapy on non-malignant disorders. Part III: hyperproliferative disorders. Strahlenther Onkol. 2015;191:541-548.
- Grenfell S, Borg M. Radiotherapy in fascial fibromatosis: a case series, literature review and considerations for treatment of early-stage disease. J Med Imag Radiat Oncol. 2014;58 641-647.
- Seegenschmiedt MH, Micke O, Muecke R. Radiotherapy for non-malignant disorders: state of the art and update of the evidence-based practice guidelines. Br J Radiol. 2015;88:20150080.
Citation
SC G, B F, J C, M C, M B. Radiation Therapy in the Treatment of Plantar Fibromatosis – Two Clinical Cases of Recurrent Disease. Appl Radiat Oncol. 2021;(3):63-67.
October 5, 2021