Neoadjuvant chemoradiotherapy for laryngeal synovial sarcoma
Images
CASE SUMMARY
Synovial cell sarcoma of the head and neck comprise less than 0.1% of all head and neck cancers.1 The first case of head and neck synovial sarcoma was described in 1954 by Jernstrom. The larynx is the least common site of occurrence of synovial sarcomas, making laryngeal synovial sarcoma an extremely rare disease entity.
Due to the paucity of cases, the optimal treatment of laryngeal synovial sarcoma is yet to be established. Based on reports, wide local excision with or without adjuvant radiation therapy is usually the first treatment of choice. The role of chemotherapy remains controversial.
We present a patient diagnosed with unresectable laryngeal synovial sarcoma who underwent neoadjuvant radiation therapy with concurrent weekly chemotherapy.
The patient is a 22-year-old Filipino man, ECOG 0, with no known medical comorbidities or vices. He presented with a 7-month history of foreign body sensation in his throat. Thereafter, he developed hoarseness, pooling of saliva, and solid-food dysphagia. He then experienced difficulty breathing and sought consult.
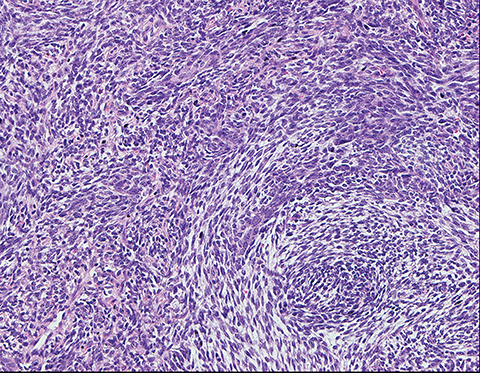
The patient underwent tracheostomy, nasogastric tube insertion and simple supraglottic excisional biopsy for what was thought to be a benign process. Pathologic examination demonstrated a malignant spindle cell tumor (Figure 1).
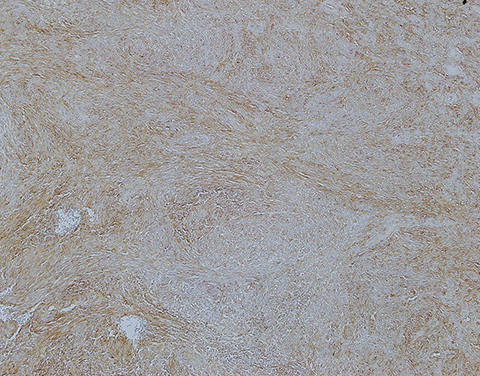
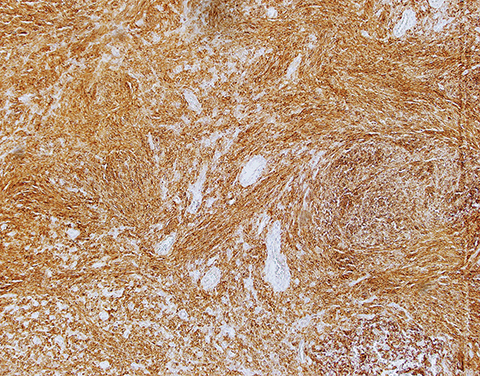
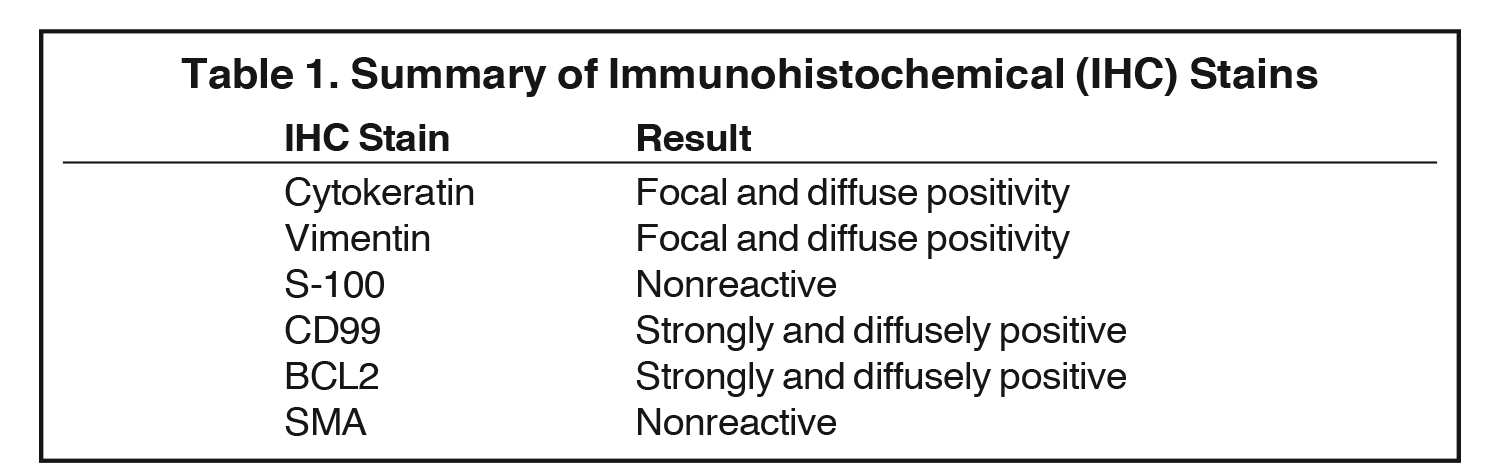
Although a sarcomatoid squamous cell carcinoma was initially included in the histologic differential diagnosis, the patient’s age and the characteristic appearance in the hematoxylin and eosin (H&E) stain favored the diagnosis of synovial sarcoma. Multiple immuno-stains were done for confirmation (Table 1, Figure 2).
