Pre-operative chemoradiation for rectosigmoid cancers: Where do we draw the line?
Images
CASE SUMMARY
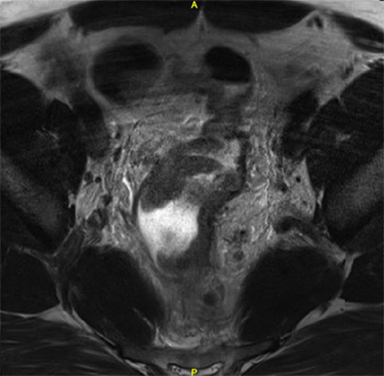
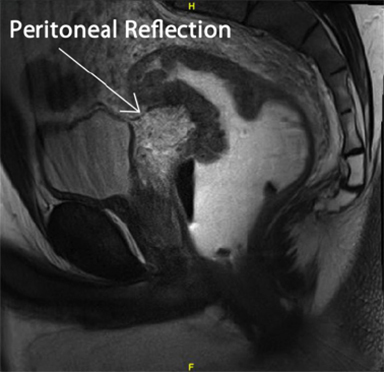
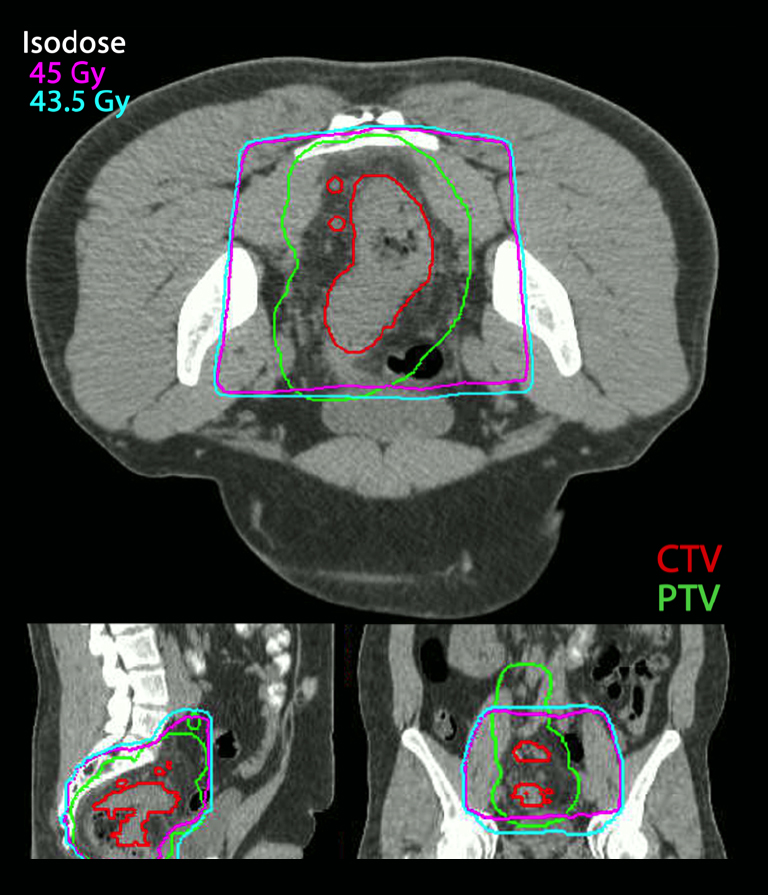
A 39-year-old male presented with a 6-month history of blood in his stools, accompanied by a persistent sensation of fullness and pain in his rectum. He was evaluated with blood work, a colonoscopy, a CT scan of his abdomen and pelvis, and an MRI scan. Serum carcinoembryonic antigen (CEA) was 1.3 ng/ml. The colonoscopy revealed a mass extending from 9 cm to 16 cm as measured from the anal verge. The mass was circumferential, constricting and friable with some bleeding. A biopsy revealed moderately differentiated adenocarcinoma. On clinical radiological correlation, the mass was found to extend into the sigmoid colon but was centered below the peritoneal reflection (Figure 1). He was treated with neoadjuvant chemoradiation to 50.40 Gy in 28 fractions and concurrent Capecitabine 825 mg/m2 twice daily for 5 days a week during radiation (Figures 2 and 3). This was to be followed by total mesorectal excision after 4 to 6 weeks. The patient proceeded for sperm banking before initiating chemoradiation.
IMAGING FINDINGS
MRI of his pelvis revealed a mass located in the upper rectum extending into the sigmoid colon. The mass was noted to be circumferential and involving almost 95% of the luminal circumference (Figue 1). There was significant extra rectral spread noted along with a number of involved nodes throughout the mesorectum. A CT scan of the abdomen and pelvis was negative for liver metastases.
DIAGNOSIS
Locally advanced adenocarcinoma of the rectosigmoid (T3N2bM0, stage IIIC).
DISCUSSION
The current paradigm for treating locally advanced rectal cancer is neoadjuvant chemoradiotherapy followed by total mesorectal excision. A number of studies have shown that this approach for the treatment of advanced rectal cancers has a number of benefits, which include a possible increase in overall survival, a definite increase in the disease-free survival, an increase in local control, and an increase in the rate of sphincter salvage.1
Whether this paradigm also applies to high rectal or rectosigmoid cancers is not known. We describe a case of high rectal cancer that was treated with neoadjuvant chemoradiotherapy, and explore the benefits of this approach.