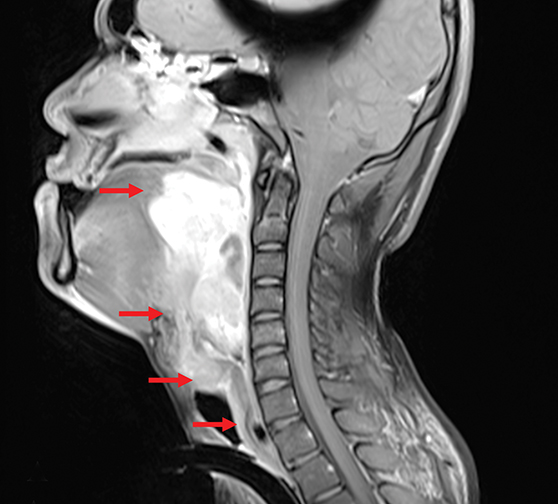
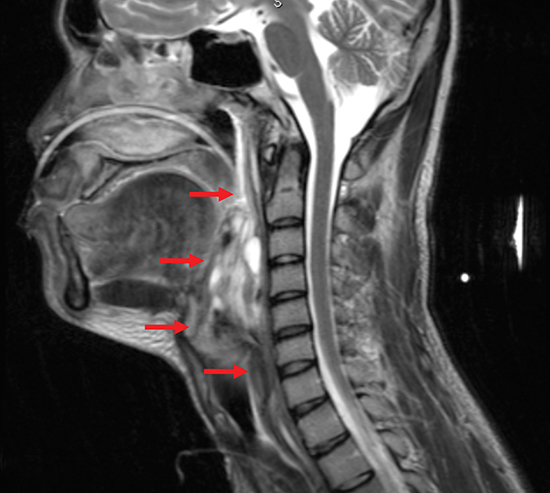
Head and neck magnetic resonance imaging (MRI) with gadolinium revealed a complex 10.4 × 7.6 × 5.1 cm (CC × W × AP) mass occupying the posterior oral cavity, oropharynx and larynx (Figure 3A). Few subcentimeter cervical lymph nodes were also seen. A 0.6-cm pulmonary nodule in the left upper lobe was noted on chest computed tomography (CT), deemed to be inflammatory.
Direct laryngoscopy under general anesthesia was then attempted. However, during the procedure, profuse bleeding (~500 cc) from the mass was encountered. The procedure was terminated after achieving adequate hemostasis.
In light of the tumor’s friability and propensity to bleed, and after careful review of the scarce literature regarding this rare malignancy, the head and neck tumor board decided to pursue neoadjuvant radiation therapy with concurrent chemotherapy consisting of 30 mg of weekly intravenous doxorubicin. Surgery was planned for after assessment of response to neoadjuvant treatment, should the sarcoma become resectable.
During initial consult with radiation oncology, a foul-smelling, fungating mass was visualized in the posterior oropharynx, while a subcentimeter cervical lymph node was palpable on the left submandibular area.
Three-dimensional conformal radiation therapy (3DCRT) was delivered using a linear accelerator with a 6-MV photon beam. The gross tumor volume (GTV) included all gross disease on physical examination and contrast- enhanced MRI. The clinical target volume (CTV) was generated by adding 2 cm to the GTV, shaving off bone and muscle. The planning target volume (PTV) was the CTV plus 0.5 cm. Bilateral neck levels 2 to 4 were electively covered due to the propensity of synovial sarcoma to metastasize to the lymphatics. The plan was to administer 200 cGy daily fractions for 7 weeks to a total dose of 7000 cGy to the PTV. However, by the sixth week, the patient developed excessive secretions that prevented him from lying supine for his radiation therapy treatment in spite of suctioning, anticholinergic medications and sedation. Treatment was stopped at 6 weeks, having delivered a total dose of 6000 cGy to the tumor and bilateral neck.
There was grade 3 radiation dermatitis, which eventually healed after local wound care. The chemotherapy course was unremarkable.
Six weeks after treatment, repeat MRI with gadolinium showed a decrease in the mass to 7.8 × 4.5 × 4.8 cm (CC × W × AP) with interval resolution of its oropharyngeal, oral cavity and glottic extensions (Figure 3B).
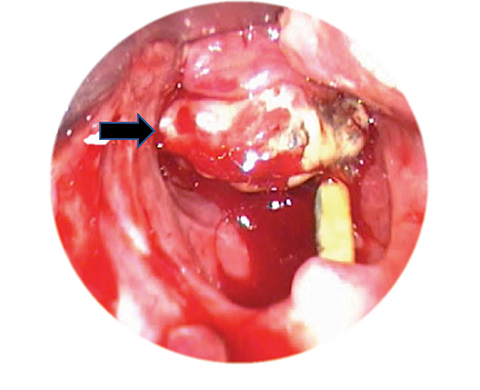
The planned stepwise surgical resection then commenced beginning with excision of the supraglottic mass via transoral approach (Figure 4). Completion laryngectomy was done 3 weeks after. No untoward events were noted intraoperatively or postoperatively. The final histopathology report showed monophasic synovial sarcoma, 4 cm in greatest dimension, with 60% tumor necrosis involving the left glottis and supraglottis. No lymphovascular space or perineural invasion was noted. All surgical margins were clear, ranging 0.7 to 2 cm. No further adjuvant treatment was warranted. Thereafter, close follow-up with clinical examination and imaging as per National Cancer Center Network (NCCN) guidelines will ensue.
DISCUSSION
Laryngeal synovial sarcoma is a rare head and neck malignancy with approximately 20 cases reported in the literature.2 The most reported site of presentation is the parapharyngeal space, while the larynx is the least frequent site of occurrence.
The term “synovial” refers to the tumor’s microscopic resemblance to the synovium, but these tumors do not in fact arise from synovial structures. Immunohistochemistry plays a major role in diagnosing synovial sarcoma, of which there are 2 main variants: monophasic and biphasic. Molecular testing can also be done, as synovial sarcoma harbors a specific chromosomal translocation, t(X:18) (p11.2; q11.2). This leads to fusion between the SYT gene on chromosome 18 and the SSX1 and SSX2 genes on the X chromosome.3
There is very limited literature on head and neck synovial sarcomas, with the majority being case reports and single-institution studies. The management of these rare tumors, and of head and neck sarcomas in general, has largely been extrapolated from studies on the more common extremity sarcomas.
The primary treatment modality for head and neck sarcomas is surgery, with adjuvant radiation therapy reserved for large tumors, high-grade sarcomas and positive margins.4 According to NCCN guidelines, the primary goal in oncologic resection of head and neck sarcomas is complete en bloc excision, while minimizing functional and aesthetic complications. There is no consensus regarding margin width, but on average, a 2-cm margin is considered acceptable.5
However, the unique anatomic considerations of the head and neck limit the ability to obtain negative surgical margins, which may explain the higher local recurrence rate and lower disease-specific survival of head and neck sarcomas.6
The role of radiation therapy in the treatment of head and neck sarcomas came out of the high rates of local recurrence following inadequate surgery. Adjuvant radiation therapy is usually given to patients with high-grade tumors of any size. Studies on preoperative vs postoperative radiation therapy for head and neck sarcomas are limited; thus, the optimal timing of radiation therapy is yet to be determined.7
Adjuvant radiation therapy has been shown to benefit R1 or R2 surgeries the most, with local control rates increasing from 25% to 54%.8 Definitive radiation therapy alone has no role in primary treatment of head and neck sarcomas.9 A report on 112 patients with unresectable soft-tissue sarcomas who underwent radiation therapy, of which 3% were synovial sarcomas, used a median radiation dose of 64 Gy. Twenty percent of patients received chemotherapy. Five-year local control was 51% for tumors < 5 cm and 9% for tumors > 10 cm (p < 0.001). Doses of > 63 Gy resulted in better local control, disease-free survival and overall survival compared to lesser doses, while doses of 68 Gy or more led to more complications.10
Although surgery followed by adjuvant radiation therapy is standard treatment for soft-tissue sarcomas, it is associated with high rates of local recurrence particularly in patients with head and neck sarcoma, incompletely resected sarcomas and large soft-tissue sarcomas. Hence, studies investigating treatment alternatives—including the addition of chemotherapy and concurrent chemoradiotherapy—have been pursued.