The inclusion criteria for the German and Dutch trials studying neoadjuvant chemoradiation and neoadjuvant radiation, respectively, provide guidance in this respect. While the German trial included tumors up to 16 cm from the anal verge, the Dutch trial included tumors up to 15 cm from the anal verge and below S1-S2.2 However, only 21% and 32% of patients included in the preoperative and postoperative chemoradiotherapy groups, respectively, for the German rectal cancer trial had tumors more than 10 cm from the anal verge. 1 In the Dutch trial, for patients with tumors located from 10 to 15 cm from the anal verge, the difference in local recurrence rate was not significantly different (3.8% for surgery alone vs. 1.3% for multi-modality treatment).The Swedish rectal cancer trial included patients with tumors below the sacral promontory,3 and the European Organization for the Research and Treatment of Cancer (EORTC) rectal cancer trial included patient with tumors up to 15 cm from the anal verge.4 Notably, all 4 arms of the EORTC trial had less than 10% of patients with tumors > 10 cm from the anal verge. The National Comprehensive Cancer Network (NCCN) 2014 guidelines define true rectal cancers as those within 12 cm from the anal verge.5 Hence, there is considerable debate as to what constitutes an upper rectal cancer vs. a true sigmoid colon cancer, and as to whether preoperative therapy benefits this subset of cancers or not. Mulcahy summarizes these observations as well.6
Measuring rectal tumors from the anal verge is difficult for high rectal/rectosigmoid cancer, as is correlation of these measurements with imaging findings. A useful anatomical landmark is the peritoneal reflection. Tumors below the peritoneal reflection may be called rectal cancers, while those above may be called sigmoid cancers. MRI scans help define the peritoneal reflection and also have the ability to predict a negative radial margin.7 Apart from the loco regional control benefit noted with preoperative therapy, the 2 main advantages of such therapy are to increase the rate of sphincter preservation, and optimally cytoreduce the disease, enabling easier and margin-negative resections. While the first reason is not an issue with high rectal cancers, the argument can be made that optimal preoperative therapy may help greatly in reducing R1 resections for locally advanced rectal cancers, as demonstrated in a retrospective review of T3-T4 high rectal cancers by O’Neill.8
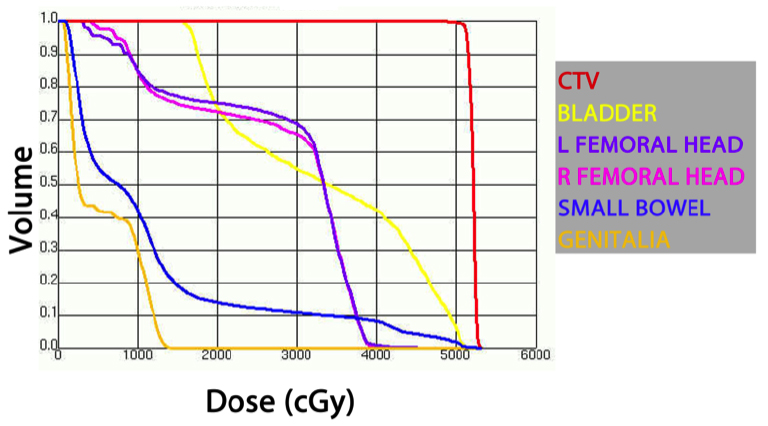
Additionally, the latest analysis from the German rectal cancer trial reported superior disease-free survival and a decreased distant metastases rate for patients achieving high tumor regression after long-course preoperative chemoradiation, further underscoring the value of this treatment.9 Lastly, there is increasing concern to limit the late effects of radiation to normal tissues, particularly the small bowel. We believe that a gentler chemoradiation fractionation (at 1.8 Gy per fraction instead of 5 Gy per fraction3) achieves reasonable cytoreduction of the tumor while avoiding considerable toxicity to the small bowel for high rectal tumors. This is even more so the case with techniques like 3D conformal therapy and intensity-modulated radiotherapy (IMRT).
CONCLUSION
While there is no clear consensus as to the management of high rectal/rectosigmoid cancers, the current approach for tumors located below the peritoneal reflection is a course of neoadjuvant chemoradiation, especially for locally advanced tumors encroaching upon the mesorectal fascia in which preoperative MRI suggests the possibility of an R1 resection. Tumors above the peritoneal reflection and truly sigmoid cancers may be treated as primary colon cancers with surgery and adjuvant systemic therapy per histology and stage.
REFERENCES
- Sauer R, Liersch T, Merkel S, et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30:1926-1933.
- Kapiteijn E, Marijnen C, Nagtegaal I, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345:638-646.
- Improved survival with preoperative radiotherapy in resectable rectal cancer. Swedish Rectal Cancer Trial. N Engl J Med. 1997;336:980–987.
- Bosset JF, Collette L, Calais G, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355:1114–1123.
- NCCN Clinical Practice Guidelines in Oncology, Rectal cancer, version 3.2014, http://www.nccn.org/professionals/physician_gls/PDF/rectal.pdf.
- Mulcahy M. Radiotherapy for cancer of the rectum: which patients stand to benefit? Gastrointest Cancer Res. 2009;3(2):81-83.
- Burton S, Brown G, Daniels I, et al. MRI identified prognostic features of tumors in distal sigmoid, rectosigmoid, and upper rectum: treatment with radiotherapy and chemotherapy. Int J Radiat Oncol Biol Phys. 2006;65:445-451.
- O’Neill B, Brown G, Wotherspoon A, et al. Successful downstaging of high rectal and recto-sigmoid cancer by neo-adjuvant chemo-radiotherapy. Clin Med Oncol. 2008;2:135-144.
- Fokas E, Liersch T, Fietkau R, et al. Tumor regression grading after preoperative chemoradiotherapy for locally advanced rectal carcinoma revisited: updated results of the CAO/ARO/AIO-94 trial. J Clin Oncol. 2014; epub ahead of print.