Patients with head and neck synovial sarcoma have a high likelihood of harboring distant metastasis; therefore, more effective systemic therapy is necessary. While synovial sarcomas are known to be chemosensitive tumors, the role and benefit of chemotherapy in the treatment of soft-tissue sarcomas in general is not as well defined as that of radiation therapy. There is even less literature concerning chemotherapy in head and neck sarcomas, more so the case for neoadjuvant chemoradiation for head and neck cancers. This approach aims to primarily improve local control without compromising function, and to abate micrometastatic disease early on.
A retrospective study of 29 cases of head and neck sarcoma, including synovial sarcoma, showed longer mean survival with chemoradiotherapy treatment (71.5 months) compared to without (42.3 months). Age was the only statistically significant survival predictor.11
Our patient with primary laryngeal synovial sarcoma represents a rare case that was treated with neoadjuvant chemoradiation therapy. This case demonstrated how neoadjuvant chemoradiation therapy successfully facilitated oncologic surgical resection of an initially unresectable laryngeal synovial sarcoma. Close patient follow-up will continue to monitor disease outcomes.
CONCLUSION
Head and neck synovial sarcoma is a rare disease entity posing several diagnostic and treatment challenges. Hence, a multidisciplinary team approach consisting of head and neck surgeons, radiation oncologists and medical oncologists is optimal. In the absence of evidence-based guidelines and randomized prospective studies, management decisions for head and neck synovial sarcoma should be individualized. Novel approaches to treatment should be investigated for their potential to improve patient outcomes.
REFERENCES
- Sturgis EM, Potter BO. Sarcomas of the head and neck region. Curr Opin Oncol. 2003;15(3):239-252.
- Narayanan G, Baby A, Somanathan T, Konoth S. Synovial sarcoma of the larynx: report of a case and review of literature. Case Rep Otolaryngol. 2017;1-6. doi:10.1155/2017/6134845.
- Tos AP, Sciot R, Giannini C, et al. Synovial sarcoma of the larynx and hypopharynx. Ann Otol Rhinol Laryngol. 1998;107(12),1080-1085. doi: 10.1177/000348949810701215.
- Mucke T, Mitchell DA, Tannapfel A, et al. Outcome in adult patients with head and neck sarcomas: 10-year analysis. J Surg Oncol. 2010;102(2):170-174.
- National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology (NCCN guidelines) soft tissue sarcoma. http://www.nccn.org/
- professionals/physician_gls/pdf/sarcoma.pdf.
- De Bree R, van der Waal I, de Bree E, Leemans CR. Management of adults of tissue sarcomas of the head and neck. Oral Oncol. 2010;46(11):786-790.
- Shellenberger TD, Sturgis EM. Sarcomas of the head and neck region. Curr Oncol Rep. 2009;11(2),135-142. doi:10.1007/s11912-009-0020-8.
- Barker Jr JL, Paulino AC, Feeney S, McCulloch T, Hoffman H. Locoregional treatment for adult soft tissue sarcomas of the head and neck: an institutional review. Cancer J. 2003;9(1):49-57.
- Eeles RA, Fisher C, A’Hern RP, et al. Head and neck sarcomas: prognostic factors and implications for treatment. Br J Cancer. 1993;68(1):201-207.
- Kepka L, Delaney TF, Suit HD, Goldberg SI. Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys. 2005;63(3),852-859. doi:10.1016/j.ijrobp. 2005. 03.004
- Barosa J, Ribeiro J, Afonso L, Fernandes J, Monteiro E. Head and neck sarcoma: analysis of 29 cases. Euro Ann Otorhinolaryngol, Head Neck Dis. 2014;131(2),83-86. doi:10.1016/j.anorl.2012.11.007.
Citation
EM C, JP A, BG DG, ACF L, AR V. Neoadjuvant chemoradiotherapy for laryngeal synovial sarcoma. Appl Radiat Oncol. 2019;(1):45-48.
March 18, 2019